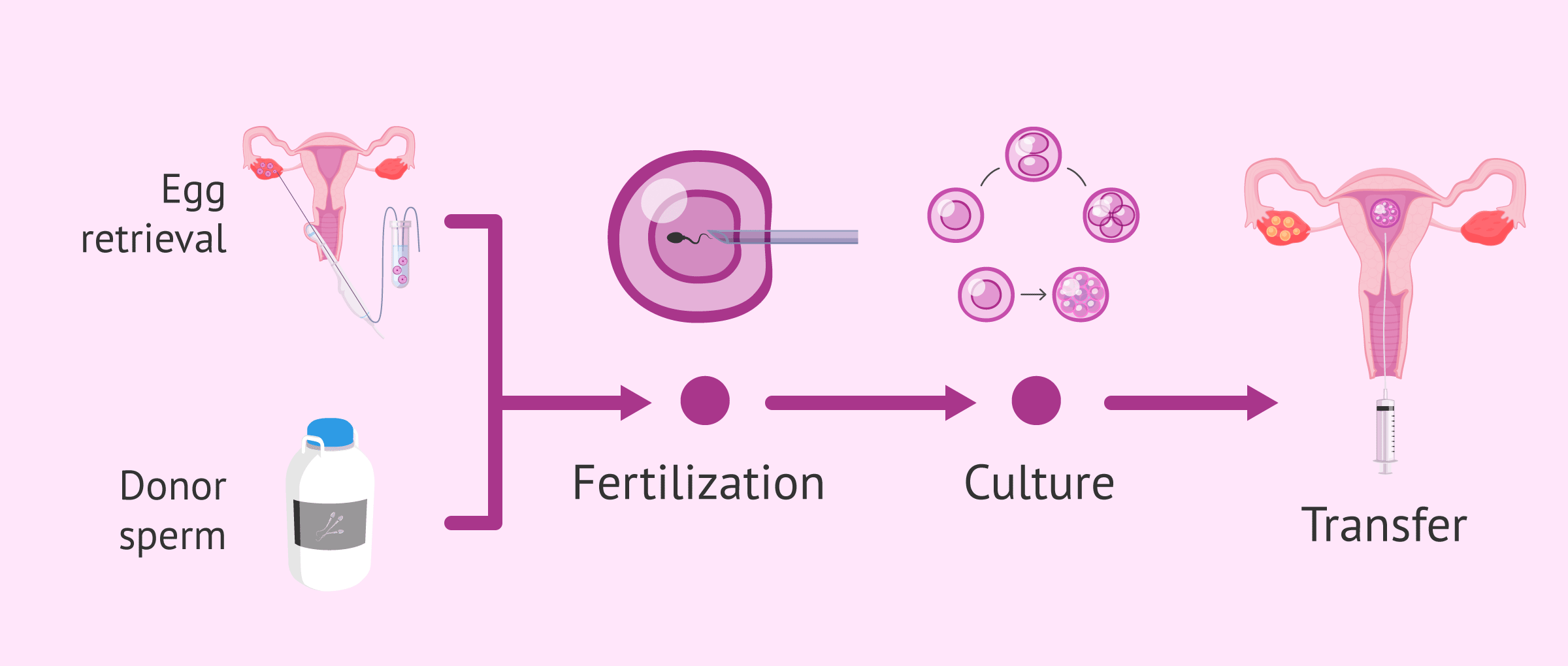
A recent legal recognition of a lesbian couple’s parental status highlights the medical reality of reciprocal IVF. This procedure allows one partner to provide eggs while the other carries the pregnancy. Understanding the clinical protocol, safety profiles, and regulatory landscape is essential for patients considering this path to parenthood in 2026.
The intersection of family law and reproductive medicine often obscures the physiological complexities involved. When news breaks regarding parental recognition, the underlying medical technology—specifically assisted reproductive technology (ART)—requires scrutiny. For patients, this legal milestone translates to tangible changes in health record accessibility and insurance coverage for fertility treatments. As we navigate this week’s developments, the focus must remain on clinical efficacy and patient safety rather than solely on social validation.
In Plain English: The Clinical Takeaway
- Shared Biological Connection: Reciprocal IVF enables one partner to contribute genetically (via egg) while the other experiences pregnancy, sharing the medical journey.
- Standard Safety Profile: The risks mirror standard IVF, including ovarian hyperstimulation and multiple gestation, requiring strict monitoring.
- Regulatory Compliance: Donor sperm used in these procedures must meet FDA or EMA infectious disease screening standards to ensure safety.
