Cardiovascular disease remains the leading cause of mortality globally. Although omega-3 fatty acids show promise in lipid management, recent 2026 clinical data suggests benefits are specific to prescription-grade EPA rather than over-the-counter fish oil blends for secondary prevention.
As we navigate the complexities of preventive cardiology this spring, patients frequently ask whether a daily capsule can replace lifestyle modifications or statin therapy. The confusion stems from conflicting headlines regarding marine-derived polyunsaturated fatty acids. Understanding the distinction between dietary supplementation and pharmacological intervention is critical for reducing myocardial infarction risk without exposing patients to unnecessary polypharmacy or financial burden.
The Biochemical Pathway of Triglyceride Reduction
Omega-3 fatty acids, specifically eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), operate through distinct mechanisms of action within hepatic metabolism. These molecules inhibit diacylglycerol acyltransferase, an enzyme crucial for triglyceride synthesis. By reducing the liver’s production of incredibly-low-density lipoproteins (VLDL), omega-3s lower circulating triglyceride levels. However, lowering triglycerides does not automatically equate to reduced cardiovascular events. The anti-inflammatory properties of EPA specifically stabilize plaque within the arterial walls, reducing the likelihood of rupture that leads to stroke or heart attack.
It is vital to distinguish between oxidation states. Over-the-counter supplements often lack the purity required to prevent lipid peroxidation, which can paradoxically increase oxidative stress. Prescription formulations undergo rigorous stabilization to ensure the molecule remains bioavailable without degrading into pro-inflammatory byproducts before absorption.
In Plain English: The Clinical Takeaway
- Not All Fish Oil Is Equal: Prescription strength omega-3s are purified and regulated like drugs, whereas store-bought supplements vary widely in actual EPA content.
- Triglycerides vs. Events: Lowering fat levels in the blood does not guarantee protection against heart attacks unless specific high-risk criteria are met.
- Interaction Risks: High doses can thin the blood slightly, requiring monitoring if you are already on anticoagulants or preparing for surgery.
Divergent Outcomes in Phase III Cardiovascular Trials
The clinical landscape is defined by two pivotal trials that shaped our 2026 understanding. The REDUCE-IT trial demonstrated a 25% relative risk reduction in major adverse cardiovascular events using high-dose icosapent ethyl (pure EPA). Conversely, the STRENGTH trial, which used a combination of EPA and DHA in a carboxylic acid formulation, was stopped early for futility. This dichotomy suggests that DHA may attenuate the beneficial effects of EPA in high-risk populations, or that the carrier oil matters significantly.
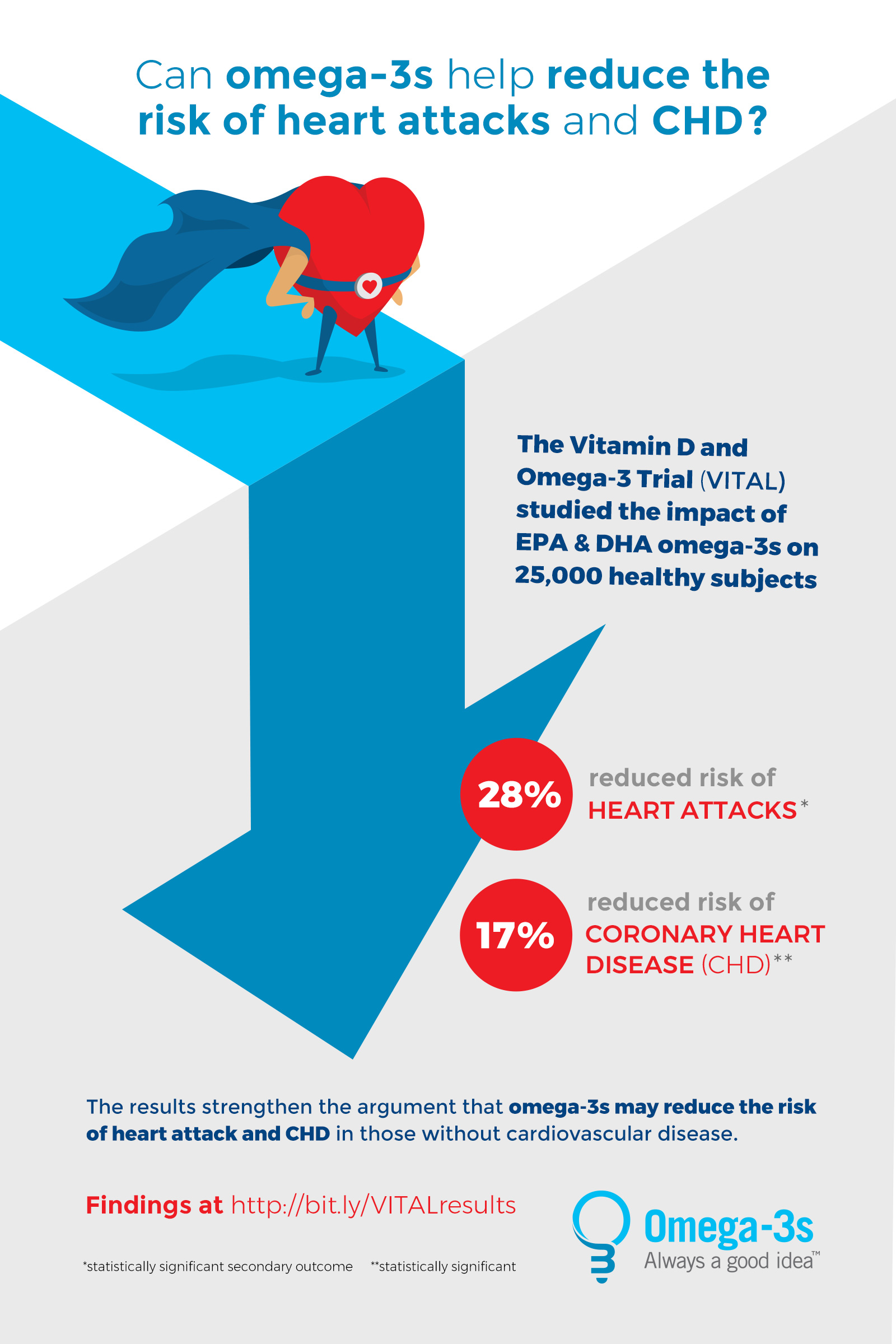
Funding transparency is essential here. REDUCE-IT was funded by Amarin Pharma, while STRENGTH was funded by AstraZeneca. Independent meta-analyses published in The Lancet confirm that the benefit is largely confined to patients with established cardiovascular disease and elevated triglycerides despite statin therapy. For the general wellness population, the VITAL study long-term follow-up indicates no significant reduction in primary prevention events.
