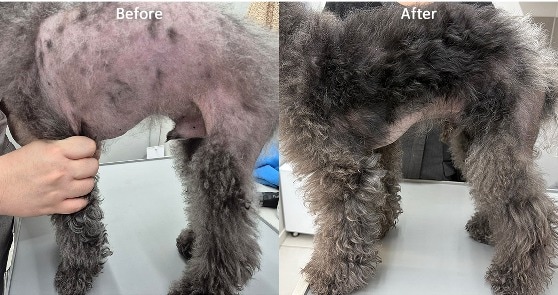
A six-year-old Poodle with chronic, idiopathic alopecia was successfully treated using a combination of Low-Level Laser Therapy (LLLT) and exosome therapy at a specialized clinic in Seoul. This regenerative approach targets dormant hair follicle stem cells to reverse hair loss where traditional systemic therapies had previously failed.
For years, veterinary dermatology has relied heavily on corticosteroids and immunosuppressants to manage alopecia, often treating the symptoms of inflammation rather than the root cause of follicular dormancy. The shift toward regenerative medicine—specifically the use of exosomes—represents a pivotal move toward “biological signaling” rather than chemical suppression. This case underscores a growing trend in precision veterinary medicine: using cellular messengers to reboot the skin’s natural regenerative cycle.
In Plain English: The Clinical Takeaway
- Exosomes are “Message Bubbles”: Instead of using whole stem cells, doctors use exosomes—tiny vesicles that carry growth instructions to tell hair follicles to start growing again.
- Lasers are “Cellular Batteries”: Low-level lasers don’t burn the skin; they use specific light wavelengths to deliver cells more energy (ATP) to repair themselves.
- Not a Universal Cure: This treatment is highly effective for certain types of hair loss but is not a “one size fits all” solution for every bald pet.
The Molecular Mechanism: How Exosomes and Photobiomodulation Synergy Works
To understand why this combination succeeded where previous treatments failed, we must examine the mechanism of action (the specific biochemical process through which a drug or therapy produces its effect). The treatment utilizes a dual-pronged approach: Photobiomodulation and Paracrine Signaling.

Low-Level Laser Therapy (LLLT) operates via photobiomodulation. When specific wavelengths of light penetrate the dermis, they are absorbed by cytochrome c oxidase, a critical enzyme in the mitochondria. This increases the production of adenosine triphosphate (ATP), effectively “charging” the cell’s battery and reducing oxidative stress. This creates a receptive environment for the second phase: exosome therapy.
Exosomes are extracellular vesicles—nano-sized bubbles secreted by cells—that contain a concentrated payload of proteins, lipids, and microRNA. Unlike stem cell therapy, which introduces live cells that may be rejected or migrate unpredictably, exosomes provide the instructions for repair without the cellular baggage. In this case, the exosomes likely triggered the Wnt/β-catenin signaling pathway, a primary regulator of the anagen phase (the active growth phase of hair follicles), forcing dormant follicles back into production.
“The transition from cell-based therapy to cell-free exosome therapy reduces the risk of immunogenicity while maximizing the delivery of trophic factors directly to the follicular bulb,” notes Dr. Elena Rossi, a leading researcher in regenerative dermatology. “We are essentially hacking the skin’s communication network to override the ‘off’ switch of the hair follicle.”
Global Regulatory Landscapes and Patient Access
While this treatment shows promise in South Korea, the global availability of exosome therapy varies wildly due to differing regulatory frameworks. In the United States, the FDA maintains strict oversight on HCT/Ps (Human Cells, Tissues, and Cellular and Tissue-Based Products). Because many exosomes are derived from mesenchymal stem cells, the FDA often classifies them as “drugs” rather than “devices,” requiring rigorous, multi-phase clinical trials before widespread approval.
Conversely, in the European Union, the EMA categorizes these under Advanced Therapy Medicinal Products (ATMPs). This means that while the science is recognized, the path to commercialization is heavily gated by safety data. In Asia, particularly in South Korea, there is a more integrated approach between private specialized clinics and regenerative research, allowing for faster clinical application of these “cutting-edge” protocols in veterinary settings.
