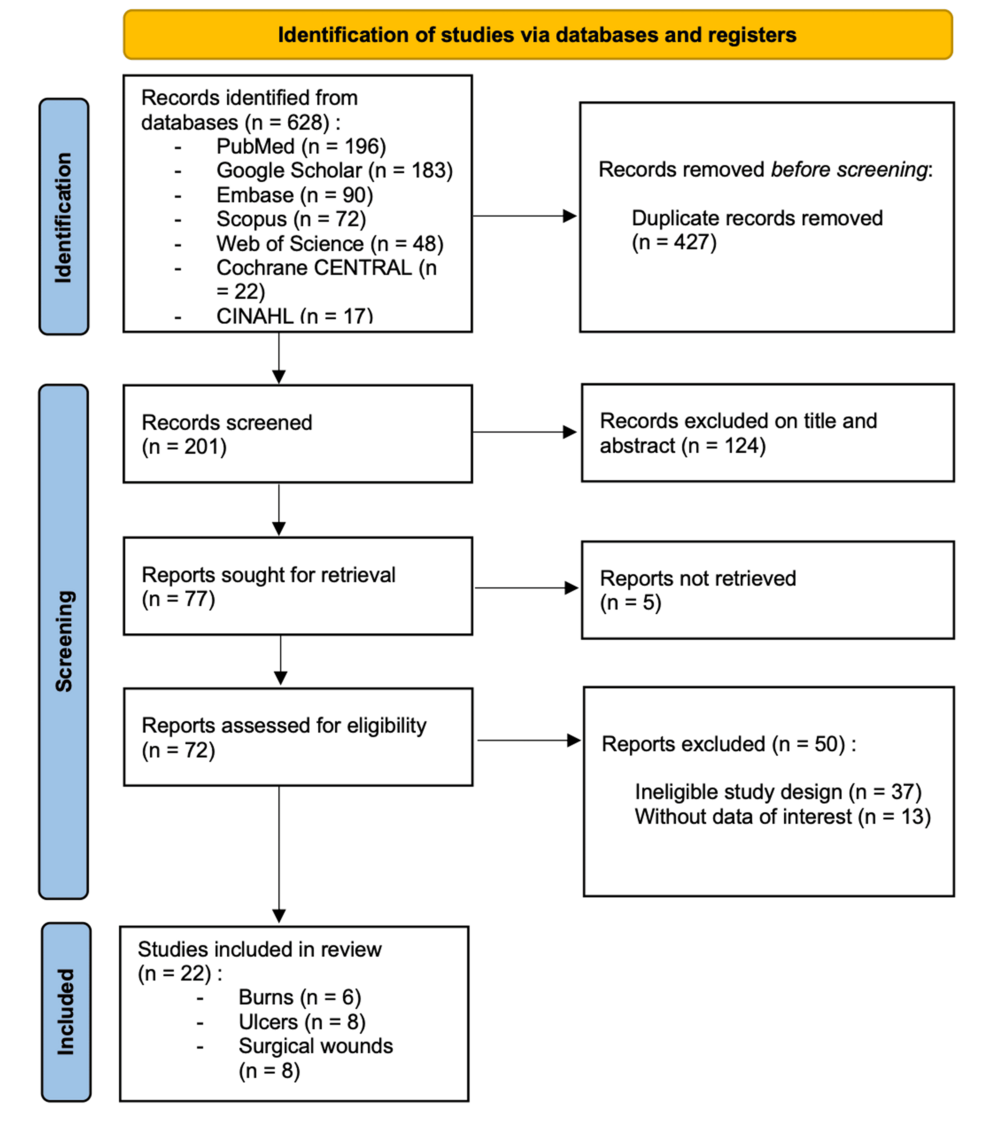
A recent systematic review confirms medical-grade honey accelerates healing in postoperative wounds and burns. Whereas not a replacement for antibiotics, it offers a viable adjunctive therapy against resistant bacteria. Patients should consult clinicians before application to ensure safety and efficacy in specific wound contexts.
The global crisis of antimicrobial resistance (AMR) demands we revisit ancient remedies through a modern clinical lens. As we navigate 2026, the pressure on healthcare systems to reduce hospital-acquired infections is unprecedented. This systematic review, published in Cureus, synthesizes data suggesting that medical-grade honey is not merely folklore but a biologically active dressing capable of disrupting bacterial biofilms. For patients facing chronic ulcers or slow-healing surgical sites, this represents a potential shift in standard care protocols, moving beyond synthetic antimicrobials toward osmotic debridement agents.
