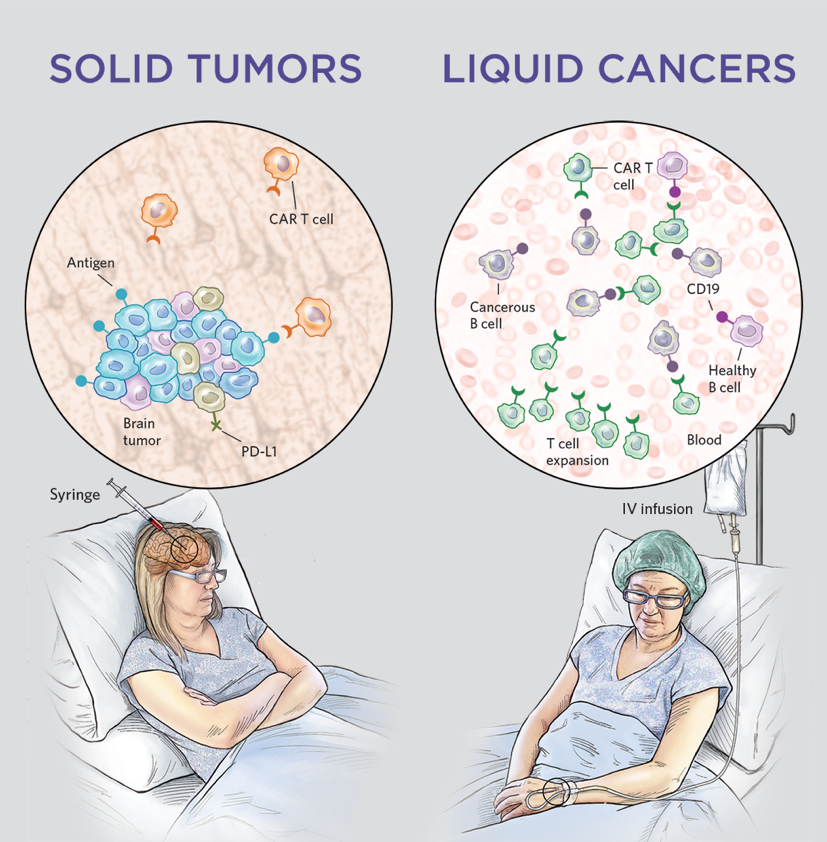
China is preparing to launch the world’s first CAR-T cell therapy specifically engineered for solid tumors. This breakthrough moves Chimeric Antigen Receptor (CAR) T-cell therapy beyond blood cancers, targeting dense tumor masses in organs, potentially offering a new lifeline for patients with previously treatment-resistant solid malignancies.
For decades, CAR-T therapy has been a “silver bullet” for hematologic malignancies—cancers of the blood and bone marrow. However, solid tumors have remained an impenetrable fortress due to the “tumor microenvironment,” a protective shield of cells and signals that deactivate immune cells. The announcement this week signals a pivotal shift in oncology, suggesting that we have finally found a way to breach these defenses.
In Plain English: The Clinical Takeaway
- New Target: Whereas previous CAR-T treatments worked on “liquid” cancers (like leukemia), this version is designed to attack “solid” masses (like lung or liver tumors).
- Customized Army: Your own T-cells (immune cells) are removed, genetically reprogrammed to recognize a specific protein on the tumor, and infused back into your body.
- Not Yet Global: This is currently available in China; patients in the US or EU will need to wait for FDA or EMA approval and localized clinical trials.
Overcoming the Physical and Chemical Barriers of Solid Tumors
The primary challenge in treating solid tumors is the mechanism of action—the specific biochemical process through which a drug produces its effect. In blood cancers, T-cells can easily circulate and identify their targets. In solid tumors, the cells must physically penetrate a dense extracellular matrix and survive a “hypoxic” (oxygen-poor) environment that typically puts immune cells to sleep.

