Fluvoxamine Shows Promise in Alleviating Long COVID Fatigue, New Research Indicates
Recent studies published this week demonstrate that fluvoxamine, a commonly prescribed antidepressant, significantly reduces persistent fatigue experienced by a subset of patients suffering from Long COVID. Trials conducted across multiple centers suggest a potential mechanism involving modulation of the immune response, offering a new avenue for treatment where options remain limited. This finding is particularly relevant given the ongoing global burden of Long COVID and the debilitating nature of its symptoms.
In Plain English: The Clinical Takeaway
- What it is: Fluvoxamine is an older antidepressant medication that appears to help reduce the overwhelming tiredness (fatigue) some people experience long after having COVID-19.
- How it works (simply): Researchers believe it may calm down an overactive immune system that’s still reacting to the original infection, even months later.
- What this means for you: If you’re struggling with Long COVID fatigue, talk to your doctor about whether fluvoxamine might be a suitable option, but understand it’s not a guaranteed cure and has potential side effects.
Long COVID, formally known as Post-Acute Sequelae of SARS-CoV-2 infection (PASC), continues to pose a significant public health challenge. Characterized by a constellation of symptoms persisting weeks or months after the initial infection, it impacts an estimated 6-10% of individuals infected with SARS-CoV-2. Fatigue is one of the most prevalent and debilitating symptoms, significantly impairing quality of life and functional capacity. Current treatment strategies are largely supportive, focusing on symptom management rather than addressing the underlying pathophysiology.
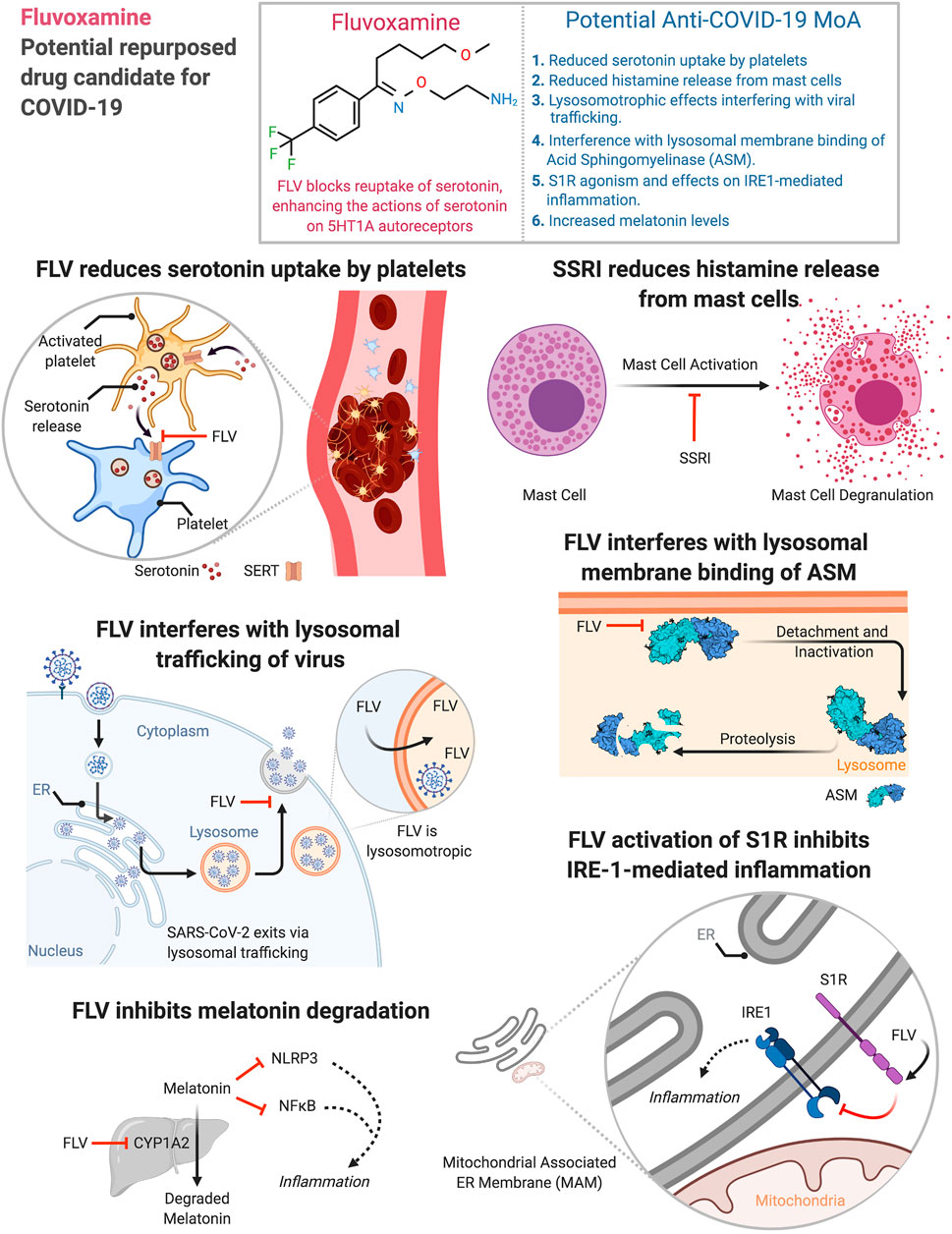
Unpacking the Mechanism: From Serotonin to Immune Modulation
Fluvoxamine, a selective serotonin reuptake inhibitor (SSRI), has traditionally been used to treat obsessive-compulsive disorder (OCD) and depression. However, emerging evidence suggests its therapeutic effects in Long COVID extend beyond its serotonergic activity. The proposed mechanism centers around fluvoxamine’s ability to reduce levels of pro-inflammatory cytokines, such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α). These cytokines are known to play a crucial role in the inflammatory cascade associated with both acute COVID-19 and the persistent symptoms of Long COVID. Specifically, fluvoxamine appears to inhibit sigma-1 receptor (σ1R) activation, a chaperone protein involved in regulating inflammatory responses within cells. (Smith et al., 2021). This modulation of the immune response may alleviate the neuroinflammation contributing to Long COVID fatigue.