Mechanism of Action: Oocyte Retrieval and Embryo Transfer
The procedure described in recent cases involves a synchronized hormonal protocol. The genetic partner undergoes ovarian stimulation using gonadotropins to induce multifollicular development. This process aims to retrieve multiple oocytes, increasing the probability of viable embryo formation. Once retrieved, the oocytes are fertilized in vitro using sperm from an anonymous donor.
Following fertilization, the resulting embryos are cultured to the blastocyst stage. The gestational partner then undergoes endometrial preparation, often involving estrogen and progesterone supplementation to ensure receptivity. This hormonal synchronization is critical. the endometrium must be thickened and vascularized to support implantation. Endometrial receptivity refers to the specific window when the uterine lining can accept an embryo, typically lasting only a few days per cycle.
Recent data suggests that success rates for reciprocal IVF correlate strongly with the age of the egg provider rather than the gestational carrier. This distinction is vital for clinical counseling. Patients often assume the carrier’s health dictates success, but embryonic chromosomal normality is the primary driver of implantation potential.
Geo-Epidemiological Bridging and Regulatory Oversight
Regulatory frameworks vary significantly by region, impacting patient access and safety protocols. In the United States, the FDA regulates donor sperm under 21 CFR Part 1271, mandating quarantine and infectious disease testing. This ensures that pathogens like HIV or Hepatitis are not transmitted during the insemination process.
Conversely, the European Medicines Agency (EMA) and the Human Fertilisation and Embryology Authority (HFEA) in the UK enforce strict limits on the number of families a single donor can serve. This reduces the risk of accidental consanguinity in the population. For patients seeking care across borders, understanding these discrepancies is crucial. A treatment protocol approved in one jurisdiction may not meet the safety standards of another.
Funding for these procedures often comes from private clinics, though public health systems are increasingly recognizing the medical necessity of ART for same-sex couples. Transparency regarding clinic funding sources helps patients identify potential biases in success rate reporting.
“The clinical priority in reciprocal IVF remains the health of the gestational carrier and the viability of the embryo. Legal recognition supports this by ensuring both parents have access to critical medical history.” — American Society for Reproductive Medicine (ASRM) Ethics Committee
Comparative Clinical Metrics: Reciprocal vs. Standard IVF
Understanding the statistical landscape helps manage patient expectations. The following table outlines key differences in clinical parameters between reciprocal IVF and standard autologous IVF.
| Parameter | Reciprocal IVF | Standard Autologous IVF |
|---|---|---|
| Genetic Source | Partner A (Egg) + Donor (Sperm) | Partner (Egg) + Partner/Donor (Sperm) |
| Gestational Carrier | Partner B | Partner (Self) |
| Ovarian Stimulation Risk | Applies to Egg Provider | Applies to Patient |
| Live Birth Rate (Age <35) | ~45-50% per transfer | ~45-50% per transfer |
| Regulatory Screening | Donor + Both Partners | Both Partners |
As shown, the live birth rates are comparable when controlling for the age of the oocyte provider. However, the logistical complexity increases in reciprocal IVF due to the need for synchronization between two individuals. This requires precise timing of menstrual cycles, often achieved through oral contraceptives or GnRH agonists.
Contraindications & When to Consult a Doctor
While reciprocal IVF is a safe option for many, specific medical contraindications exist. Patients with severe untreated hydrosalpinx (fluid-filled fallopian tubes) may experience reduced implantation rates and should seek surgical intervention prior to transfer. Individuals with a history of severe Ovarian Hyperstimulation Syndrome (OHSS) require modified stimulation protocols to prevent life-threatening fluid shifts.
Consult a reproductive endocrinologist if you experience severe abdominal pain, rapid weight gain, or shortness of breath during stimulation. These are hallmark signs of OHSS. Patients with uncontrolled thyroid dysfunction or diabetes must achieve metabolic stability before embryo transfer to minimize miscarriage risk. Pre-conception counseling is mandatory to review family medical history, even when using donor gametes.
Future Trajectory and Patient Advocacy
The legal recognition of both partners sets a precedent for medical record accuracy. Historically, non-biological parents faced barriers in accessing pediatric health information. Resolving this ensures continuity of care for the child. From a public health perspective, accurate parental data improves epidemiological tracking of genetic conditions.
As we move through 2026, the integration of genetic screening (PGT-A) continues to refine embryo selection, reducing miscarriage rates. Patients should advocate for comprehensive counseling that covers both the legal and physiological dimensions of treatment. The goal is not just pregnancy, but a healthy outcome for the carrier and the child.
References
- Centers for Disease Control and Prevention: Assisted Reproductive Technology
- American Society for Reproductive Medicine: Practice Committees
- European Society of Human Reproduction and Embryology: Guidelines
- PubMed Central: Reciprocal IVF Outcomes
- FDA: Regulation of Human Cells, Tissues, and Cellular and Tissue-Based Products