The Korean Otolaryngological Society has initiated legal proceedings against marketers promoting unproven tinnitus cures. Clinical review indicates these agents function primarily as sedatives, masking symptoms rather than repairing auditory nerve damage. Patients risk dependency even as the underlying neurological condition remains untreated.
This regulatory crackdown underscores a critical public health vulnerability where patient desperation intersects with aggressive pharmaceutical marketing. While the immediate news originates from East Asia, the implications resonate globally, particularly in markets where off-label prescribing for chronic otologic conditions lacks stringent oversight. For the millions suffering from chronic subjective tinnitus, the distinction between palliative sedation and curative intervention is not merely semantic; We see a matter of long-term neurological safety and quality of life.
In Plain English: The Clinical Takeaway
- No Universal Cure Exists: Currently, no medication is FDA-approved to specifically cure chronic tinnitus; treatments focus on management.
- Sedation is Not Healing: Drugs that make you sleepy may hide the noise temporarily but do not fix the ear or brain pathways causing it.
- Regulatory Action Protects Patients: Legal moves against false advertising prevent vulnerable patients from wasting money on ineffective, potentially harmful drugs.
The Neurobiology of Auditory Phantom Perception and Pharmacological Interference
Tinnitus is not a disease itself but a symptom of underlying auditory pathway dysfunction, often originating in the dorsal cochlear nucleus of the brainstem. When hair cells in the inner ear are damaged, the brain attempts to compensate for the lack of sensory input by increasing neural gain, resulting in the perception of sound where none exists. Pharmacological interventions often target the central nervous system to dampen this hyperactivity.
Yet, many compounds marketed as “miracle cures” operate through non-specific mechanisms of action, primarily involving gamma-aminobutyric acid (GABA) modulation. While this induces sedation and may reduce the patient’s awareness of the ringing, it does not reverse the synaptic plasticity changes responsible for the condition. This distinction is vital; treating the symptom via sedation leaves the neuroplastic maladaptation intact, potentially leading to tolerance and withdrawal issues without resolving the core pathology.
Global Regulatory Standards for Otologic Drug Claims
The regulatory landscape for otologic medications varies significantly between jurisdictions. In the United States, the Food and Drug Administration (FDA) requires rigorous double-blind placebo-controlled trials to substantiate claims of efficacy. Currently, no drug holds specific FDA approval for the cure of tinnitus, though some are used off-label. In contrast, other regions may have different thresholds for supplement classification versus pharmaceutical regulation.
The recent legal actions in Korea mirror enforcement trends seen by the European Medicines Agency (EMA) regarding unauthorized health claims. When a manufacturer claims a product is a “special effect drug” (特効薬) without Phase III clinical trial data, it violates consumer protection laws designed to prevent medical fraud. This geo-epidemiological bridging highlights that patient access to safe care depends on robust regulatory bodies capable of distinguishing between evidence-based medicine and predatory marketing.
Evaluating Efficacy Through Double-Blind Placebo-Controlled Metrics
To understand why these advertisements are problematic, one must examine the statistical significance of existing treatments. Most studies involving benzodiazepines or antidepressants for tinnitus show high variability in patient-reported outcomes. The placebo effect in tinnitus trials is notoriously high, often exceeding 30%, making subjective claims of “cures” scientifically unreliable without objective audiometric confirmation.
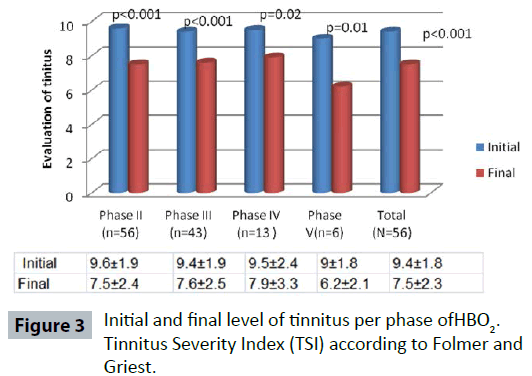
Funding transparency is also crucial. Research funded by pharmaceutical entities often reports higher efficacy rates than independent studies. Patients must scrutinize whether the underlying research supporting a treatment was conducted by independent academic institutions or sponsored by the manufacturer. The lack of disclosed funding sources in many direct-to-consumer advertisements remains a primary vector for medical misinformation.
| Drug Class | Proposed Mechanism | Clinical Efficacy (Evidence Level) | Common Adverse Effects |
|---|---|---|---|
| Benzodiazepines | GABA Receptor Agonist | Low (Symptom Masking Only) | Sedation, Dependency, Cognitive Decline |
| Tricyclic Antidepressants | Serotonin/Norepinephrine Reuptake Inhibition | Moderate (Comorbidity Management) | Dry Mouth, Weight Gain, Cardiac Risks |
| Anticonvulsants | Sodium Channel Blockade | Low/Conflicting | Dizziness, Fatigue, Rash |
| Placebo | Psychological Expectation | High (Subjective Relief) | None |
Expert consensus emphasizes the need for multimodal management rather than pharmacological reliance.
“Current evidence does not support the use of medication as a primary curative strategy for tinnitus. Management should focus on cognitive behavioral therapy and sound enrichment to facilitate habituation,”
states the Clinical Practice Guideline issued by the American Academy of Otolaryngology-Head and Neck Surgery. This position reinforces that sedation is not a therapeutic endpoint.
Contraindications & When to Consult a Doctor
Patients considering any pharmacological intervention for tinnitus must be aware of specific contraindications. Individuals with a history of substance use disorder should avoid sedative-hypnotics due to the high risk of dependency. Elderly patients are at increased risk for falls and cognitive impairment when using centrally acting agents.

Immediate medical consultation is warranted if tinnitus presents unilaterally (in one ear only), pulsates in rhythm with the heartbeat, or is accompanied by sudden hearing loss or vertigo. These symptoms may indicate underlying vascular anomalies or acoustic neuromas requiring imaging and specialized intervention rather than empirical drug therapy. Never initiate off-label medication without a comprehensive audiological evaluation.
The Future Trajectory of Tinnitus Therapeutics
The legal actions taken this week signal a shift toward stricter accountability in health advertising. As research into bimodal neuromodulation and targeted drug delivery systems advances, the hope is to move beyond sedation toward genuine neural repair. Until then, patients must remain vigilant against claims that sound too excellent to be true. The path to relief lies in evidence-based management, not in pills that merely induce sleep.
References
- American Academy of Otolaryngology-Head and Neck Surgery. Clinical Practice Guideline: Tinnitus.
- National Center for Biotechnology Information. Tinnitus: Pathophysiology and Treatment Options.
- U.S. Food and Drug Administration. Warning Letters on Unauthorized Health Claims.
- World Health Organization. World Report on Hearing.
- JAMA Otolaryngology–Head & Neck Surgery. Pharmacological Interventions for Tinnitus.

