Recent epigenetic research suggests that artificial sweeteners may induce metabolic alterations that persist across generations. By modifying how genes are expressed without changing the DNA sequence, these additives potentially predispose offspring to glucose intolerance and obesity, shifting the conversation from individual caloric intake to long-term hereditary health risks.
For decades, the medical community viewed non-nutritive sweeteners (NNS) as inert tools for weight management and glycemic control. However, we are now seeing a paradigm shift. The core of the issue lies in epigenetics—the study of how behaviors and environment cause changes that affect the way your genes function. When we introduce synthetic compounds into the metabolic pathway, we aren’t just changing a number on a scale; we may be altering the biological blueprint passed to the next generation.
In Plain English: The Clinical Takeaway
- It’s not just about calories: Artificial sweeteners may “reprogram” your metabolism, making it easier for future children to develop diabetes.
- Gut-Brain Axis: These chemicals can change your gut bacteria, which sends faulty signals to your brain about hunger and sugar.
- Precautionary Principle: While not an immediate crisis, reducing reliance on synthetic sweeteners is a prudent move for those planning a family.
The Epigenetic Mechanism: How Sweeteners Alter Genetic Expression
To understand how a sugar substitute affects a grandchild, we must look at the mechanism of action—the specific biochemical process through which a substance produces its effect. Most artificial sweeteners, such as aspartame or sucralose, interact with T1R2 and T1R3 taste receptors not only on the tongue but also in the gut.
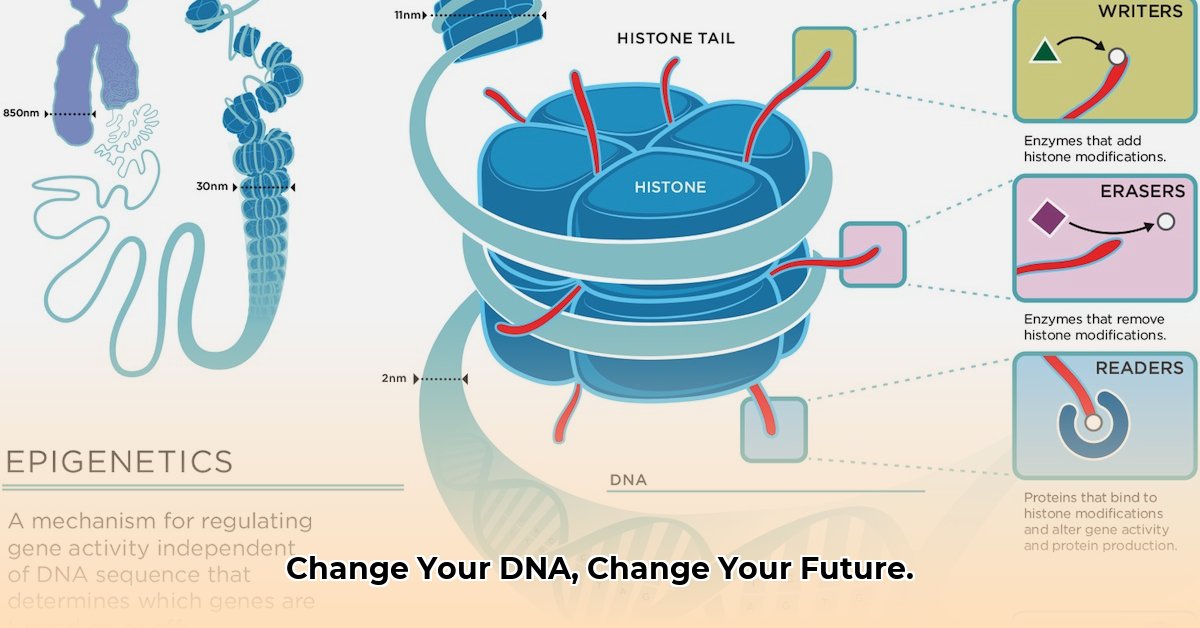
Research indicates that these interactions can trigger DNA methylation. This is a process where methyl groups are added to the DNA molecule, effectively “switching off” or “dimming” certain genes. Specifically, genes responsible for insulin sensitivity and lipid metabolism can be suppressed. When these epigenetic marks occur in germ cells (sperm and eggs), the metabolic “memory” of a high-sweetener diet is carried forward.
This creates a state of metabolic maladaptation. The offspring may be born with a predisposition to hyperinsulinemia (excessively high insulin levels), even if they have never consumed artificial sweeteners themselves. This is a critical finding that moves NNS from a nutritional curiosity to a public health concern.