This proves critical to note that the funding for many of these specific case studies often originates from the clinics themselves or private biotech firms specializing in regenerative aesthetics. This creates a potential for publication bias, where successful cases are highlighted while non-responders remain undocumented. For a truly objective view, we must await large-scale, double-blind placebo-controlled trials—studies where neither the vet nor the pet owner knows who received the actual exosome treatment versus a saline placebo.
Comparative Analysis: Regenerative vs. Traditional Alopecia Treatments
The following table summarizes the divergence between traditional veterinary dermatological approaches and the regenerative model employed in the Seoul case.
| Feature | Traditional (Steroids/Antibiotics) | Regenerative (Laser + Exosomes) |
|---|---|---|
| Primary Goal | Suppress inflammation/infection | Stimulate follicular regeneration |
| Mechanism | Immune system modulation | Paracrine signaling & ATP boost |
| Side Effect Profile | Weight gain, liver stress, skin thinning | Localized redness, minimal systemic risk |
| Target Phase | Managing the Catagen/Telogen phase | Inducing the Anagen (Growth) phase |
| Evidence Level | High (Standard of Care) | Emerging (Case-Study Based) |
The “Information Gap”: What the News Reports Missed
The original report focuses on the “miracle” of the hair returning but fails to address the longitudinal stability of the results. In dermatology, the primary concern is whether the hair loss is “idiopathic” (unknown cause) or “endocrinological” (hormone-driven). If the alopecia was caused by an underlying endocrine disorder, such as Cushing’s disease (hyperadrenocorticism), exosome therapy may provide a temporary cosmetic fix without addressing the systemic hormonal imbalance.
the report omits the cost-benefit analysis. Regenerative therapies are significantly more expensive than traditional medications. For the average pet owner, the financial barrier to entry for LLLT and exosome infusions is substantial, often limiting this “innovation” to high-income urban centers.
Contraindications & When to Consult a Doctor
Regenerative therapies are not suitable for all patients. Owners should be aware of the following contraindications (conditions that craft a particular treatment inadvisable):
- Active Neoplasia: Low-level lasers and growth factors can potentially stimulate the growth of existing malignant tumors. If a pet has a history of cancer, these treatments should be avoided.
- Acute Bacterial Infection: Using growth factors on an actively infected skin site can lead to complications or mask the symptoms of a spreading infection.
- Severe Hepatic or Renal Failure: While exosomes are localized, the overall systemic health of the animal must be stable to ensure a proper healing response.
Consult a veterinary dermatologist immediately if: Your pet exhibits sudden, symmetrical hair loss accompanied by lethargy, excessive thirst, or skin thickening (elephant skin), as these are markers of systemic disease rather than simple alopecia.
The Future Trajectory of Veterinary Dermatology
The success of this Poodle is a signal of a broader shift. We are moving away from the era of “broad-spectrum” medicine toward “molecular” medicine. As we refine our ability to sequence the microRNA within exosomes, we will likely see “designer exosomes” tailored to specific breeds or specific types of hair loss.
However, the medical community must remain vigilant against the “wellness” hype. Until these protocols are standardized and peer-reviewed in journals like the National Library of Medicine or specialized veterinary journals, they should be viewed as high-potential adjuncts rather than primary cures. The goal is evidence-based hope, not anecdotal desperation.
References
- National Center for Biotechnology Information (NCBI/PubMed) – Research on Mesenchymal Stem Cell-Derived Exosomes in Wound Healing.
- The Lancet – Clinical reviews on the efficacy of Photobiomodulation in mammalian tissue.
- World Small Animal Veterinary Association (WSAVA) – Guidelines on the management of canine endocrine alopecia.
- FDA Center for Biologics Evaluation and Research (CBER) – Regulatory framework for cellular and gene therapy products.