Iptacopan, a first-in-class oral factor B inhibitor, is demonstrating promising results in treating C3 glomerulopathy (C3G), a rare kidney disease. Recent correspondence addresses the interpretation of complement biomarkers during iptacopan treatment, clarifying data from the APPEAR-C3G trial and offering crucial context for clinicians managing this complex condition. This clarification is vital for optimizing treatment strategies and monitoring patient response.
C3 glomerulopathy represents a significant challenge in nephrology. It’s characterized by the abnormal deposition of complement protein C3 in the glomeruli – the kidney’s filtering units – leading to inflammation and progressive kidney damage. Historically, treatment options have been limited, often relying on broad immunosuppression with significant side effects. Iptacopan offers a more targeted approach by specifically inhibiting factor B, a key protein in the alternative complement pathway, which is overactive in C3G. Understanding how biomarkers respond to this targeted therapy is paramount to assessing its effectiveness and tailoring treatment plans.
In Plain English: The Clinical Takeaway
- Targeted Kidney Treatment: Iptacopan is a new pill that aims to stop a specific part of your immune system from attacking your kidneys in a rare disease called C3 glomerulopathy.
- Biomarker Monitoring is Key: Doctors are learning how to best track the levels of certain proteins in your blood to see if the medicine is working correctly.
- Not a Cure, But Promising: While not a cure, iptacopan is showing potential to slow down kidney damage and improve quality of life for people with C3G.
Decoding Complement Biomarkers in Iptacopan Treatment
The recent correspondence from the authors of the APPEAR-C3G trial addresses concerns raised by Felix Poppelaars and colleagues regarding the interpretation of complement biomarkers. Specifically, the discussion centers on the expected changes in biomarkers like Bb and C3d following iptacopan initiation. Bb is a marker of alternative pathway activation, while C3d indicates complement activation more broadly. The authors clarify that a rapid decrease in Bb is anticipated with iptacopan, reflecting its mechanism of action – inhibiting factor B, a precursor to Bb. However, C3d levels may decrease more slowly due to its longer half-life and the complex interplay of complement pathways. This nuanced understanding is crucial to avoid misinterpreting biomarker fluctuations as treatment failure.
The mechanism of action of iptacopan is particularly noteworthy. Unlike traditional immunosuppressants that broadly suppress the immune system, iptacopan selectively targets factor B, a protein central to the alternative complement pathway. This pathway is often dysregulated in C3G, leading to uncontrolled complement activation and kidney damage. By inhibiting factor B, iptacopan effectively “turns down” this overactive pathway, reducing inflammation and protecting the glomeruli. This targeted approach minimizes off-target effects and potentially reduces the risk of infections associated with broader immunosuppression.
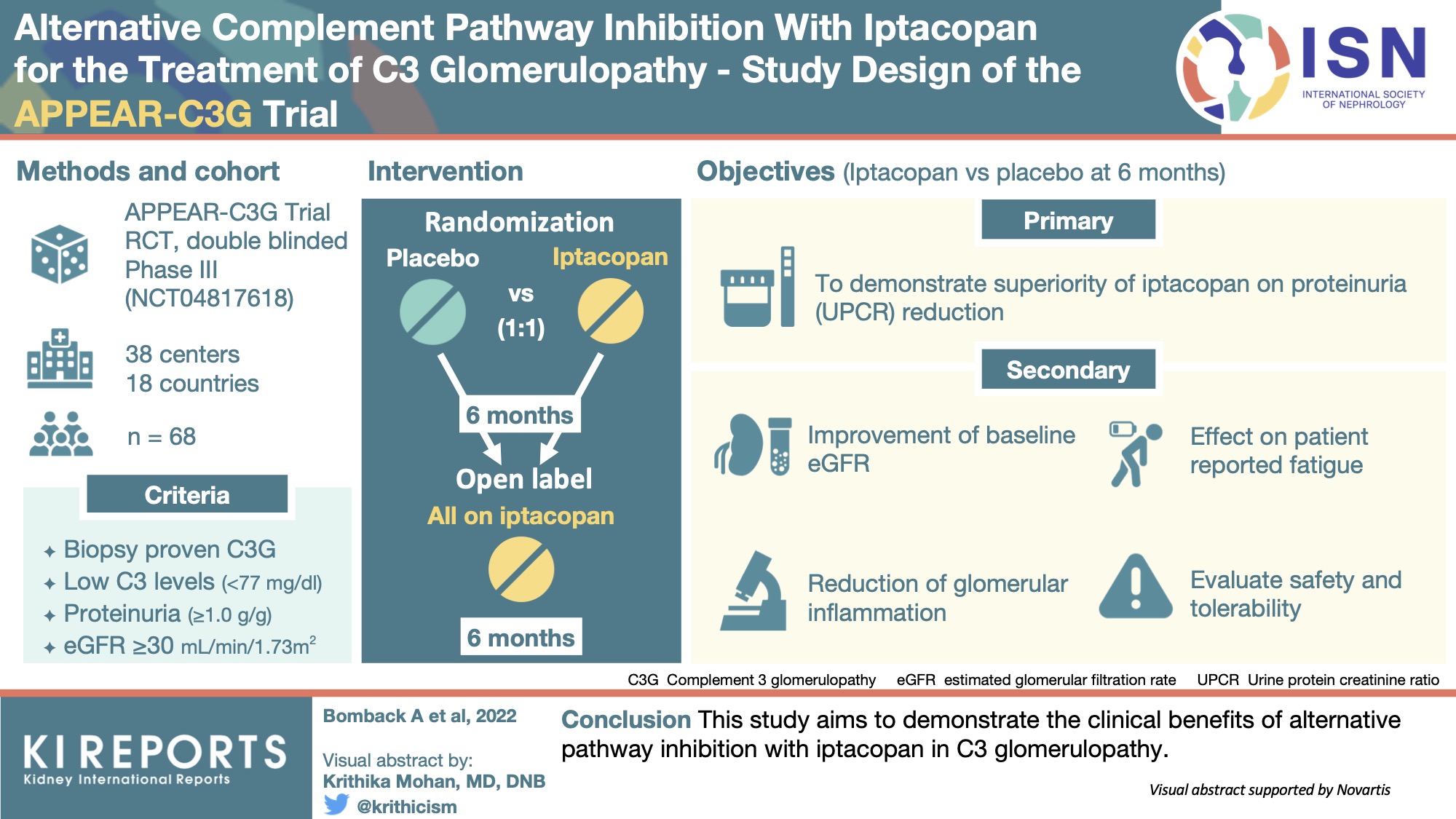
The APPEAR-C3G Trial: A Closer Look at Efficacy and Safety
The APPEAR-C3G trial (N=86) was a Phase III, randomized, double-blind, placebo-controlled study that demonstrated the efficacy and safety of iptacopan in patients with C3G. The trial met its primary endpoint, showing a statistically significant reduction in proteinuria – protein in the urine, a marker of kidney damage – in patients treated with iptacopan compared to placebo. Iptacopan demonstrated improvements in estimated glomerular filtration rate (eGFR), a measure of kidney function. However, it’s important to note that iptacopan is not without potential contraindications. Patients with active infections or a history of severe infections should be carefully evaluated before initiating treatment, as iptacopan may increase the risk of infection due to its immunosuppressive effects.
