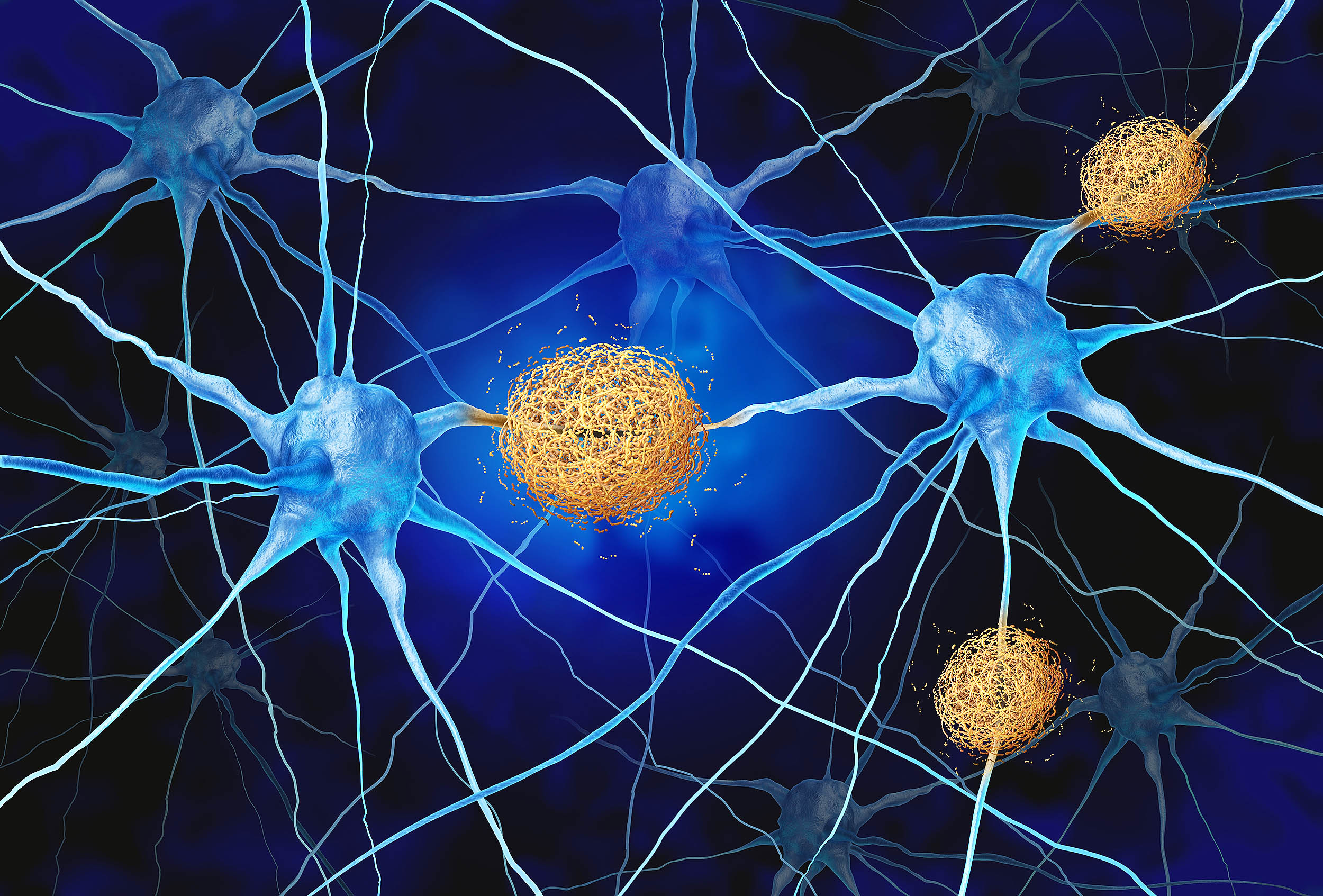
A study published this week in the Proceedings of the National Academy of Sciences reveals that brain immune cells, known as microglia, may actively generate Alzheimer’s plaques rather than clearing them. This finding challenges current therapeutic strategies that aim to boost immune activity, suggesting that such interventions could inadvertently accelerate neurodegeneration in early-stage patients.
For decades, the medical consensus viewed microglia as the brain’s janitors—cells that patrol neural tissue to sweep away toxic debris. However, new research led by Professor Joost Schymkowitz at the VIB biotech institute in Belgium indicates a darker reality: under specific conditions, these cells may act as architects of the disease. By reshaping loose amyloid-beta proteins into hard, sticky fibers, microglia can inadvertently seed the very plaques they were meant to destroy. This discovery forces a critical re-evaluation of drug development pipelines, particularly for monoclonal antibodies designed to stimulate immune clearance.
In Plain English: The Clinical Takeaway
- The “Janitor” Myth: Scientists previously believed brain immune cells only cleaned up waste; we now know they can accidentally manufacture the waste (plaques) themselves.
- Treatment Risks: Medications that simply “boost” the immune system in the brain might make early Alzheimer’s worse, not better, by fueling plaque production.
- Early Detection: The study suggests inflammation starts before plaques are visible, meaning future treatments must target this earlier “distress signal” window.
