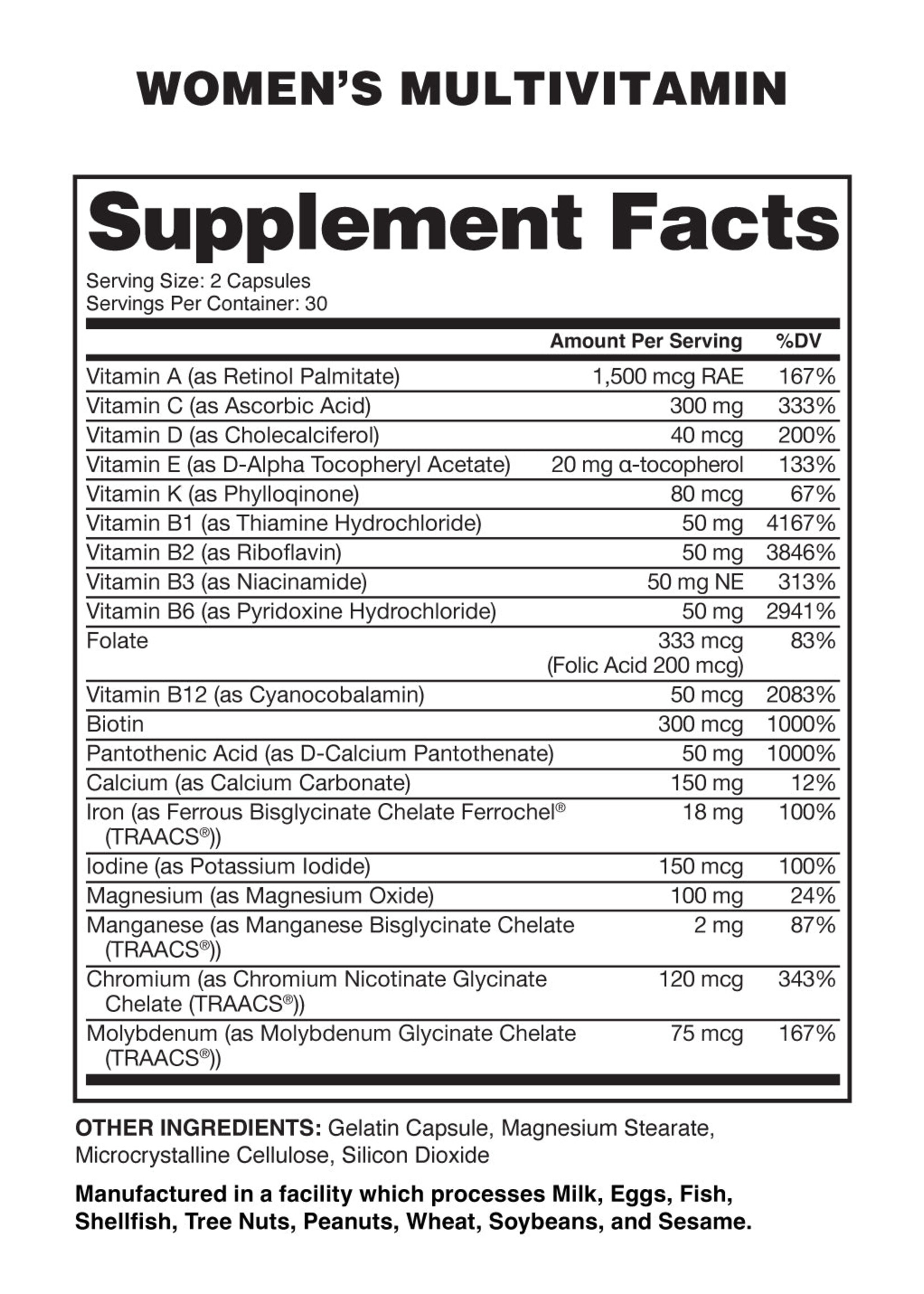
Recent investigations into high-demand UK vitamin supplements reveal a discrepancy between “100% Daily Value” labels and actual bioavailability. This regulatory gap highlights how supplement potency often fails to meet labeled claims, potentially misleading consumers about their nutritional intake and the efficacy of over-the-counter wellness products.
For the average consumer, a label claiming “100% of your daily requirement” suggests a guaranteed biological outcome. However, in the world of pharmacology and nutrition, there is a critical distinction between nominal dose (what is on the label) and bioavailability (the proportion of the nutrient that enters the circulation and is available for physiological activity). When supplements fail to meet these markers, patients with genuine deficiencies may inadvertently remain undertreated, while those over-supplementing risk toxicity.
In Plain English: The Clinical Takeaway
- Labels aren’t guarantees: “100%” refers to the amount added during manufacturing, not necessarily what your body absorbs.
- Quality varies: Not all supplements are regulated like medicines; some contain significantly less of the active ingredient than claimed.
- Food first: Whole foods provide “cofactors” (helper nutrients) that make vitamins easier for your body to use compared to isolated pills.
