Androgenetic alopecia affects approximately 50% of men by age 50, driven by genetic sensitivity to dihydrotestosterone. Whereas personal narratives highlight emotional distress, clinical management relies on FDA and EMA-approved interventions. Patients must distinguish between cosmetic concerns and underlying pathological conditions requiring medical triage.
The story of Luis, a 48-year-old man battling hair loss since youth, resonates with millions globally. As shared recently with Vita Health Media, his experience underscores the profound psychological burden of alopecia. But, anecdotal evidence alone cannot guide treatment. As we navigate the healthcare landscape of 2026, it is imperative to separate marketing narratives from peer-reviewed clinical consensus. Hair loss is rarely just cosmetic; it is often a visible marker of hormonal metabolism and genetic predisposition that requires rigorous medical evaluation.
In Plain English: The Clinical Takeaway
- Mechanism: Most common hair loss is caused by hormones shrinking hair follicles, not by stress or dirt.
- Expectations: Treatments maintain existing hair rather than regrowing a full head; consistency is critical for results.
- Safety: Prescription medications require monitoring for systemic side effects, particularly regarding hormonal balance.
The Pathophysiology of Follicular Miniaturization
To understand why life without hair feels unthinkable to patients like Luis, one must understand the biology of Androgenetic Alopecia (AGA). The primary driver is the conversion of testosterone into dihydrotestosterone (DHT) by the enzyme 5-alpha-reductase. In genetically susceptible individuals, DHT binds to androgen receptors in the scalp follicles. This binding triggers a process called follicular miniaturization, where the growth phase (anagen) shortens, and the resting phase (telogen) lengthens. Over time, thick terminal hairs are replaced by thin, unpigmented vellus hairs.
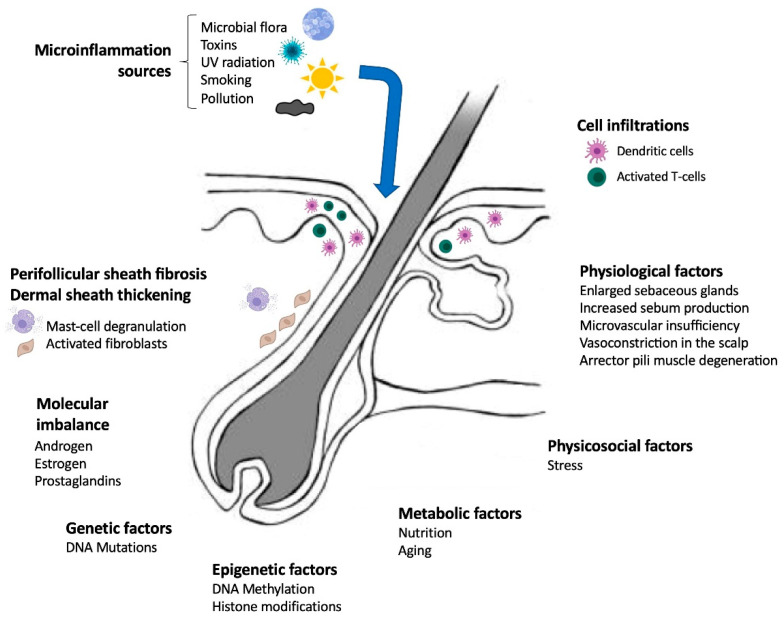
Recent longitudinal studies continue to validate that this process is irreversible without pharmacological intervention. While social media often promotes nutritional supplements, the clinical data indicates that unless a patient has a verified deficiency in iron, zinc, or biotin, supplementation yields negligible impact on AGA. The information gap here is critical: patients often spend years on ineffective wellness trends before seeking evidence-based dermatological care.
Regulatory Landscapes: FDA vs. EMA Approvals
Access to treatment varies significantly by region, impacting patient outcomes. In the United States, the Food and Drug Administration (FDA) has approved topical minoxidil and oral finasteride as first-line therapies. Conversely, the European Medicines Agency (EMA) maintains strict oversight on compounded formulations. As of early 2026, topical finasteride has gained traction in specific markets to reduce systemic absorption, though long-term safety data remains under post-marketing surveillance.
Geo-epidemiological bridging reveals disparities in care. In the UK, NHS guidelines often restrict hair loss treatments to cases causing severe psychological distress, whereas private clinics in Luxembourg and the US offer broader access. This creates a tiered system where economic status dictates access to emerging therapies like Janus Kinase (JAK) inhibitors, currently under investigation for alopecia areata but sometimes off-labelled for AGA.
“Patients must understand that hair loss treatments are maintenance therapies, not cures. Discontinuation typically results in the loss of gained benefits within four to six months.” — American Academy of Dermatology Guidelines
Funding Transparency and Clinical Bias
When evaluating fresh hair restoration technologies, funding sources must be scrutinized. Many pivotal trials for emerging hair loss drugs are industry-sponsored by pharmaceutical giants seeking market expansion. While this funding accelerates development, it introduces potential bias in efficacy reporting. Independent replication of data is essential. For instance, while Low-Level Laser Therapy (LLLT) devices are cleared by the FDA, independent meta-analyses suggest modest efficacy compared to pharmacological standards. Patients should prioritize treatments backed by double-blind placebo-controlled trials over anecdotal testimonials.
| Treatment Modality | Mechanism of Action | Common Side Effects | Regulatory Status (2026) |
|---|---|---|---|
| Topical Minoxidil | Potassium channel opener; vasodilation | Scalp irritation, hypertrichosis | FDA Approved (OTC) |
| Oral Finasteride | Type II 5-alpha-reductase inhibitor | Sexual dysfunction, mood changes | FDA Approved (Rx) |
| Low-Level Laser | Cellular energy (ATP) stimulation | Minimal; heat sensation | FDA Cleared (Device) |
Contraindications & When to Consult a Doctor
Not all hair loss is Androgenetic Alopecia. Sudden shedding, patchy loss, or scarring indicates conditions like alopecia areata, telogen effluvium, or lichen planopilaris, which require distinct management. Self-medicating with hormonal inhibitors like finasteride is contraindicated in women of childbearing potential due to the risk of teratogenicity (birth defects) if handled crushed or ingested. Patients with a history of depression should consult a psychiatrist before initiating 5-alpha-reductase inhibitors, given the documented association with mood disorders.
If you notice rapid onset hair loss accompanied by fatigue, weight changes, or skin alterations, immediate consultation is necessary. These may signal thyroid dysfunction or autoimmune disorders. A board-certified dermatologist can perform a scalp biopsy or trichoscopy to confirm the diagnosis. Do not rely on telehealth algorithms alone for prescription hormonal therapies; physical examination remains the gold standard for ruling out scarring alopecias which cause permanent destruction of the follicle.
The Future of Restorative Dermatology
The trajectory for hair loss treatment is moving towards personalized medicine. Genetic profiling may soon determine which patients respond best to anti-androgens versus growth stimulants. Until then, the standard of care remains rooted in established pharmacology. For patients like Luis, the path forward involves accepting the chronic nature of the condition while utilizing safe, regulated tools to manage progression. The goal is not merely aesthetic restoration, but the preservation of self-esteem through medically sound practices.

