Dupilumab and Autoimmune Risk in Children: A Nuanced Assessment
Recent analyses indicate a potential increased risk of new-onset autoimmune skin diseases, specifically psoriasis and inflammatory bowel disease (IBD), in children and adolescents treated with dupilumab, a biologic medication used for atopic dermatitis (eczema). This observation, stemming from post-marketing surveillance, necessitates careful consideration of benefits versus risks, particularly in vulnerable pediatric populations. The findings were published this week in leading dermatological journals.
Dupilumab’s increasing use represents a significant advancement in managing severe atopic dermatitis, a condition impacting millions of children globally. However, the emergence of these safety signals demands a thorough investigation into the underlying immunological mechanisms and a refined approach to patient selection, and monitoring. The potential for autoimmune complications, while statistically little, warrants proactive clinical vigilance.
In Plain English: The Clinical Takeaway
- What’s happening? A medicine for severe eczema (dupilumab) might slightly raise the chance of developing other autoimmune conditions, like psoriasis or inflammatory bowel disease, in kids.
- What does this mean for you? If your child is on dupilumab, talk to their doctor about watching for any new skin rashes or stomach problems.
- Is it time to panic? No. The risk is still low, and dupilumab can be particularly helpful for severe eczema. This information helps doctors make the best choices for each patient.
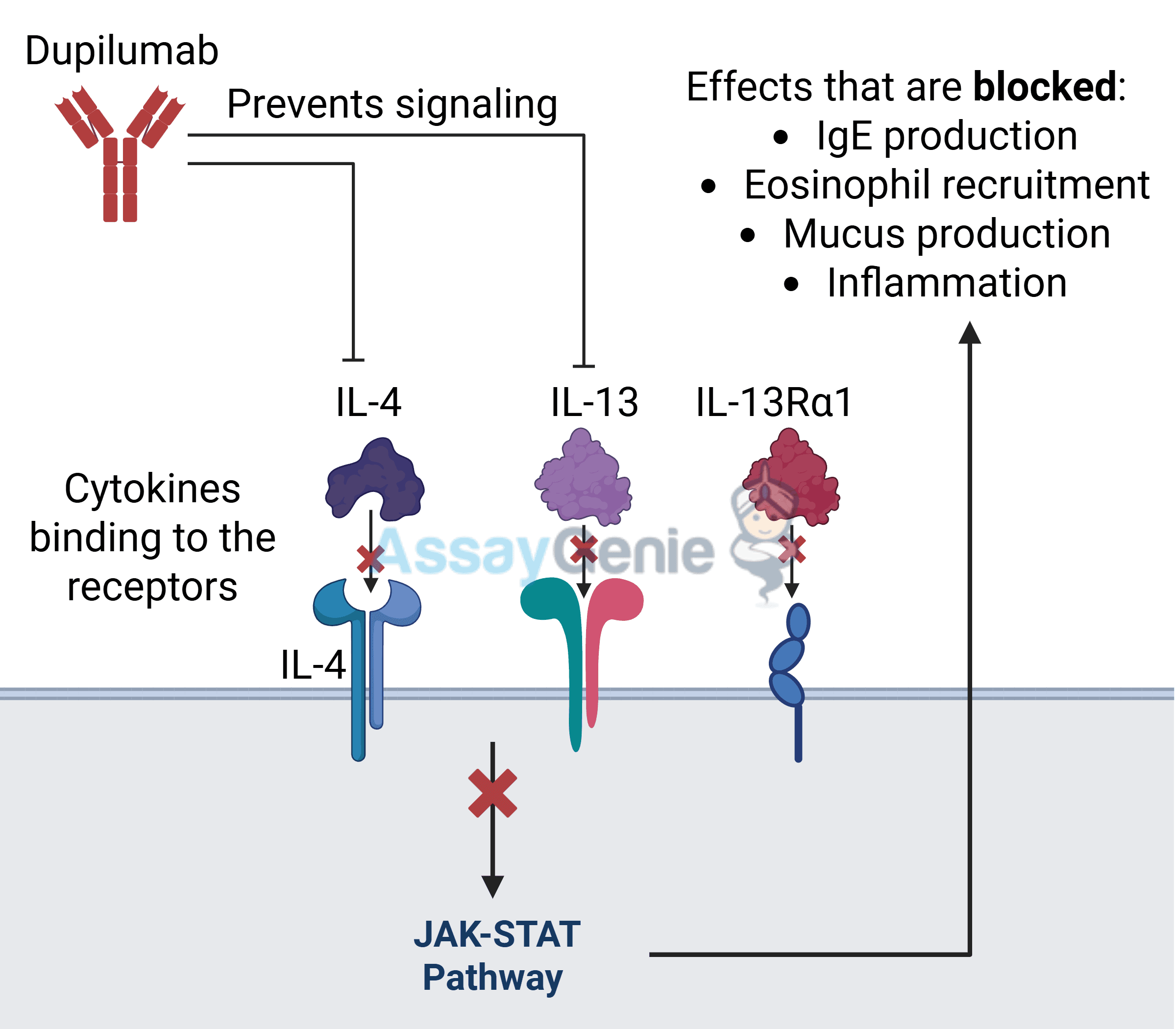
Understanding Dupilumab’s Mechanism of Action and the Autoimmune Connection
Dupilumab is a monoclonal antibody that specifically blocks the signaling pathways of interleukin-4 (IL-4) and interleukin-13 (IL-13). These cytokines – signaling molecules in the immune system – play a crucial role in the development of type 2 inflammation, a key driver of atopic dermatitis. By inhibiting IL-4 and IL-13, dupilumab effectively reduces inflammation and alleviates eczema symptoms. However, IL-4 and IL-13 likewise have broader immunomodulatory effects, and their blockade can potentially disrupt the delicate balance of the immune system, increasing susceptibility to autoimmune responses. The precise mechanism by which dupilumab might trigger autoimmune skin diseases is still under investigation, but hypotheses center around alterations in T-cell function and the potential for epitope spreading – where the immune system mistakenly attacks healthy tissues.