| Parameter | Iptacopan Group (N=43) | Placebo Group (N=43) | p-value |
|---|---|---|---|
| Change in Proteinuria (mg/g creatinine) | -58.2% | -19.1% | <0.001 |
| Change in eGFR (mL/min/1.73m2) | +7.8 | -3.2 | 0.04 |
| Serious Adverse Events | 16% | 14% | NS |
Regulatory Landscape and Global Access
Iptacopan has received accelerated approval from the U.S. Food and Drug Administration (FDA) and is currently under review by the European Medicines Agency (EMA). The FDA’s accelerated approval pathway allows for earlier access to promising therapies for serious conditions with unmet medical needs, based on surrogate endpoints like proteinuria reduction. However, continued monitoring and confirmatory trials are required to demonstrate long-term clinical benefit. In the UK, the National Health Service (NHS) is currently evaluating iptacopan for potential inclusion in its formulary, which would determine its availability to patients through the public healthcare system. Access to iptacopan remains a significant challenge globally, particularly in low- and middle-income countries where C3G may be underdiagnosed and resources for specialized treatment are limited.
“The development of iptacopan represents a paradigm shift in the treatment of C3 glomerulopathy. For years, we’ve relied on broad immunosuppression, which often comes with debilitating side effects. Iptacopan’s targeted approach offers the potential to improve patient outcomes while minimizing these risks.” – Dr. Brad Spellberg, Chief Medical Officer, Los Angeles County + USC Medical Center.
Funding and Potential Biases
The APPEAR-C3G trial was sponsored by Novartis, the manufacturer of iptacopan. While Novartis provided funding for the research, the authors have declared no competing interests. It is crucial to acknowledge this funding source, as it may introduce potential biases, though the study was rigorously designed and conducted according to established scientific standards. Transparency regarding funding is essential for maintaining public trust in medical research.
Contraindications & When to Consult a Doctor
Iptacopan is not suitable for everyone. Individuals with active infections, a history of severe infections, or known hypersensitivity to iptacopan should avoid this medication. Patients experiencing symptoms such as fever, chills, cough, or any signs of infection while on iptacopan should immediately consult their physician. Individuals with liver or kidney impairment may require dose adjustments, and close monitoring is essential. Pregnant or breastfeeding women should discuss the risks and benefits of iptacopan with their healthcare provider.
Looking ahead, further research is needed to optimize iptacopan treatment strategies and identify biomarkers that can predict treatment response. Long-term studies are too crucial to assess the durability of iptacopan’s effects and monitor for potential long-term side effects. The ongoing investigation into complement biomarkers, as highlighted in the recent correspondence, will undoubtedly play a vital role in refining our understanding of C3G and improving the lives of patients affected by this debilitating disease.
References
- Complement C3 glomerulopathy: diagnosis, pathogenesis, and treatment. https://pubmed.ncbi.nlm.nih.gov/34626788/
- Iptacopan for C3 glomerulopathy. https://www.nejm.org/doi/full/10.1056/NEJMoa2215639
- FDA Approves Iptacopan for C3 Glomerulopathy. https://www.fda.gov/drugs/fda-approves-iptacopan-c3-glomerulopathy
- Alternative pathway of complement. https://www.ncbi.nlm.nih.gov/books/NBK562223/