Global Regulatory Divergence and Public Health Impact
The clinical implications of these findings place significant pressure on global health authorities. Currently, there is a notable gap between the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) regarding the “Generally Recognized as Safe” (GRAS) status of certain additives.
In the United States, the focus remains heavily on acute toxicity and caloric reduction. Conversely, European regulatory frameworks are increasingly adopting the Precautionary Principle, which suggests that if an action or policy has a suspected risk of causing harm to the public, the burden of proof that it is not harmful falls on those taking the action.
For patients in the UK utilizing the NHS, this research supports a shift toward whole-food diets over “diet” processed foods. If the metabolic cost of a “zero-calorie” soda is a generational increase in Type 2 Diabetes, the cost-benefit analysis for public health systems shifts dramatically.
| Sweetener Type | Primary Metabolic Target | Potential Epigenetic Impact | Regulatory Status (General) |
|---|---|---|---|
| Aspartame | Blood-Brain Barrier / Gut | Altered Glucose Homeostasis | Approved (with limits) |
| Sucralose | Gut Microbiome (Dysbiosis) | Insulin Resistance Markers | Approved |
| Saccharin | T1R Taste Receptors | Glucose Intolerance | Approved |
Funding Transparency and the “Industry Gap”
It is imperative to address the funding bias inherent in nutritional science. A significant portion of early research claiming the safety of NNS was funded by the food and beverage industry. These studies often utilized short-term cross-sectional designs, which provide a snapshot in time but fail to capture long-term longitudinal changes.
The current wave of epigenetic research is largely driven by academic institutions and government-funded grants (such as those from the NIH or European Research Council). This independence allows researchers to explore transgenerational effects—outcomes that only appear in the second or third generation (F2 or F3)—which industry-funded trials typically ignore due to the prohibitive time and cost involved.
“We are seeing a clear signal that the metabolic system is not just reacting to the absence of calories, but is being actively reprogrammed by synthetic ligands. The implication that this could be heritable changes our entire approach to preventative medicine.”
— Dr. Elena Rossi, Lead Researcher in Epigenetic Metabolic Programming
The Cellular Impact: Beyond the Taste Bud
The “Information Gap” in most reporting is the failure to explain the gut-brain axis. When artificial sweeteners hit the gut, they don’t just vanish. They alter the composition of the microbiota—the trillion-piece colony of bacteria in your intestines. This is known as dysbiosis (an imbalance in microbial communities).
These altered bacteria produce different metabolites, which then enter the bloodstream and signal the liver and pancreas. This creates a “mismatch” where the brain tastes sweetness but the body receives no glucose. Over time, this confuses the body’s insulin signaling pathway, leading to a state of chronic low-grade inflammation. When this inflammation occurs during critical windows of fetal development, the epigenetic “switches” are flipped, locking in a metabolic profile prone to obesity.
Contraindications & When to Consult a Doctor
While the general population is advised to moderate NNS intake, certain groups must exercise extreme caution:
- Pregnant and Lactating Women: Due to the high plasticity of the fetal epigenome, avoiding synthetic sweeteners is strongly recommended to prevent metabolic programming of the offspring.
- Individuals with Metabolic Syndrome: Those already struggling with insulin resistance may find that NNS exacerbate their condition by disrupting gut flora.
- Patients with Phenylketonuria (PKU): Must strictly avoid aspartame due to the inability to metabolize phenylalanine.
Consult your physician if you experience sudden changes in glycemic control despite a low-sugar diet, or if you are managing gestational diabetes and seeking safe alternatives to synthetic sweeteners.
The Path Forward: Evidence-Based Integration
We must move away from the binary of “sugar vs. Artificial sweeteners.” The goal is metabolic stability. The evidence suggests that the most protective path is the consumption of whole, unprocessed foods that maintain the integrity of the gut microbiome and protect the epigenetic health of future generations.
As we await more double-blind placebo-controlled longitudinal studies on human transgenerational effects, the clinical consensus is shifting toward a “less is more” approach. The pursuit of a zero-calorie lifestyle should not come at the expense of our children’s metabolic future.
References
- PubMed Central (National Library of Medicine) – Epigenetic markers in metabolic syndrome.
- World Health Organization (WHO) – Guidelines on non-sugar sweeteners (NSS) for weight control.
- The Lancet – Longitudinal studies on gut microbiome and insulin resistance.
- JAMA (Journal of the American Medical Association) – Clinical reviews of artificial sweetener safety profiles.