| Feature | Prescription Omega-3 (Icosapent Ethyl) | Over-the-Counter Fish Oil |
|---|---|---|
| Regulatory Status | FDA Approved Drug (NDA) | Dietary Supplement (DSHEA) |
| Purity & Oxidation | Highly Purified, Stabilized | Variable, Potential for Oxidation |
| Indication | High Triglycerides + CVD Risk | General Wellness / Dietary Gap |
| Insurance Coverage | Often Covered (Prior Auth) | Out-of-Pocket Expense |
Regulatory Discrepancies Between FDA and EMA Guidelines
Geo-epidemiological bridging reveals significant disparities in patient access. In the United States, the FDA maintains approval for icosapent ethyl specifically for risk reduction in statin-treated adults. However, the European Medicines Agency (EMA) has been more conservative, often restricting reimbursement to severe hypertriglyceridemia cases only. This impacts local patient access; a patient in London may not qualify for the same pharmacological intervention as a patient in Morrisville, NC, despite identical clinical profiles.
Public health officials emphasize that supplements are not held to the same Good Manufacturing Practices (GMP) as pharmaceuticals. Variability in capsule content means a patient might consume rancid oil unknowingly.
“Patients should understand that dietary supplements are not evaluated for safety and efficacy by the FDA in the same way as prescription drugs,”
states a consistent warning from the FDA Office of Dietary Supplements. This regulatory gap necessitates physician oversight before initiating high-dose regimens.
Contraindications & When to Consult a Doctor
While generally safe, omega-3 supplementation carries specific risks that require triage. Patients with a known hypersensitivity to fish or shellfish should avoid marine-derived products, though synthetic algae-based options exist. More critically, high doses (greater than 4 grams daily) have been associated with an increased incidence of atrial fibrillation, a chaotic heart rhythm that increases stroke risk.
due to mild antiplatelet effects, patients scheduled for surgical procedures should discontinue employ at least seven days prior to incision to minimize bleeding complications. Those currently taking warfarin or direct oral anticoagulants must consult their cardiologist, as additive effects could alter INR stability. If you experience palpitations, unexplained bruising, or gastrointestinal distress, discontinue use and seek professional medical intervention immediately.
The trajectory for omega-3 therapy is moving toward precision medicine. Future guidelines in late 2026 will likely incorporate genetic markers to determine who metabolizes EPA most effectively. Until then, the consensus remains clear: prioritize whole food sources like fatty fish for general health, and reserve high-dose supplementation for confirmed hypertriglyceridemia under strict medical supervision.
References
- Bhatt DL, et al. Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. NEJM. 2019.
- Nicholls SJ, et al. Effect of High-Dose Omega-3 Fatty Acids vs Corn Oil on Major Adverse Cardiovascular Events. JAMA. 2020.
- American Heart Association. Fish and Omega-3 Fatty Acids. 2025 Update.
- U.S. Food and Drug Administration. Dietary Supplements. 2026.
- VITAL Study. Long-term Follow-up Results. Brigham and Women’s Hospital.