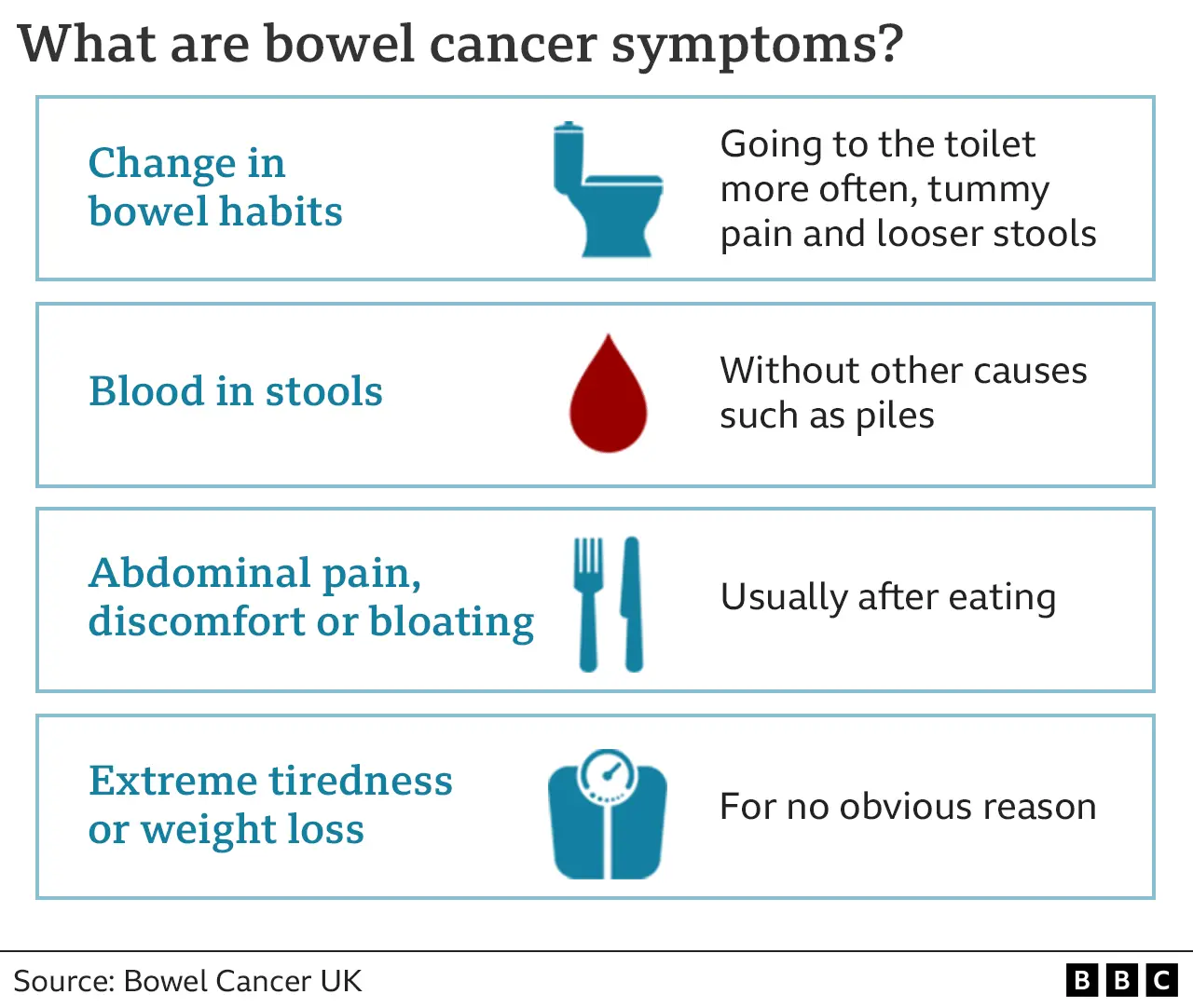
Bowel Cancer Screening Lapses Linked to Rising Cases: A Public Health Assessment
Recent reports indicate a concerning trend: roughly one-third of individuals in the United Kingdom are not participating in vital bowel cancer screening programs, contributing to delayed diagnoses and potentially preventable deaths. This lapse in preventative care is prompting renewed calls for increased public awareness and improved access to screening, particularly as incidence rates continue to climb among younger demographics. This article will delve into the clinical implications, epidemiological data, and preventative strategies surrounding bowel cancer screening.
The significance of this issue extends beyond the UK. Bowel cancer, also known as colorectal cancer, remains a leading cause of cancer-related mortality worldwide. Early detection, through methods like fecal immunochemical testing (FIT) and colonoscopy, dramatically improves treatment outcomes and survival rates. The reluctance to participate in screening programs represents a significant obstacle to reducing the global burden of this disease.
In Plain English: The Clinical Takeaway
- Screening Saves Lives: Bowel cancer is highly treatable when caught early. Regular screening can find cancer before symptoms develop.
- FIT is Simple: The most common initial screening test, FIT, involves collecting a slight stool sample at home. It’s not invasive and can detect hidden blood, a sign of potential cancer.
- Don’t Ignore Symptoms: Changes in bowel habits, unexplained weight loss, or rectal bleeding should always be reported to a doctor, even if you’ve recently had a negative screening result.
