The Centers for Disease Control and Prevention (CDC) has temporarily suspended diagnostic testing for high-priority infectious diseases, including rabies and mpox, citing critical staffing shortages. This pause, effective immediately as of early April 2026, forces reliance on state laboratories and private sectors, potentially delaying life-saving post-exposure prophylaxis for patients exposed to fatal zoonotic pathogens.
As a physician and health editor, I view this administrative pause not merely as a logistical hiccup, but as a fracture in the national biosafety net. When the federal reference laboratory—the ultimate arbiter for complex infectious disease cases—goes dark, the ripple effect is felt in emergency rooms from rural Kansas to urban New York. The immediate concern is not just the backlog, but the mechanism of action for these diseases; rabies, for instance, is nearly 100% fatal once clinical symptoms manifest, making rapid diagnostic confirmation via the Direct Fluorescent Antibody (DFA) test a matter of life and death.
In Plain English: The Clinical Takeaway
- Diagnostic Delays: Confirmatory testing for rabies and mpox is currently paused at the federal level, meaning results may take longer as samples are routed to alternative labs.
- Do Not Wait: If you suspect exposure to rabies (animal bite) or mpox (unexplained rash/lesions), seek immediate medical care; do not wait for federal confirmation to commence preventive treatment.
- State Lab Reliance: Many state public health laboratories remain operational, but they may experience higher volumes and slower turnaround times due to the CDC pause.
The Mechanism of the Pause: Why Staffing Impacts Virology
The suspension of services stems from a depletion in specialized personnel required to handle Biosafety Level 3 (BSL-3) pathogens. Unlike routine blood perform, testing for viruses like the Orthopoxvirus (which causes mpox) or the Rabies lyssavirus requires highly trained virologists and specific containment infrastructure. When the CDC’s specialized workforce shrinks, the capacity to perform Polymerase Chain Reaction (PCR) assays and immunofluorescence staining diminishes proportionally.
This creates a bottleneck in the diagnostic cascade. In a standard workflow, ambiguous samples from state labs are sent to the CDC for confirmation. With this pathway blocked, state laboratories must operate at maximum capacity without their usual federal backup. This increases the risk of false negatives or prolonged turnaround times, during which a patient with rabies exposure could miss the critical window for effective Post-Exposure Prophylaxis (PEP).
“The interruption of reference testing services compromises the surveillance network essential for detecting emerging variants. We are effectively flying blind on specific zoonotic threats until staffing levels are restored.”
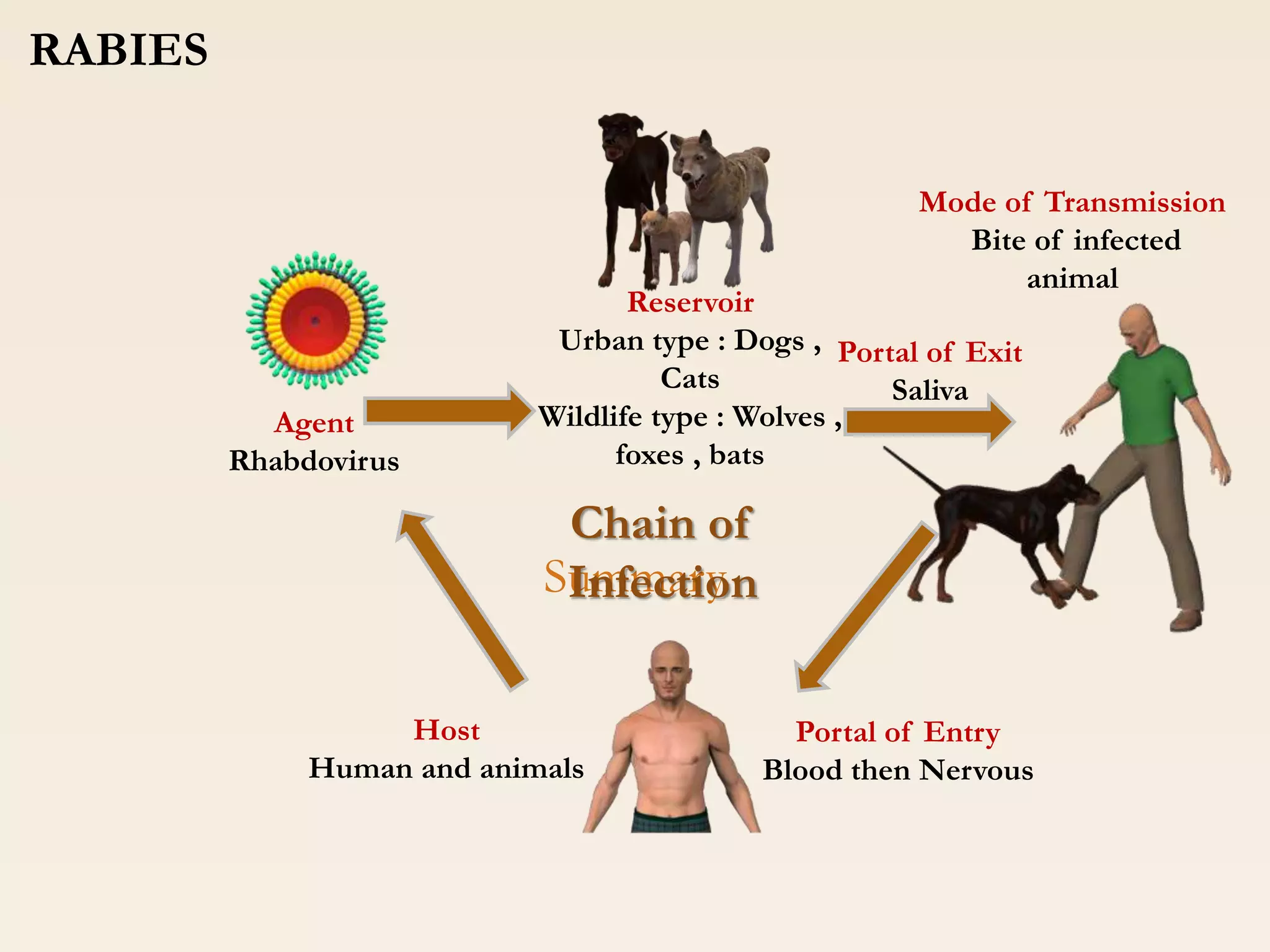
Clinical Implications: Rabies and the Time-Sensitive Window
The most urgent clinical implication involves rabies. The virus travels from the site of inoculation (usually a bite) along peripheral nerves to the central nervous system. Once the virus breaches the blood-brain barrier, the blood-brain barrier prevents antibodies from entering, rendering treatment futile. The CDC pause impacts the ability to test animal vectors (the biting animal) quickly.