The Bioavailability Gap: Why “100%” is Often a Mathematical Illusion
The discrepancy found in these UK-marketed vitamins often stems from the stability of the compounds. Many vitamins are chemically unstable and degrade over time due to exposure to light, heat, or oxygen—a process known as oxidation. If a manufacturer adds 100% of a vitamin at the start of production, the actual concentration at the time of consumption may be significantly lower.
the mechanism of action for nutrient absorption is complex. For instance, fat-soluble vitamins (A, D, E, and K) require dietary fats to be absorbed across the intestinal lumen. If a patient takes a “100%” dose on an empty stomach, the actual systemic uptake may be negligible, regardless of the label’s accuracy.
This issue is compounded by the use of synthetic versus natural forms. For example, synthetic folic acid must be converted by the enzyme methylenetetrahydrofolate reductase (MTHFR) into a bioactive form. Patients with genetic polymorphisms in the MTHFR gene cannot efficiently process synthetic versions, rendering a “100%” label clinically irrelevant for their specific biology.
Regulatory Divergence: UK (MHRA) vs. USA (FDA) vs. EU (EMA)
The regulatory landscape for supplements is vastly different from that of pharmaceutical drugs. In the UK, the Medicines and Healthcare products Regulatory Agency (MHRA) and the Food Standards Agency (FSA) oversee supplements, but they generally do not pre-approve supplements for efficacy before they hit the market. Here’s a “post-market” surveillance system.
Similarly, in the United States, the FDA regulates supplements under the Dietary Supplement Health and Education Act (DSHEA), which treats them more like food than drugs. This means the burden of proof for “purity” and “potency” often falls on the manufacturer rather than a government auditor. When a UK-based product claims 100% potency, it is often a self-certified claim.
In contrast, the European Medicines Agency (EMA) maintains stricter guidelines on the pharmacopeia (the official book of drug standards) for substances that cross the line between “supplement” and “medicine.” This regulatory fragmentation allows companies to market products in regions with the least oversight, creating a “geographic lottery” for patient safety.
| Regulatory Body | Classification | Pre-Market Approval | Enforcement Mechanism |
|---|---|---|---|
| FDA (USA) | Dietary Supplement | No (Self-Affirmed) | Post-market surveillance |
| MHRA/FSA (UK) | Food Supplement | No | Compliance audits |
| EMA (EU) | Nutraceutical/Medicine | Variable (Strict) | Rigorous quality standards |
Funding, Bias, and the “Wellness” Industrial Complex
It is imperative to note that much of the data supporting the “necessity” of high-dose supplements comes from industry-funded trials. When research is funded by the supplement manufacturers themselves, there is a documented “funding bias” that tends to overstate efficacy and underreport the lack of clinical significance.
True clinical gold standards are double-blind, placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving the treatment. These trials often show that for healthy adults with a balanced diet, high-dose supplements provide no significant reduction in mortality or chronic disease risk.
“The over-reliance on supplement labels as a proxy for nutritional health is a public health fallacy. We must shift the focus from ‘percentage of daily value’ to ‘biomarker verification’ through blood function to ensure patients are not treating imagined deficiencies or ignoring real ones.” — Dr. Arisbe Moore, PhD in Nutritional Epidemiology
The Metabolic Pathway: From Ingestion to Cellular Utility
To understand why labels fail, we must look at the metabolic pathway. Once a vitamin is ingested, it must survive the acidic environment of the stomach, be absorbed in the modest intestine, and be transported via the bloodstream to the target organ. This is where competitive inhibition occurs; if a supplement contains too much of one nutrient (e.g., Zinc), it can block the absorption of another (e.g., Copper).
This interaction explains why a “100% Multivitamin” can actually create a new deficiency. By flooding the system with one specific isolate, the body’s natural homeostatic balance is disrupted, potentially leading to hypervitaminosis—a state of vitamin toxicity that can cause organ damage, particularly in the liver and kidneys.
Contraindications & When to Consult a Doctor
Supplementation is not benign. Make sure to seek immediate medical consultation if you fall into the following categories:
- Patients on Warfarin or Anticoagulants: High doses of Vitamin K can neutralize blood-thinning medications, increasing the risk of clotting.
- Individuals with Chronic Kidney Disease (CKD): Impaired renal clearance can lead to the dangerous accumulation of fat-soluble vitamins and minerals.
- Pregnant Women: Excess Vitamin A (retinol) is teratogenic, meaning it can cause birth defects. Always use prenatal vitamins approved by an OB-GYN.
- Patients with Hypercalcemia: Excessive Vitamin D supplementation can lead to dangerously high calcium levels in the blood.
Final Clinical Verdict
The “100%” label on popular UK vitamins should be viewed as a target, not a guarantee. As we move toward 2026, the trend in precision medicine is shifting toward nutrigenomics—tailoring nutrient intake based on an individual’s genetic blueprint rather than generic labels. Until regulatory bodies mandate third-party verification of potency for every batch, the most reliable “supplement” remains a diverse, whole-food diet.