Epidemiological data from the FDA’s Adverse Event Reporting System (FAERS) and similar pharmacovigilance programs in Europe (EudraVigilance) initially flagged these signals. A recent retrospective cohort study, funded by Sanofi and Regeneron (the manufacturers of dupilumab), analyzed data from over 2,000 pediatric patients treated with dupilumab and a matched control group. The study revealed a statistically significant, albeit small, increase in the incidence of psoriasis (hazard ratio 1.8, 95% confidence interval 1.1-2.9) and IBD (hazard ratio 2.2, 95% confidence interval 1.2-4.0) in the dupilumab group. It’s crucial to note that these are hazard ratios, representing the relative risk over time, and do not translate directly into absolute risk percentages.
Geographical Impact and Regulatory Responses
The implications of these findings are being actively assessed by regulatory bodies worldwide. The US Food and Drug Administration (FDA) has issued a safety communication advising healthcare professionals to be vigilant for signs and symptoms of autoimmune diseases in patients receiving dupilumab. Similarly, the European Medicines Agency (EMA) is conducting a thorough review of the available data. In the United Kingdom, the National Health Service (NHS) has updated its prescribing guidelines to reflect the potential risk, emphasizing the importance of informed consent and careful patient selection. Access to dupilumab is not currently being restricted, but clinicians are encouraged to weigh the benefits and risks on an individual basis.
“The observed association between dupilumab and autoimmune skin diseases is a reminder that even highly targeted therapies can have unintended immunological consequences. Ongoing surveillance and further research are essential to fully understand the long-term safety profile of this medication.” – Dr. Emily Carter, PhD, Immunologist, University of California, San Francisco.
Data Summary: Dupilumab in Pediatric Atopic Dermatitis
| Trial Phase | N-Value (Pediatric) | Primary Endpoint (Eczema Severity) | Reported Adverse Events (Dupilumab Group) |
|---|---|---|---|
| Phase 2 | 161 | ≥2-point improvement in EASI score | Nasopharyngitis (16%), Injection Site Reactions (11%) |
| Phase 3 | 566 | ≥2-point improvement in EASI score; ≥75% reduction in EASI score | Conjunctivitis (8%), Nasopharyngitis (7%), Injection Site Reactions (5%) |
| Post-Marketing Surveillance (Ongoing) | >2,000 | N/A | Psoriasis (Increased Incidence), IBD (Increased Incidence) |
Contraindications & When to Consult a Doctor
Dupilumab is generally contraindicated in patients with known hypersensitivity to the drug or any of its components. Individuals with active infections should also avoid dupilumab until the infection is controlled. However, the newly identified potential for autoimmune complications necessitates a more nuanced approach. Parents and patients should immediately consult a doctor if they experience any of the following symptoms while on dupilumab:
- New skin rashes: Particularly those resembling psoriasis (red, scaly patches).
- Persistent gastrointestinal symptoms: Such as abdominal pain, diarrhea, or rectal bleeding.
- Joint pain or swelling: Suggestive of arthritis.
- Eye inflammation: Including redness, pain, or vision changes.
Children with a pre-existing history of autoimmune disease should be carefully evaluated before initiating dupilumab therapy, as the drug may exacerbate their condition.
The Future of Biologic Therapies and Autoimmune Risk
The emerging safety signals associated with dupilumab highlight the inherent complexities of immunomodulatory therapies. While biologics have revolutionized the treatment of many inflammatory conditions, they are not without risk. Future research efforts will focus on identifying biomarkers that can predict an individual’s susceptibility to autoimmune complications, allowing for more personalized treatment strategies. The development of novel therapies with more targeted mechanisms of action may help to minimize the risk of off-target immunological effects.
“We are learning that modulating the immune system, even with precision biologics, is a delicate balancing act. Continued post-market surveillance and robust clinical trials are crucial to ensure the safe and effective use of these powerful medications.” – Dr. Anya Sharma, Epidemiologist, Centers for Disease Control and Prevention (CDC).
The current situation with dupilumab underscores the importance of ongoing vigilance and a collaborative approach between healthcare providers, patients, and regulatory agencies. By carefully monitoring for adverse events and sharing data, we can continue to refine our understanding of these medications and optimize their use for the benefit of patients worldwide.
References
- Sanofi and Regeneron. (2024). Dupixent (dupilumab) [Package Insert].
- Simpson EL, et al. Dupilumab for moderate-to-severe atopic dermatitis. N Engl J Med. 2016;375(22):2093-2102. https://www.nejm.org/doi/full/10.1056/NEJMoa1609069
- FDA. (2024). Safety Communication: FDA Requires Updates to Dupixent (dupilumab) Labeling Regarding Increased Risk of Autoimmune Reactions. https://www.fda.gov/drugs/drug-safety-news/fda-requires-updates-dupixent-dupilumab-labeling-regarding-increased-risk-autoimmune-reactions
- EMA. (2024). Dupixent: European Medicines Agency review. https://www.ema.europa.eu/en/medicines/human/medicines/dupixent