The Mechanism of Action: From Cleanup to Construction
The core of this discovery lies in the mechanism of action—the specific biochemical process by which a drug or biological entity produces an effect. In this case, the biological entity is the microglia. In a healthy brain, these cells engulf amyloid-beta, a protein fragment that, if left unchecked, aggregates into plaques. However, the VIB study demonstrates that inside the microglia, this protein is not merely digested. Instead, This proves structurally reshaped into “seed” fibers.
When these cells release the fibers back into the extracellular space, they act as a template. Much like a crystal forming around a seed in a solution, these cell-made fibers prompt surrounding dissolved proteins to stick to them, rapidly accelerating plaque growth. This process, known as nucleation, explains why plaque buildup often spreads so aggressively once it begins. Crucially, the fibers generated by these cells more closely resemble the structures found in actual patient brains than the synthetic fibers typically created in laboratory vials, lending high clinical validity to the model.
The Therapeutic Paradox: When Defense Becomes Damage
This finding lands in a complex regulatory landscape. Currently, the FDA and EMA have approved therapies like lecanemab and donanemab, which utilize antibodies to clear amyloid plaques. Some experimental approaches have also sought to hyper-activate microglia to enhance this clearance. The new data suggests a contraindication for such “booster” strategies in the earliest phases of the disease.
If microglia are the source of the seeds, stimulating them before the plaques are fully formed could be counterproductive. It creates a therapeutic paradox: the very cells recruited to defend the brain may be feeding the pathology. This necessitates a shift toward modulation rather than activation. Future clinical trials must distinguish between the “protective” phenotype of microglia (which clears debris) and the “disease-associated” phenotype (which seeds plaques).
“For a long time, we’ve studied amyloid plaques in the lab, where they form spontaneously in small vials. Patient-derived structures differ in shape and behavior, so a closer match could sharpen tests of early plaque formation.” — Prof. Joost Schymkowitz, VIB-KU Leuven Center for Brain & Disease Research
Genetic Susceptibility and the TREM2 Connection
The study also illuminates the role of genetics, specifically variants in the TREM2 gene. This gene is essential for microglial survival and function. In families carrying rare variants of TREM2, the risk for Alzheimer’s is significantly elevated. The research indicates that without proper TREM2 function, microglia struggle to manage the protein load, shifting their behavior from protective to pathogenic. This bridges the gap between genetic risk factors and the physical manifestation of the disease, offering a potential biomarker for identifying patients who might react poorly to immune-boosting therapies.
the study observed that these cell-made fibers trigger the aggregation of tau, another protein involved in Alzheimer’s that forms tangles inside neurons. This confirms the “amyloid cascade hypothesis” even as refining it: microglia-driven amyloid seeding appears to be the upstream trigger that eventually pulls tau into the destructive process, tightening the link between early immune response and later cognitive decline.
Comparative Analysis: Microglial Function in Health vs. Disease
| Feature | Healthy Microglial Response | Pathogenic Response (New Findings) |
|---|---|---|
| Primary Action | Phagocytosis (engulfing and digesting debris) | Reshaping and secreting amyloid “seeds” |
| Effect on Amyloid-Beta | Reduces soluble protein levels | Converts soluble protein into insoluble fibers |
| Inflammatory State | Transient, resolved quickly | Chronic, persistent distress signaling |
| Therapeutic Goal | Support clearance mechanisms | Inhibit seeding without stopping clearance |
Contraindications & When to Consult a Doctor
While this research is currently at the pre-clinical and early mechanistic stage, it has immediate implications for patient advocacy and monitoring. There are no new contraindications for existing FDA-approved medications based solely on this report; however, patients participating in clinical trials for “immune-boosting” Alzheimer’s therapies should discuss these findings with their principal investigators.
Who should be vigilant: Individuals with a family history of early-onset Alzheimer’s or known TREM2 variants should remain closely monitored by neurologists. When to seek facilitate: If you or a loved one experiences subtle changes in memory, executive function, or spatial awareness, do not wait for “severe” symptoms. Early intervention remains the most effective strategy, regardless of the specific mechanism of plaque formation. Always consult a board-certified neurologist before making changes to supplementation or treatment regimens.
The Path Forward: Precision Timing
The ultimate value of this study lies in its potential to refine the timing of interventions. The data suggests a narrow window where the immune response switches from protective to harmful. If clinicians can identify biomarkers for this switch—perhaps through advanced PET imaging or cerebrospinal fluid analysis—they could administer therapies that suppress the “seeding” behavior while preserving the “cleaning” behavior. This level of precision medicine represents the next frontier in neurology, moving away from broad-spectrum immune activation toward targeted cellular modulation.
References
- Schymkowitz, J., et al. (2026). “Microglia-mediated nucleation of amyloid-beta fibrils in Alzheimer’s disease.” Proceedings of the National Academy of Sciences (PNAS).
- Alzheimer’s Association. (2025). “2025 Alzheimer’s Disease Facts and Figures.” Alzheimer’s & Dementia.
- U.S. Food and Drug Administration. (2024). “FDA approves new treatment for early Alzheimer’s disease.” FDA.gov.
- National Institute on Aging. (2025). “What Happens to the Brain in Alzheimer’s Disease?” NIA.nih.gov.