Discover the latest in health: evidence‑based wellness tips, medical breakthroughs, nutrition guidance, fitness insights, and expert advice for a healthier, happier life.
Recent advancements in cancer immunotherapy have illuminated the crucial role of structural design in vaccine efficacy, particularly in new research from Northwestern University focusing on human papillomavirus (HPV)-related cancers. By reengineering a therapeutic vaccine to enhance the immune response against HPV-driven tumors, researchers have made significant strides that could reshape how we approach cancer treatment.
In a study published on February 11, 2026, in the journal Science Advances, scientists demonstrated that the arrangement of vaccine components can dramatically impact the immune system’s ability to combat tumors. This innovative approach is rooted in a novel concept known as “structural nanomedicine,” pioneered by Northwestern’s Chad A. Mirkin, who emphasizes that the geometry of vaccine components can dictate their effectiveness.
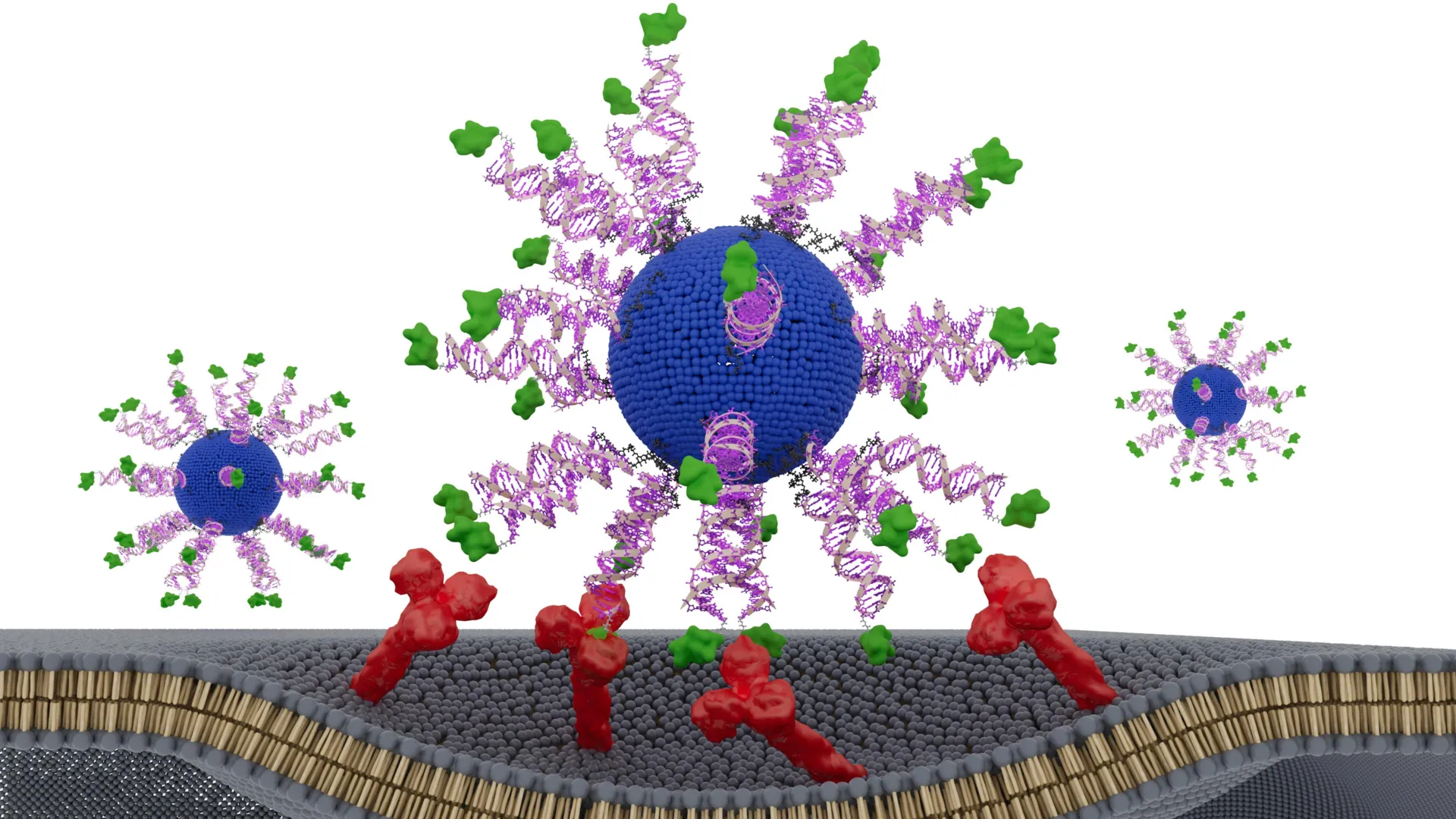
The research team developed a vaccine utilizing spherical nucleic acids (SNAs), which are globular DNA structures capable of penetrating immune cells and activating them. They systematically altered the configuration of these components and evaluated their performance in humanized animal models of HPV-positive cancers and patient-derived tumor samples.
One particular design proved to be significantly more effective than others, leading to reduced tumor growth and prolonged survival in animal models. This specific configuration also generated a higher number of active cancer-fighting T cells, highlighting how minor adjustments in molecular structure can lead to substantial differences in immune response.
Understanding the Mechanism
To create this therapeutic vaccine, researchers incorporated a lipid core, immune-activating DNA, and a short fragment of an HPV protein known to be present in tumor cells. Each version of the vaccine contained the same ingredients; although, the only variable was the positioning and orientation of the HPV-derived peptide, or antigen. Through their experiments, the team identified three different designs:
- One where the peptide was concealed inside the nanoparticle.
- Two surface-displayed versions, one with the peptide attached at the N-terminus and the other at the C-terminus.
