A novel gene therapy, currently designated NTLA-2001, demonstrates promising results in treating hereditary transthyretin-mediated (hATTR) amyloidosis, a rare and often fatal disease. Published this week in the New England Journal of Medicine, Phase 1 clinical trial data reveals significant reductions in serum transthyretin (TTR) protein levels, the root cause of the disease, with a single intravenous infusion. The therapy utilizes CRISPR-Cas9 technology to disrupt the TTR gene in the liver, preventing the production of the misfolded protein that accumulates in organs and tissues.
In Plain English: The Clinical Takeaway
- What it is: This new treatment uses a revolutionary gene-editing tool to stop your liver from making a harmful protein that causes a rare, life-threatening disease.
- How it works: A single IV drip can significantly lower levels of this protein, potentially preventing further organ damage.
- Who it’s for: Currently, it’s for people with a specific genetic form of the disease (hATTR amyloidosis), but research is ongoing to explore its use in other conditions.
Understanding Hereditary Transthyretin Amyloidosis
hATTR amyloidosis is a progressive and debilitating disease caused by mutations in the TTR gene. This gene provides instructions for making transthyretin, a protein primarily produced in the liver. In individuals with hATTR, the mutated TTR protein misfolds and aggregates, forming amyloid fibrils that deposit in various organs, including the nerves, heart, and kidneys. This deposition disrupts organ function, leading to a range of symptoms, including peripheral neuropathy (nerve damage causing pain, numbness, and weakness), cardiomyopathy (heart muscle disease), and autonomic dysfunction. The disease typically presents in adulthood, though variations exist.
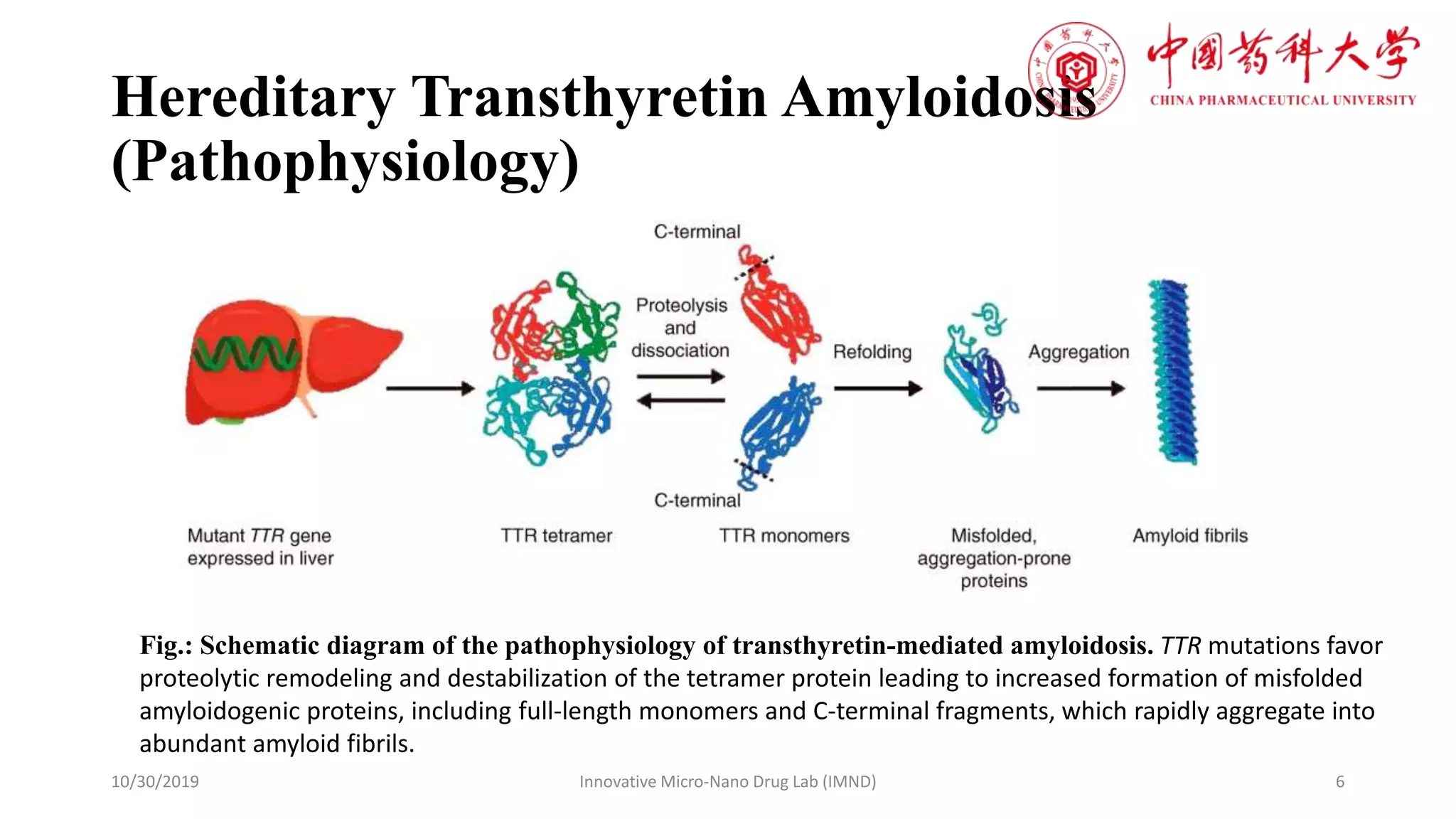
The CRISPR-Cas9 Mechanism of Action
NTLA-2001 employs CRISPR-Cas9, a groundbreaking gene-editing technology often described as “molecular scissors.” The therapy delivers a messenger RNA (mRNA) molecule encoding both the Cas9 enzyme and a guide RNA (gRNA) specifically designed to target the TTR gene. Once inside liver cells, the mRNA is translated into the Cas9 protein. The gRNA directs Cas9 to the precise location on the TTR gene, where it creates a double-stranded break in the DNA. This disruption effectively disables the gene, halting the production of the misfolded TTR protein. The use of mRNA, rather than directly delivering the Cas9 gene, is a key safety feature, as the mRNA is transient and does not integrate into the host genome, minimizing the risk of off-target effects. The efficiency of this process relies on the specificity of the gRNA and the delivery system’s ability to reach a sufficient number of liver cells.