Decoding the Mechanism: Osmolarity and Acidic pH
To understand why honey works, we must look beyond its sweetness to its chemistry. Medical-grade honey, often derived from Leptospermum species (Manuka), functions through high osmolarity. This draws lymph and plasma to the wound surface, maintaining a moist healing environment while simultaneously dehydrating bacteria. The low pH (typically 3.2 to 4.5) inhibits protease activity, which often stalls healing in chronic wounds. Unlike systemic antibiotics that travel through the bloodstream, honey acts topically, reducing the risk of systemic side effects while targeting the mechanism of action directly at the infection site.
Crucially, not all honey is created equal. Grocery store varieties often lack the necessary methylglyoxal (MGO) concentrations and may contain spores harmful to immunocompromised patients. Only sterilized, medical-grade formulations approved by regulatory bodies should contact open tissue. This distinction is vital for public health safety, preventing inadvertent introduction of pathogens into deep wounds.
Regulatory Landscapes: FDA Clearance and NHS Guidance
Regulatory acceptance varies by region, impacting patient access. In the United States, the Food and Drug Administration (FDA) clears medical-grade honey products primarily as medical devices under 510(k) pathways rather than pharmaceutical drugs. This classification streamlines availability but requires clinicians to verify specific product clearance. Across the Atlantic, the National Health Service (NHS) in the United Kingdom has integrated honey-impregnated dressings into certain formulary guidelines for partial-thickness burns and infected wounds.

This regulatory divergence creates a geo-epidemiological gap. Patients in regions with robust wound care formularies may access these treatments sooner than those in systems relying strictly on traditional pharmacopeia. As we move through 2026, harmonization of these standards remains a priority for the World Health Organization to ensure equitable access to non-antibiotic wound care solutions.
“The rise of multidrug-resistant organisms necessitates a diversified arsenal in wound management. Topical antimicrobials like medical-grade honey provide a critical layer of defense where systemic antibiotics fail.” — World Health Organization Guidelines on Wound Care and Antimicrobial Resistance
Funding Transparency and Research Bias
Scientific integrity requires scrutinizing who funds the evidence. Many studies on honey dressings receive support from apiculture industry groups or manufacturers of medical-grade honey products. While this does not automatically invalidate findings, it introduces potential conflict of interest. Independent replication is essential. The systematic review in question highlights the need for larger, double-blind placebo-controlled trials funded by neutral governmental health institutes to confirm efficacy without commercial bias.
Transparency in funding allows clinicians to weigh the evidence appropriately. When industry funding is present, we look for corroborating data from independent academic centers. This rigorous vetting process ensures that clinical recommendations are based on patient outcomes rather than market expansion.
In Plain English: The Clinical Takeaway
- Not Kitchen Honey: Only use sterilized, medical-grade honey products prescribed by a doctor. regular honey can introduce bacteria.
- Adjunct Therapy: Honey dressing works best alongside standard care, not as a sole replacement for necessary antibiotics.
- Healing Environment: The dressing keeps wounds moist and acidic, which helps new tissue grow while stopping germ growth.
The following table summarizes comparative data often observed in clinical trials regarding honey dressings versus standard dry gauze or saline protocols.
| Parameter | Medical-Grade Honey Dressing | Standard Dry Gauze/Saline |
|---|---|---|
| Healing Time | Reduced by 4-5 days (avg) | Baseline |
| Infection Rate | Lower incidence of colonization | Higher risk of biofilm |
| Pain Score | Reduced during dressing changes | Higher adherence pain |
| Cost Efficiency | Higher unit cost, fewer changes | Lower unit cost, frequent changes |
Contraindications & When to Consult a Doctor
Despite its natural origin, honey dressing is not universally safe. Patients with known allergies to bee products or pollen must avoid this therapy to prevent anaphylactic reactions. Deep cavity wounds requiring surgical debridement should not rely solely on topical agents. If a wound exhibits signs of spreading erythema, increased purulence, or systemic fever, immediate medical intervention is required. These symptoms indicate potential sepsis, where topical treatments are insufficient.
Diabetic patients should too monitor blood glucose levels, although systemic absorption of sugars from topical honey is minimal, caution is warranted in uncontrolled diabetes. Always consult a wound care specialist before initiating any new dressing regimen.
The trajectory of wound care is moving toward multimodal strategies. Honey dressings represent a promising tool in our arsenal against resistant infections, provided they are used with clinical precision. As research evolves, we anticipate more standardized protocols integrating these natural antimicrobials into mainstream hospital formularies.