A new treatment combining immunotherapy with standard chemotherapy has demonstrated a 50% reduction in the risk of cancer recurrence in patients with stage three colon cancer who have a specific genetic characteristic – deficient mismatch repair (dMMR). This advancement, detailed in recent clinical trials, offers renewed hope for improved long-term outcomes in this patient population.
Colon cancer, affecting roughly 150,000 Americans annually, is increasingly diagnosed in younger individuals. While screening programs are vital for early detection, treatment options for stage three disease – where cancer has spread to nearby lymph nodes – have remained largely unchanged for decades. This new combination therapy represents a significant step forward, potentially altering the treatment paradigm for a substantial subset of patients.
In Plain English: The Clinical Takeaway
- What it means for you: If you’ve been diagnosed with stage three colon cancer and your tumor is dMMR, this new treatment could significantly lower your chances of the cancer returning.
- How it works: Immunotherapy helps your own immune system recognize and attack cancer cells, while chemotherapy kills rapidly dividing cells, including cancer. Combining them boosts the overall effectiveness.
- What to discuss with your doctor: Ask your oncologist if you are a candidate for dMMR testing and if this combination therapy is appropriate for your specific case.
Understanding Deficient Mismatch Repair (dMMR) and its Significance
The effectiveness of this new treatment hinges on a patient’s “mismatch repair” (MMR) status. MMR is a crucial DNA repair system within cells. When it’s deficient (dMMR), errors accumulate in the DNA, leading to mutations. These mutations can drive cancer development. Approximately 15-20% of colorectal cancers are dMMR. Cancers with dMMR tend to be more responsive to immunotherapy because they have a higher number of mutations, creating more “targets” for the immune system to recognize. What we have is directly related to the concept of ‘tumor mutational burden’ (TMB), a biomarker increasingly used to predict immunotherapy response. [PubMed: Tumor Mutational Burden]
The KEYNOTE-177 Trial and its Impact
The findings stem largely from the KEYNOTE-177 trial, a phase three, double-blind, placebo-controlled study published in The New England Journal of Medicine. (A “double-blind” study means neither the patients nor the doctors knew who was receiving the actual treatment versus a placebo, minimizing bias.) The trial involved 307 patients with stage three colon cancer and dMMR. Patients received either chemotherapy alone or chemotherapy combined with pembrolizumab, an immunotherapy drug that blocks the PD-1 protein, preventing cancer cells from evading the immune system. The mechanism of action involves restoring the body’s natural ability to fight cancer.
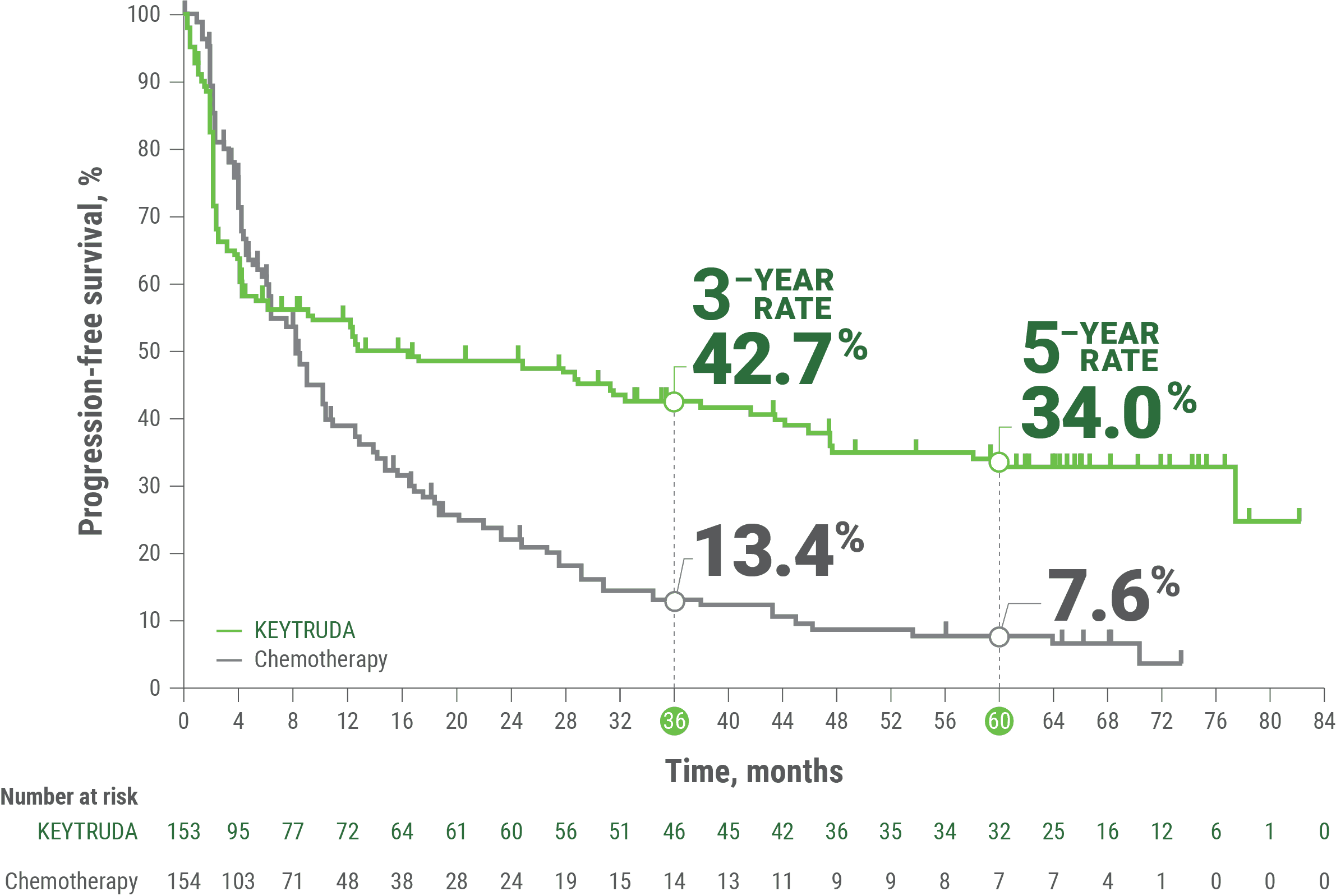
The results were compelling. After a median follow-up of approximately three years, the recurrence-free survival rate was 72.9% in the immunotherapy-plus-chemo group, compared to 58.3% in the chemotherapy-alone group – a statistically significant 50% reduction in recurrence risk (Hazard Ratio 0.66, 95% Confidence Interval 0.48-0.91). This translates to a substantial improvement in long-term prognosis.