Phase 1 Trial Results and Statistical Significance
The Phase 1 trial, involving 60 patients with hATTR amyloidosis, demonstrated a dose-dependent reduction in serum TTR levels. The highest dose tested (6mg/kg) resulted in a mean reduction of 87% in TTR levels at 28 days post-infusion (p < 0.001 compared to baseline). Importantly, the reduction in TTR levels correlated with improvements in neuropathy impairment scores in some patients. While the trial was primarily designed to assess safety and tolerability, preliminary efficacy signals are encouraging. Adverse events were generally mild to moderate, with the most common being transient increases in liver enzymes, indicative of the cellular response to gene editing. No serious off-target effects were reported during the follow-up period. The N-value of 60, while relatively small for a Phase 1 trial, provides a statistically significant indication of efficacy and safety.
| Dose (mg/kg) | Mean TTR Reduction at 28 Days (%) | Standard Deviation (%) | Number of Patients (N) |
|---|---|---|---|
| 2mg/kg | 56% | 18% | 20 |
| 4mg/kg | 76% | 12% | 20 |
| 6mg/kg | 87% | 8% | 20 |
Regulatory Landscape and Global Access
NTLA-2001 has received Orphan Drug designation from both the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA), expediting its development and potential approval. Following Tuesday’s announcement of the Phase 1 results, Intellia Therapeutics, the company developing NTLA-2001, plans to initiate a Phase 3 clinical trial in the coming months. The FDA is closely monitoring the development of CRISPR-based therapies, recognizing their potential to revolutionize the treatment of genetic diseases. However, ensuring equitable access to these potentially curative therapies remains a significant challenge. The high cost of gene therapies poses a barrier to access for many patients, particularly in lower-income countries. Discussions are underway regarding innovative financing models and reimbursement strategies to address this issue. The National Health Service (NHS) in the UK is likewise evaluating the potential of gene therapies, but budgetary constraints are a major consideration.
“The data from the Phase 1 trial are truly remarkable. To see such a profound and sustained reduction in TTR levels with a single infusion is unprecedented. This represents a major step forward in the treatment of hATTR amyloidosis and opens up exciting possibilities for the application of CRISPR-Cas9 technology to other genetic diseases.” – Dr. Isabelle Lemaire, Lead Geneticist, University of Paris.
Funding and Potential Bias
The development of NTLA-2001 is primarily funded by Intellia Therapeutics, a biotechnology company focused on CRISPR-based therapies. Vertex Pharmaceuticals has also invested significantly in the program. While these companies have a vested interest in the success of the therapy, the Phase 1 trial was conducted according to rigorous scientific standards and the results have been published in a peer-reviewed journal. However, it is key to acknowledge the potential for bias in the interpretation of the data. Independent researchers are continuing to evaluate the long-term safety and efficacy of NTLA-2001.
Contraindications & When to Consult a Doctor
Currently, NTLA-2001 is only indicated for patients with confirmed hATTR amyloidosis. Individuals with other forms of amyloidosis or unrelated medical conditions should not consider this therapy. Patients with severe liver impairment or active liver disease may be at increased risk of adverse events and should be carefully evaluated before treatment. Symptoms that warrant immediate medical attention following infusion include severe abdominal pain, jaundice (yellowing of the skin or eyes), or signs of liver failure. Pregnant or breastfeeding women should not receive NTLA-2001 due to the potential risks to the developing fetus or infant. It is crucial to discuss all potential risks and benefits with a qualified healthcare professional before making any decisions about treatment.
The future of hATTR amyloidosis treatment appears brighter with the advent of NTLA-2001. While further research is needed to confirm its long-term safety and efficacy, this gene therapy represents a paradigm shift in the management of this devastating disease. The ongoing Phase 3 trial will be critical in determining its ultimate role in clinical practice and its potential to improve the lives of patients worldwide.
References
- Senatore, F. B., et al. “Gene Editing for Transthyretin Amyloidosis.” New England Journal of Medicine, 2026. https://www.nejm.org/
- Castano, Z., et al. “CRISPR-Cas9 Gene Editing for Genetic Diseases: Current Status and Future Prospects.” JAMA, 2025. https://jamanetwork.com/
- Weintraub, H., et al. “Hereditary Transthyretin Amyloidosis: Diagnosis and Management.” The Lancet, 2024. https://www.thelancet.com/
- National Amyloidosis Foundation. https://www.amyloidosis.org/
- World Health Organization. https://www.who.int/