New South Wales has expanded access to the nasal spray influenza vaccine for children, rolling out the jab-free alternative to general practitioners across the Riverina region. This initiative aims to increase pediatric vaccination rates by removing the barrier of needle phobia and improving accessibility within community primary care settings.
For parents and healthcare providers, this shift is more than a matter of convenience; it is a strategic public health maneuver. By transitioning from intramuscular injections to an intranasal delivery system, the healthcare system is targeting “vaccine hesitancy”—the delay or refusal of vaccines—specifically driven by the psychological trauma of needles in young children. When we lower the friction of administration, we increase the statistical probability of community-wide immunity, which is the only viable defense against the seasonal volatility of influenza.
In Plain English: The Clinical Takeaway
- No Needles: The vaccine is administered as a spray up the nose, making it significantly less stressful for children.
- Same Goal: It protects against the same seasonal flu strains as the traditional shot.
- Easier Access: You can now obtain this at your local GP in the Riverina region rather than only at specialized clinics.
The Mechanism of Action: How Mucosal Immunity Works
To understand why a nasal spray is effective, we must glance at the mechanism of action—the specific biochemical process through which a drug or vaccine produces its effect. Traditional flu shots are “inactivated” vaccines, meaning they contain killed viruses that trigger a systemic immune response in the bloodstream.
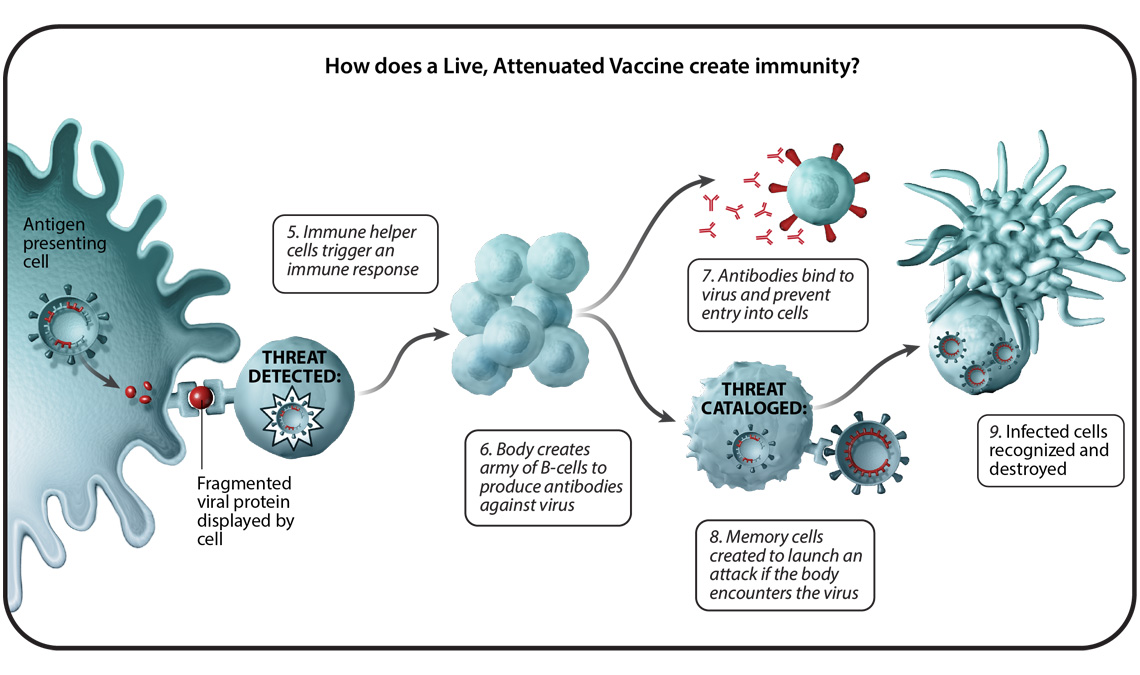
The nasal spray, however, is a Live Attenuated Influenza Vaccine (LAIV). “Attenuated” means the virus has been weakened so it cannot cause the disease in a healthy person. Instead of just triggering antibodies in the blood, LAIV mimics a natural infection by targeting the nasal mucosa (the lining of the nose). This induces mucosal immunity, creating a first line of defense exactly where the flu virus typically enters the body.