The surface version with the N-terminal attachment generated the strongest immune response, triggering up to eight times more interferon-gamma, a critical signaling molecule produced by T cells that plays a key role in anti-tumor immunity. This resulted in a significant increase in the destruction of HPV-positive cancer cells in both humanized mouse models and tumor samples from HPV patients.

Shifting Paradigms in Vaccine Development
The traditional approach to vaccine development often involves a “blender approach,” where key ingredients are mixed without precise structural organization. Mirkin pointed out that although this method has yielded effective vaccines, such as the COVID-19 vaccines, there is room for improvement in terms of efficacy and safety.
By employing structured nanomedicine, researchers can design vaccines that not only improve immune responses but also minimize potential toxicity. The insights gained from this research could lead to enhanced therapeutic strategies for various cancers, including melanoma, triple-negative breast cancer, colon cancer, and prostate cancer, with several SNA-based drugs already in clinical trials.
Future Directions and Implications
Mirkin’s team plans to revisit earlier vaccine candidates that previously showed promise but failed to elicit strong immune responses. By demonstrating the importance of nanoscale structure, the findings offer a blueprint for refining existing therapeutic cancer vaccines. The researchers are also exploring the potential of artificial intelligence in vaccine design, leveraging machine learning to analyze countless structural combinations and identify the most effective configurations.
This evolving approach to vaccine formulation has broad implications for cancer treatment, potentially leading to more effective and tailored therapies. As the field of structural nanomedicine continues to grow, it could significantly alter the landscape of cancer immunotherapy.
With ongoing research and clinical trials, the prospect of vaccines that can efficiently target and destroy cancer cells is becoming more tangible. As these innovations unfold, they promise to enhance the landscape of cancer treatment and improve patient outcomes.
For further developments in this area, readers are encouraged to stay informed and engage in discussions regarding these advancements in cancer immunotherapy.
Disclaimer: The content provided is for informational purposes only and does not constitute medical advice.