Typically, if a biting animal tests negative, the human victim avoids the PEP regimen, which involves a series of painful injections. If testing is delayed due to the CDC pause, physicians must assume the animal is positive and administer PEP immediately as a precaution. This strains hospital resources and subjects patients to unnecessary medical interventions due to a lack of diagnostic data.
Regarding mpox, the pause affects the confirmation of clade-specific variants. With the global spread of different mpox clades, distinguishing between them is vital for public health tracking, even if the clinical treatment (supportive care and antivirals like tecovirimat) remains similar. The lack of federal sequencing capacity hampers our ability to track viral evolution in real-time.
Contraindications & When to Consult a Doctor
Patients should not interpret this pause as a reason to delay care. The risk of untreated exposure far outweighs the inconvenience of testing delays.
- Rabies Exposure: Consult a doctor immediately after any bite or scratch from a wild animal (bats, raccoons, skunks) or an unvaccinated domestic animal. Do not wait for test results to start PEP if the animal cannot be tested immediately.
- Mpox Symptoms: Seek care if you develop a new, unexplained rash, fever, or swollen lymph nodes, especially if you have had close contact with a confirmed case.
- Immunocompromised Status: Patients with weakened immune systems should be particularly vigilant, as their window for effective intervention is narrower.
Geo-Epidemiological Bridging and Resource Allocation
This staffing crisis highlights a fragility in the U.S. Public health infrastructure compared to international counterparts. While the U.S. CDC pauses, the European Centre for Disease Prevention and Control (ECDC) and the World Health Organization (WHO) continue to coordinate global reference testing. This disparity may lead to a reliance on international reference labs for complex U.S. Cases, introducing logistical delays in sample transport.
the funding transparency regarding this shortage points to broader budgetary constraints within federal health agencies. Unlike pharmaceutical research funded by private grants, public health surveillance is tax-funded. When appropriations do not match the inflation-adjusted cost of retaining high-level virologists, the “brain drain” to the private sector (evident in the surge of medical communication jobs seeking similar expertise) accelerates the depletion of government labs.
| Pathogen | Standard Test Method | Clinical Urgency | Impact of CDC Pause |
|---|---|---|---|
| Rabies Virus | Direct Fluorescent Antibody (DFA) | Critical (Hours matter) | Delayed animal testing; increased empirical PEP use. |
| Mpox (Orthopoxvirus) | PCR (Polymerase Chain Reaction) | High (Isolation required) | Slower variant identification; reliance on state labs. |
| Other Pox Viruses | Viral Culture / PCR | Moderate to High | Limited capacity for rare zoonotic identification. |
The Path Forward: Restoring Diagnostic Integrity
Resolving this pause requires more than temporary hiring sprees; it demands a structural reevaluation of how we value public health labor. The current job market shows a high demand for medical editors and scientific communicators, suggesting that the expertise exists but is migrating to private communications firms rather than remaining in public service. Until the CDC can guarantee 24/7 reference testing capability, the burden falls on local health departments to maintain vigilance.
For the patient, the directive is clear: trust your local provider’s clinical judgment over the availability of a federal test result. In the absence of a confirmed negative from the CDC, the standard of care defaults to the highest safety protocol. We must treat the silence from the federal lab not as an all-clear signal, but as a call for heightened clinical suspicion.
References
- Centers for Disease Control and Prevention. “Rabies: Post-Exposure Prophylaxis.” CDC.gov.
- World Health Organization. “Mpox (formerly monkeypox) Diagnostic Testing Guidelines.” WHO.int.
- Seet, B. T., et al. “Pathogenesis and Virulence of Orthopoxviruses.” The Lancet Infectious Diseases.
- Jackson, A. C. “Current Understanding of the Pathogenesis of Rabies.” Current Opinion in Virology.
- American Association for the Advancement of Science. “Science Translational Medicine: Public Health Infrastructure.” AAAS.org.