
Historical Exhibit Showcases Moon Landing and Civil War History
A Utah man is assembling a trove of over 100 rare historical newspapers—from the Civil War to the moon landing—to launch an exhibit in Spanish Fork on June 6, 2026. ... Read More

Saturday Edition
Stay updated with Archyde – your source for breaking news, global headlines, economy, entertainment, health, technology, and sports. Fresh stories daily.

A Utah man is assembling a trove of over 100 rare historical newspapers—from the Civil War to the moon landing—to launch an exhibit in Spanish Fork on June 6, 2026. ... Read More
Continuous Coverage
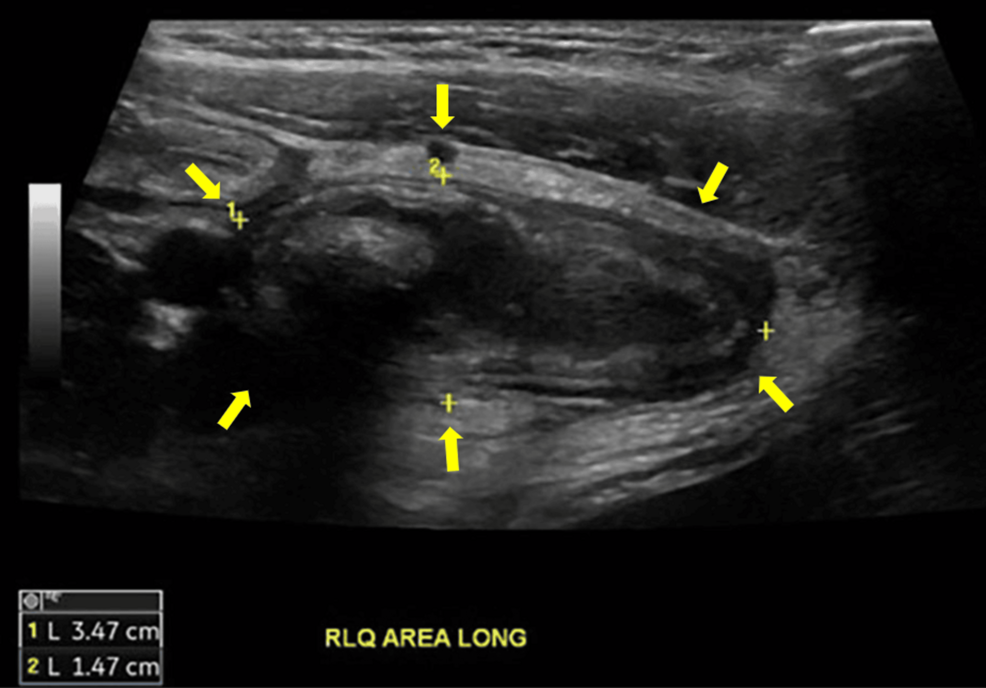
A rare case of Amyand hernia mimicking acute appendicitis highlights diagnostic challenges in pediatric surgery, emphasizing the need…

Sir Alex Ferguson reportedly messaged PSG president Nasser Al-Khelaifi following the 2026 Champions League final, labeling Arsenal’s tactical…

Ireland’s summer festival season is gearing up to be a cultural and economic powerhouse, with Slane Castle’s headline-grabbing…

Elon Musk’s SpaceX IPO Drives Saudi Al-Mamlaka Holding to 10-Year High Elon Musk’s SpaceX IPO sparks a 22.3%…

On May 31, 2026, Hong Kong’s Chief Executive, John Lee, announced a renewed economic partnership with Central Asian…

In the quiet, high-altitude meadows of the Swiss Alps, the rhythm of the seasons is shifting. For centuries,…
Global Affairs

A 2023 Volkswagen California Edition TDI 150 DSG, currently listed on the French marketplace Leboncoin, highlights the shifting…
Markets And Money

Platinum-group metal spikes 10x gold as geopolitical tensions tighten supply. A rare platinum-group metal (PGM) now trades at…
Digital Culture

Finansavisen reports on Ny Solon Eiendom’s emergency measures, revealing systemic risks in real estate tech ecosystems. This analysis…
Science And Wellbeing

Taiwan’s Tongxiao Township is launching a one-stop, free health screening program this week, targeting a population with a…
Screen And Sound

On May 31, 2026, skywatchers will witness a rare “blue micromoon”—a full moon that is both a second…
Fixtures And Form

Breaking the CPBL’s 3-game series revenue record, the Fubon Guardians’ “攻蛋” event generated 1 billion NTD in ticket…