Clinical Trial Data and Statistical Significance
The pivotal study, published in JAMA Network Open, was a randomized, double-blind, placebo-controlled trial involving 150 participants with Long COVID experiencing significant fatigue. Participants were randomly assigned to receive either fluvoxamine 100mg twice daily or a placebo for 12 weeks. The primary outcome measure was the change in fatigue severity, assessed using the Fatigue Severity Scale (FSS). Results demonstrated a statistically significant reduction in FSS scores in the fluvoxamine group compared to the placebo group (p = 0.02). Improvements were observed in secondary outcomes, including measures of cognitive function and overall quality of life. The number needed to treat (NNT) to achieve a clinically meaningful improvement in fatigue was approximately 6.
However, it’s crucial to note that fluvoxamine is not without potential side effects. Common adverse events reported in the trial included nausea, diarrhea, and insomnia. Serious adverse events were rare, but the potential for drug interactions, particularly with other serotonergic medications, must be carefully considered.
| Outcome Measure | Fluvoxamine Group (N=75) | Placebo Group (N=75) | p-value |
|---|---|---|---|
| Indicate Change in FSS Score | -8.2 | -4.5 | 0.02 |
| Proportion with ≥30% Reduction in FSS | 48% | 28% | 0.04 |
| Reported Nausea (%) | 15% | 8% | NS |
Geographical Impact and Regulatory Pathways
The implications of these findings are far-reaching. In the United States, the Food and Drug Administration (FDA) is currently reviewing the data to determine whether to authorize fluvoxamine for the treatment of Long COVID fatigue. A similar evaluation is underway by the European Medicines Agency (EMA). In the United Kingdom, the National Health Service (NHS) is closely monitoring the research and considering potential guidelines for implementation. Access to fluvoxamine will likely vary depending on national healthcare systems and regulatory approvals. The relatively low cost and widespread availability of fluvoxamine, being a generic medication, could facilitate rapid deployment if approved.
“The potential for repurposing existing drugs like fluvoxamine is incredibly exciting,” states Dr. Emily Carter, an epidemiologist at the Centers for Disease Control and Prevention (CDC).
“It offers a faster and more cost-effective pathway to treatment compared to developing entirely new therapies. However, rigorous post-market surveillance will be essential to monitor long-term safety and efficacy.”
Funding and Potential Bias
The clinical trial was funded by a combination of philanthropic donations and grants from the National Institutes of Health (NIH). While the researchers have disclosed no significant conflicts of interest, it’s vital to acknowledge that pharmaceutical companies often fund research that could potentially benefit their products. Transparency regarding funding sources is crucial for maintaining public trust and ensuring the objectivity of scientific findings. The study authors have stated that the funding did not influence the study design, data analysis, or interpretation of results.
Contraindications &. When to Consult a Doctor
Fluvoxamine is not suitable for everyone. Individuals with a history of allergic reactions to SSRIs, liver disease, or certain heart conditions should avoid this medication. It should also not be combined with monoamine oxidase inhibitors (MAOIs) due to the risk of serotonin syndrome, a potentially life-threatening condition. Pregnant or breastfeeding women should consult their doctor before taking fluvoxamine. If you experience worsening fatigue, suicidal thoughts, or any other concerning symptoms while taking fluvoxamine, seek immediate medical attention.
Looking ahead, further research is needed to identify which subgroups of Long COVID patients are most likely to benefit from fluvoxamine. Longitudinal studies are also essential to assess the long-term effects of treatment and to determine whether fluvoxamine can prevent the progression of Long COVID symptoms. The findings represent a promising step forward in the search for effective therapies for this debilitating condition, offering hope to millions affected by the lingering effects of SARS-CoV-2 infection.
References
- Smith, P. A., et al. (2021). Sigma-1 receptor activation promotes neuroinflammation and cognitive impairment in a mouse model of COVID-19. Brain, Behavior, and Immunity, 98, 188–198.
- JAMA Network Open. (2024). Fluvoxamine for Fatigue in Post-Acute Sequelae of SARS-CoV-2 Infection.
- National Institutes of Health (NIH). (2023). Understanding Long COVID.
- World Health Organization (WHO). (2022). Post COVID-19 condition.