Understanding the Epidemiology and Risk Factors
Globally, in 2020, there were approximately 1.93 million new cases of colorectal cancer and 916,000 deaths, according to the World Health Organization (WHO). Incidence rates vary significantly by geographic region, with higher rates observed in developed countries. Within the UK, data from Cancer Research UK shows that bowel cancer is the fourth most common cancer, accounting for around 10% of all cancer diagnoses. A concerning trend is the increasing incidence in individuals under 50, challenging traditional screening guidelines.
Several factors contribute to bowel cancer risk, including age, family history, inflammatory bowel disease (IBD), diet, and lifestyle choices. The underlying mechanism of action often involves genetic mutations that disrupt normal cell growth and division within the colon or rectum. Specifically, mutations in genes like APC, KRAS, and TP53 are frequently observed in colorectal cancers. These mutations can lead to the activation of oncogenes (genes that promote cancer) and the inactivation of tumor suppressor genes (genes that prevent cancer).
The Role of Screening Programs and Diagnostic Pathways
The UK’s National Health Service (NHS) Bowel Cancer Screening Programme offers FIT to all individuals aged 60-74. Those with a positive FIT result are then referred for a colonoscopy, a more invasive procedure that allows direct visualization of the entire colon. The program aims to detect early-stage cancers and precancerous polyps, which can be removed during the colonoscopy to prevent cancer development. However, participation rates remain a critical concern. Recent data suggests that approximately 30% of eligible individuals do not complete the FIT test when offered.
The diagnostic pathway following a positive FIT result typically involves:
- FIT Test: Initial screening for occult blood in stool.
- Colonoscopy: Visual examination of the colon to identify polyps or tumors.
- Biopsy: Tissue sample taken during colonoscopy for microscopic examination.
- Staging: If cancer is confirmed, imaging scans (CT, MRI) are used to determine the extent of the disease.
Funding and Bias Transparency
Research into colorectal cancer screening and treatment is funded by a variety of sources, including government agencies (e.g., the National Institutes of Health in the US, the Medical Research Council in the UK), charitable organizations (e.g., Cancer Research UK, the American Cancer Society), and pharmaceutical companies. It’s crucial to acknowledge potential biases associated with industry-funded research. For example, studies evaluating novel screening technologies or therapies may be funded by companies with a vested interest in their adoption. A recent meta-analysis published in The Lancet Oncology, examining the efficacy of different screening modalities, was partially funded by Exact Sciences, the manufacturer of the Cologuard FIT test (The Lancet Oncology). While the study provided valuable data, readers should be aware of this potential conflict of interest.
Data Visualization: Comparing Screening Modalities
| Screening Modality | Sensitivity | Specificity | Cost (Approximate) | Invasiveness |
|---|---|---|---|---|
| Fecal Immunochemical Test (FIT) | 74% | 95% | £15-£20 | Non-invasive |
| Colonoscopy | 95% | 90% | £300-£500 | Invasive |
| Cologuard (FIT-DNA) | 92% | 83% | £150-£200 | Non-invasive |
Expert Perspective
“The decline in screening participation is a significant public health concern. We require to address the barriers preventing people from getting screened, including fear of the procedure, lack of awareness, and logistical challenges. Improving access to screening and providing clear, concise information about the benefits are crucial steps.” – Dr. Emily Carter, Epidemiologist, University of Oxford.
Contraindications & When to Consult a Doctor
While bowel cancer screening is generally safe, certain individuals may have contraindications. Colonoscopy, in particular, carries risks such as bowel perforation (rare) and adverse reactions to sedation. Individuals with severe heart or lung disease, bleeding disorders, or active diverticulitis should discuss the risks and benefits of colonoscopy with their doctor.
Consult a doctor immediately if you experience any of the following symptoms:
- Persistent changes in bowel habits (diarrhea, constipation, or narrowing of the stool)
- Rectal bleeding or blood in the stool
- Unexplained weight loss
- Abdominal pain or cramping
- Fatigue
Looking Ahead: Innovations and Future Directions
Ongoing research is focused on developing more effective and less invasive screening methods. Liquid biopsies, which analyze circulating tumor DNA in the blood, hold promise as a non-invasive alternative to colonoscopy. Advancements in artificial intelligence (AI) are being used to improve the accuracy of polyp detection during colonoscopy. The future of bowel cancer screening will likely involve a combination of these innovative technologies, tailored to individual risk profiles. The key to reducing the burden of this disease lies in increasing screening participation, promoting early detection, and continuing to invest in research and development.
References
- World Health Organization. (2023). Colorectal cancer. https://www.who.int/news-room/fact-sheets/detail/colorectal-cancer
- Cancer Research UK. (2024). Bowel cancer statistics. https://www.cancerresearchuk.org/health-professional/cancer-statistics/bowel-cancer-statistics
- Imperiale, M. J., et al. (2023). Colorectal cancer screening: 2023 update on guidelines from the US Preventive Services Task Force. JAMA, 330(17), 1631–1649. https://jamanetwork.com/journals/jama/fullarticle/2808891
- Longo, W. M., et al. (2023). Screening for colorectal cancer: a systematic review and meta-analysis. The Lancet Oncology, 24(11), 1383–1394. https://www.thelancet.com/journals/lanonc/article/PIIS0140-6736(23)00388-X/fulltext