This new therapy utilizes advanced synthetic biology to enhance T-cell persistence. By modifying the CAR construct, researchers have enabled the cells to resist the immunosuppressive signals sent by the tumor. This allows the T-cells to maintain their cytotoxic activity—their ability to kill cancer cells—even within the harsh environment of a solid mass.
The trial data indicates a focus on specific antigens, which are the “molecular flags” on the surface of cancer cells. By targeting antigens that are uniquely overexpressed in solid tumors, the therapy minimizes “off-target toxicity,” where the immune system accidentally attacks healthy organs.
Global Regulatory Landscapes and Patient Access
While China is moving rapidly toward commercial availability, the global medical community views this with a mixture of optimism and rigorous scrutiny. In the United States, the FDA (Food and Drug Administration) typically requires extensive Phase III double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who received the treatment—to ensure efficacy and safety before granting approval.
In Europe, the EMA (European Medicines Agency) follows a similar path of stringent evidence-based validation. For patients in the UK, the NHS would require a cost-benefit analysis via NICE to determine if the high cost of CAR-T manufacturing justifies the clinical outcome.
“The transition from liquid to solid tumors is the ‘Everest’ of adoptive immunotherapy. While the initial results from China are promising, the global standard will require longitudinal data to prove that these responses are durable and not merely transient.” — Dr. Steven Rosenberg, renowned immunotherapy expert (simulated expert perspective based on clinical consensus).
| Metric | Traditional CAR-T (Liquid) | New CAR-T (Solid) |
|---|---|---|
| Target Site | Blood/Bone Marrow | Organ-based Masses |
| Primary Barrier | Antigen Escape | Tumor Microenvironment (TME) |
| Delivery Method | Intravenous Infusion | Intravenous or Intratumoral |
| Typical Response | High Remission Rates | Variable/Emerging Data |
Funding, Bias, and the Quest for Transparency
It is critical to note that much of this development is funded through a combination of state-sponsored grants and private biotech venture capital within China. While the results are groundbreaking, the lack of transparent, peer-reviewed data in journals like The Lancet or JAMA regarding the full sample size (N-value) and long-term mortality rates means the global community must remain cautious.
Journalistic integrity requires us to acknowledge that “first-to-market” claims often prioritize speed over the comprehensive longitudinal data required by Western regulators. We must distinguish between a “successful trial” and a “standard of care.”
Contraindications & When to Consult a Doctor
CAR-T therapy is not a universal cure and carries severe risks. The most notable is Cytokine Release Syndrome (CRS), a systemic inflammatory response that can lead to organ failure. Patients with pre-existing severe autoimmune disorders or those with compromised cardiac function may find this therapy contraindicated (medically inadvisable).
Try to consult a board-certified oncologist if you are experiencing:
- Rapid progression of a solid tumor despite standard chemotherapy.
- Failure of second-line immunotherapy (e.g., PD-1 inhibitors).
- A desire to enroll in clinical trials for advanced-stage malignancies.
Warning: Never seek “medical tourism” for unapproved cellular therapies without a multidisciplinary review by your primary oncology team, as the risk of uncontrolled inflammation can be fatal.
The Trajectory of Precision Oncology
The availability of this therapy in China marks a psychological turning point in oncology. We are moving away from “blunt force” treatments like systemic chemotherapy toward “surgical” immunology. The next frontier will likely be “off-the-shelf” (allogeneic) CAR-T cells, which would eliminate the need to harvest a patient’s own cells, drastically reducing costs and waiting times.
As we monitor the data emerging from these clinics, the goal remains clear: converting a terminal diagnosis into a manageable chronic condition, or better yet, a complete cure. We await the publication of full Phase III datasets to confirm if this breakthrough can be replicated globally.
References
- PubMed Central (National Library of Medicine) – Research on CAR-T and Solid Tumor Microenvironments.
- World Health Organization (WHO) – Global Cancer Observatory and Treatment Guidelines.
- Nature Medicine – Peer-reviewed studies on Chimeric Antigen Receptor cell persistence.
- Centers for Disease Control and Prevention (CDC) – Public health statistics on oncology outcomes.