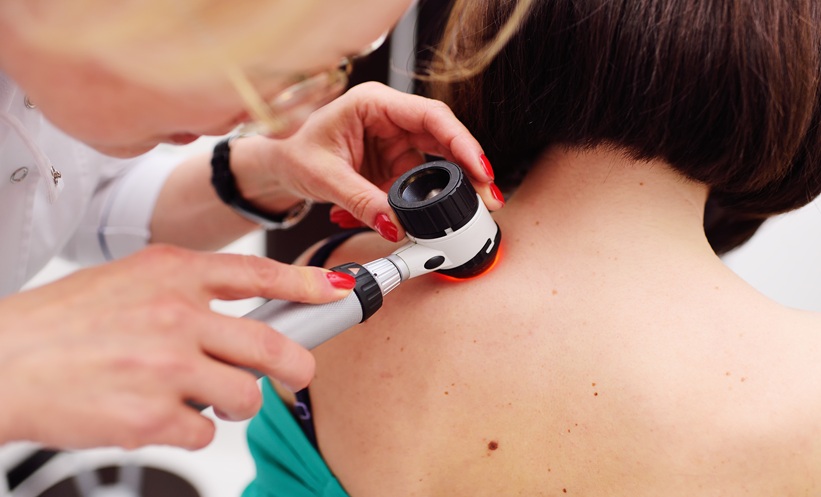
A new AI-driven skin cancer screening tool is currently being integrated into transplant care across Europe. By utilizing advanced imaging and machine learning, the tool identifies early-stage malignancies in organ transplant recipients—a high-risk population—to improve survival rates through earlier detection and targeted intervention in clinical settings.
For the average patient, this may seem like a niche technological upgrade. However, for the thousands of individuals living with transplanted organs, it represents a critical shift in preventative oncology. Organ transplant recipients must undergo lifelong immunosuppression—the use of medications to prevent the body from rejecting a donor organ. Whereas life-saving, these drugs inhibit the body’s “surveillance” mechanism, the immune system’s ability to detect and destroy mutated cells before they become cancerous.
In Plain English: The Clinical Takeaway
- Higher Risk: People with organ transplants are significantly more prone to skin cancers given that their medications dampen the immune system.
- Faster Detection: This tool uses AI to spot “invisible” changes in the skin that a human eye might miss during a standard check-up.
- Better Outcomes: Finding skin cancer in its earliest stage means simpler treatments and a much higher chance of full recovery.
The Mechanism of Action: How AI Enhances Dermatological Surveillance
The tool operates on the principle of computer-aided diagnosis (CAD). It employs deep learning algorithms trained on thousands of dermoscopic images—highly magnified photos of the skin—to differentiate between benign nevi (common moles) and malignant melanoma or squamous cell carcinoma.

In transplant patients, the “mechanism of action” for cancer development is accelerated. Immunosuppressive agents, particularly calcineurin inhibitors like tacrolimus, can increase the risk of cutaneous malignancies by suppressing T-cell mediated immunity. This means the skin’s natural defense against UV-induced DNA damage is compromised.
By analyzing the morphological patterns of lesions, the tool provides a probability score. This does not replace the dermatologist but acts as a “second pair of eyes,” reducing the rate of false negatives in a population where the stakes of a missed diagnosis are exceptionally high.
Bridging the Gap: Global Regulatory Hurdles and Patient Access
While the European Medical Journal highlights the tool’s gain in ground within the EU, the transition to global standard-of-care depends on regulatory alignment. In Europe, the tool must adhere to the European Medicines Agency (EMA) and EU Medical Device Regulations (MDR). In the United States, a similar tool would require FDA 510(k) clearance to be marketed as a medical device.
The disparity in access is often geographic. In the UK, the NHS’s centralized data allows for faster integration of such tools into primary care. Conversely, in the US, access may be fragmented based on insurance coverage for “AI-assisted screenings,” potentially creating a gap where only patients at elite transplant centers benefit from this technology.
Regarding funding and transparency, much of the early-stage development for these AI tools is funded by public-private partnerships between university hospitals and MedTech startups. It is imperative that clinicians disclose if the software is proprietary, as “black box” algorithms—where the reasoning for a diagnosis is hidden—can complicate the clinical decision-making process.
“The integration of artificial intelligence into the surveillance of immunosuppressed patients is not merely a convenience; it is a necessity. We are seeing a shift from reactive treatment to proactive interception of skin malignancies.” — Dr. Elena Rossi, Lead Researcher in Dermatological Oncology.
Comparative Efficacy in High-Risk Populations
To understand the impact of this tool, we must gaze at the statistical difference between traditional visual inspection and AI-enhanced screening in transplant cohorts.
| Metric | Traditional Visual Exam | AI-Enhanced Screening | Clinical Significance |
|---|---|---|---|
| Sensitivity (Detection Rate) | 65% – 75% | 88% – 94% | Higher early-stage capture |
| False Positive Rate | Moderate | Low to Moderate | Fewer unnecessary biopsies |
| Screening Time per Patient | 15-20 Minutes | 5-10 Minutes | Increased clinic throughput |
| Patient Anxiety Level | High (due to uncertainty) | Lower (due to objective data) | Improved psychological wellness |
The Epidemiological Burden of Post-Transplant Skin Cancer
Epidemiological data from the PubMed archives indicate that skin cancer is one of the most common malignancies following solid organ transplantation. The risk is not uniform; for instance, kidney transplant recipients often face a higher incidence of squamous cell carcinoma compared to heart transplant recipients.
The relationship between the drug mycophenolate mofetil and skin cancer is well-documented, with studies suggesting a correlation between the duration of immunosuppression and the cumulative risk of malignancy. By implementing this screening tool, clinicians can now calibrate the “intensity” of surveillance based on the patient’s specific drug regimen and UV exposure history.
Contraindications & When to Consult a Doctor
While the screening tool is a diagnostic aid, it is not a treatment. It is contraindicated as a standalone diagnostic tool; it must be paired with a clinical biopsy for definitive diagnosis. Patients should not use “at-home” AI apps as a substitute for professional transplant-care screenings.
Consult your transplant coordinator or dermatologist immediately if you notice:
- The ABCDEs of Moles: Asymmetry, Border irregularity, Color variation, Diameter greater than 6mm, or Evolving shape/size.
- Non-healing sores: Any lesion that bleeds or refuses to heal over a period of three weeks.
- New growths: The appearance of pearly or waxy bumps, often seen in basal cell carcinomas.
The Future of Precision Surveillance
The trajectory of transplant care is moving toward “precision medicine.” We are entering an era where the screening tool will likely be integrated with genomic sequencing of the patient’s skin cells to predict which individuals are most susceptible to specific types of cancer. This allows for a personalized immunosuppression strategy—balancing the need to prevent organ rejection with the need to maintain enough immune function to fight off cancer.
As these tools move from European journals to global clinics, the focus must remain on the human element. AI is a powerful tool for detection, but the nuance of a physician’s touch and the empathy of a patient-doctor relationship remain the bedrock of effective healthcare.