
Debunking the Myth of Useless Degrees
The debate over “useless” degrees ignores the fundamental economic reality: higher education is a capital investment. In 2026, the market measures value not by the credential itself, but by the ... Read More
Saturday Edition
Stay updated with Archyde – your source for breaking news, global headlines, economy, entertainment, health, technology, and sports. Fresh stories daily.

The debate over “useless” degrees ignores the fundamental economic reality: higher education is a capital investment. In 2026, the market measures value not by the credential itself, but by the ... Read More
Continuous Coverage

The Devil Wears Prada 2 arrives in theaters this weekend, reviving the high-stakes world of Miranda Priestly for…

Oklahoma Football’s 2026 win total has been set at a shocking 7.5, signaling a skeptical market outlook on…

Supermassive black holes (SMBHs) generate the universe’s most powerful cosmic jets by accelerating plasma via intense magnetic fields…
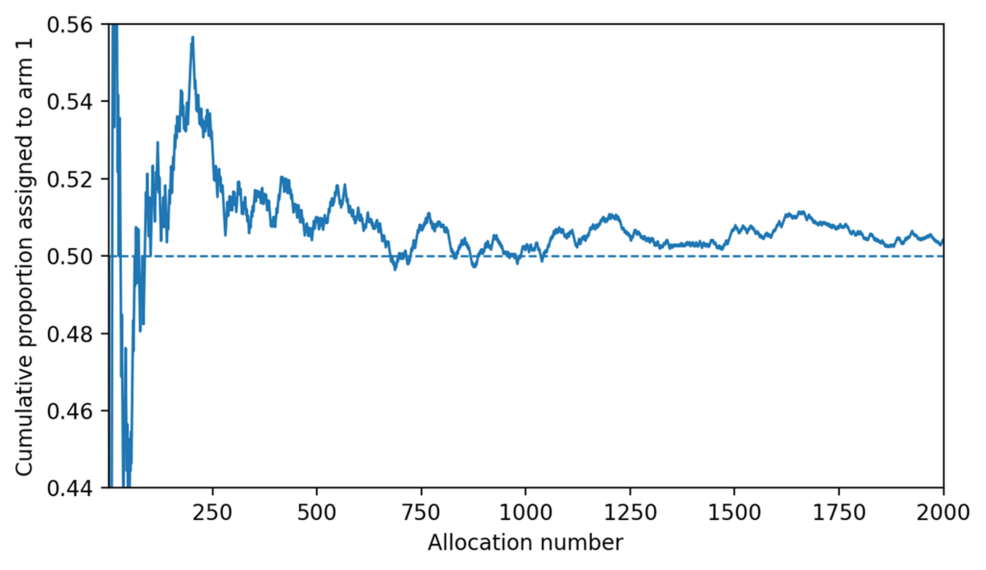
A new Python-based binary allocation prototype published in Cureus introduces a transparent, audit-ready framework for clinical trial randomization.…

The Andaman Sea is a graveyard of dreams, a stretch of turquoise water that masks a brutal, churning…
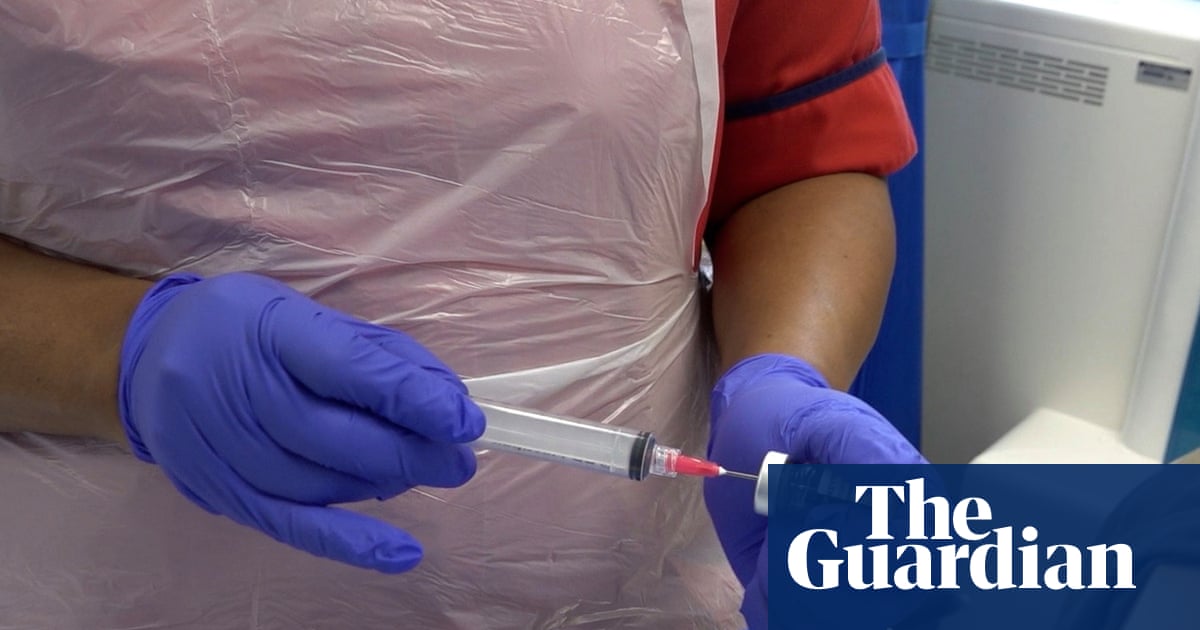
Thousands of cancer patients in England are gaining access to a breakthrough personalized immunotherapy vaccine. This treatment trains…
Global Affairs
The United States and Iran are locked in a high-stakes geopolitical gamble as Tehran sets a one-month deadline…
Markets And Money
/https://i.s3.glbimg.com/v1/AUTH_59edd422c0c84a879bd37670ae4f538a/internal_photos/bs/2026/A/4/xoiEohRsuNWx588AOLMg/captura-de-tela-2026-04-30-203104.png)
The Brazilian government is launching a new debt renegotiation package on Monday, May 4, 2026, expanding the “Desenrola”…
Digital Culture

The Insel Gruppe, Switzerland’s largest healthcare provider, is quietly rewriting the playbook for hospital IT—by deploying Epic’s KISS…
Science And Wellbeing

France is restructuring its 2026 primary care payment model, transitioning toward a “prevention economy.” General practitioners (GPs) will…
Screen And Sound

A critical piece of Liverpool’s maritime history linked to the Beatles is facing demolition as ownership disputes and…
Fixtures And Form

The 2026 HSBC BWF World Tour Super 750 Yonex French Open arrives in Paris from October 20–25, 2026,…