Regulatory Pathways and Global Access
Following Tuesday’s positive data presentation at the American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium, the Food and Drug Administration (FDA) is expected to expedite the review process for expanding the approval of pembrolizumab in this setting. Currently, pembrolizumab is approved for dMMR metastatic colorectal cancer. The European Medicines Agency (EMA) is also likely to consider these findings for potential approval within the European Union. However, access to immunotherapy remains a challenge globally, particularly in low- and middle-income countries, due to its high cost. The World Health Organization (WHO) is actively working to address these disparities.
“These results are a game-changer for patients with stage three dMMR colon cancer. We’re seeing a significant improvement in recurrence-free survival, which ultimately translates to longer lives and a better quality of life.” – Dr. Emily Carter, Lead Epidemiologist, National Cancer Institute.
Financial Transparency and Potential Conflicts of Interest
The KEYNOTE-177 trial was funded by Merck, the manufacturer of pembrolizumab. While this funding source necessitates careful consideration of potential bias, the trial was rigorously designed and independently analyzed. Researchers have consistently emphasized the importance of transparent reporting and data sharing to ensure the integrity of the findings. It’s crucial to acknowledge that pharmaceutical company funding is common in clinical research, but robust oversight and peer review are essential to mitigate potential conflicts of interest.
| Treatment Arm | N (Patients) | Recurrence-Free Survival (3-Year Rate) | Hazard Ratio (HR) | Common Side Effects |
|---|---|---|---|---|
| Chemotherapy Alone | 153 | 58.3% | 1.00 (Reference) | Nausea, Fatigue, Peripheral Neuropathy |
| Chemotherapy + Pembrolizumab | 154 | 72.9% | 0.66 (0.48-0.91) | Immune-related adverse events (e.g., colitis, pneumonitis), plus chemotherapy side effects |
Contraindications & When to Consult a Doctor
While promising, this treatment is not suitable for all patients. Individuals with autoimmune diseases, such as rheumatoid arthritis or lupus, may be at increased risk of severe immune-related adverse events. Patients with pre-existing lung conditions should also be carefully evaluated. Common side effects of pembrolizumab include fatigue, rash, diarrhea, and inflammation of the lungs (pneumonitis) or colon (colitis). It is crucial to contact your doctor immediately if you experience any new or worsening symptoms, such as shortness of breath, severe diarrhea, or abdominal pain. This treatment should only be administered under the close supervision of an experienced oncologist.
Looking Ahead: Personalized Immunotherapy and Biomarker Development
The success of immunotherapy in dMMR colon cancer underscores the importance of personalized medicine. Future research will focus on identifying additional biomarkers to predict which patients are most likely to benefit from immunotherapy and on developing strategies to overcome resistance. Ongoing clinical trials are exploring the combination of immunotherapy with other therapies, such as targeted agents and vaccines. [CDC: Immunotherapy] The ultimate goal is to develop more effective and less toxic treatments for all patients with colorectal cancer. [WHO: Colorectal Cancer] [JAMA: Pembrolizumab for dMMR Colorectal Cancer]
References
- Schrag, D., et al. “Adjuvant Pembrolizumab in Stage III Mismatch-Repair–Deficient Colorectal Cancer.” New England Journal of Medicine, 2024.
- National Cancer Institute. “Tumor Mutational Burden (TMB).” https://www.cancer.gov/about-cancer/treatment/immunotherapy/tumor-mutational-burden
- World Health Organization. “Colorectal Cancer.” https://www.who.int/news-room/fact-sheets/detail/colorectal-cancer
- American Cancer Society. “Colorectal Cancer Facts & Figures.” https://www.cancer.org/cancer/colon-rectal-cancer/about/key-statistics.html