This dual-layer protection—both local mucosal and systemic IgG antibodies—is why some clinical data suggests LAIV may provide a more robust response in children than the inactivated injection. What we have is particularly critical in pediatric populations, where the immune system is still “learning” to recognize pathogens.
Global Regulatory Alignment and Epidemiological Impact
The rollout in New South Wales aligns with global standards set by the World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC). In the United States, the FDA has approved LAIV for a similar demographic, recognizing that pediatric uptake is the primary driver of “herd immunity”—the point where enough of a population is immune to stop a virus from spreading.
In the Riverina region, the geographical dispersion of patients often creates “healthcare deserts.” By empowering local GPs to administer the nasal spray, the NSW government is reducing the socio-economic barrier to vaccination. When a parent doesn’t have to drive two hours to a major hub for a pediatric appointment, the rate of compliance increases.
“The goal of diversifying delivery methods is to eliminate every possible excuse for non-vaccination. By integrating the nasal spray into primary care, we are treating the psychological barrier of the needle as a clinical hurdle that must be cleared to ensure public safety.” — Dr. Sarah Gilbert, Vaccinology Researcher.
Comparative Efficacy and Clinical Data
The funding for these vaccines typically comes from a combination of government procurement and pharmaceutical research and development (R&D) from companies like CSL Seqirus. These trials are rigorously monitored to ensure that the “attenuation” of the virus is stable and safe across different age groups.

The following table summarizes the primary differences between the two most common pediatric delivery methods based on established clinical consensus.
| Feature | Inactivated Vaccine (Jab) | LAIV (Nasal Spray) |
|---|---|---|
| Virus State | Killed/Inactivated | Live Attenuated (Weakened) |
| Immune Response | Systemic (Bloodstream) | Mucosal & Systemic |
| Administration | Intramuscular Injection | Intranasal Spray |
| Primary Benefit | Safe for immunocompromised | Higher pediatric compliance |
Addressing the “Live Virus” Concern
A common point of confusion among parents is the “live” nature of the nasal spray. It is imperative to understand that the virus is cold-adapted. So it is engineered to replicate only in the cooler environment of the nasal passages and cannot survive the warmer temperature of the lungs. It cannot cause pneumonia or a full-scale flu infection in a healthy child.
This distinction is vital for clinical transparency. While the systemic response is similar, the local response in the nose provides a “barrier effect” that inactivated shots cannot replicate. This is why the PubMed archives frequently highlight the importance of mucosal vaccines in preventing the transmission of respiratory droplets.
Contraindications & When to Consult a Doctor
Despite the benefits, the nasal spray is not suitable for everyone. Because it contains a live attenuated virus, We find strict contraindications—specific situations where a drug should not be used because it may be harmful to the patient.
- Immunocompromised Patients: Children with severe immune deficiencies or those taking immunosuppressant medications must avoid the nasal spray. They should receive the inactivated (jab) vaccine instead.
- Severe Asthma: Children with a history of severe asthma or wheezing should consult their pediatrician, as there is a slight increase in the risk of bronchospasm.
- Severe Allergy: Anyone with a known severe allergy to gelatin or any component of the vaccine should seek an alternative.
- High Fever: If a child is currently suffering from a high fever or a severe acute illness, vaccination should be postponed until they have recovered.
If your child experiences a high fever that does not respond to antipyretics, or develops difficulty breathing following administration, contact your healthcare provider immediately.
The Future of Pediatric Preventative Care
The expansion of the nasal flu vaccine in NSW is a harbinger of a broader shift toward “patient-centric” medicine. By prioritizing the psychological comfort of the patient, we improve the epidemiological outcome of the entire community. As we move toward 2027, the integration of more non-invasive delivery systems—potentially including mRNA-based nasal sprays—will likely further reduce the burden on our healthcare systems.
the choice between a jab and a spray is a secondary concern; the primary objective is the prevention of influenza-related hospitalizations. For the families of the Riverina and beyond, this rollout represents a victory for accessibility and a commitment to evidence-based public health.