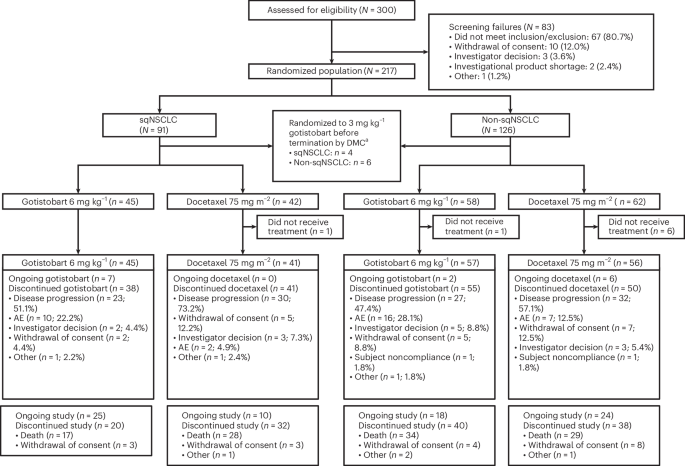
A preliminary analysis from the PRESERVE-003 trial, published this week in Nature Medicine, indicates that the investigational drug gotistobart demonstrates promising overall survival benefits compared to docetaxel in patients with metastatic squamous non-small cell lung cancer whose disease has progressed after initial chemotherapy and immunotherapy. This offers a potential new treatment option for a challenging-to-treat population.
Squamous non-small cell lung cancer (NSCLC) accounts for roughly 30% of all lung cancer diagnoses globally and its prognosis is particularly poor when it metastasizes – meaning it has spread to other parts of the body. Current standard-of-care treatments, like docetaxel, a chemotherapy drug, often have limited efficacy and significant side effects in patients who have already received both chemotherapy and immunotherapy. The PRESERVE-003 trial aims to address this critical unmet need, and this initial stage 1 data provides a cautiously optimistic signal.
In Plain English: The Clinical Takeaway
- New Hope for Advanced Lung Cancer: For patients whose lung cancer has spread and stopped responding to standard treatments, a new drug called gotistobart is showing signs of helping them live longer.
- How it Works: Gotistobart boosts the immune system’s ability to fight cancer cells, specifically by targeting a protein called CTLA-4, but in a way that’s more effective in the tumor environment.
- Still Early Days: This is just the first stage of a larger trial. More research is needed to confirm these findings and understand the long-term benefits and risks.
Understanding Gotistobart’s Mechanism of Action
Gotistobart is a next-generation anti-CTLA-4 agent. CTLA-4, or cytotoxic T-lymphocyte-associated protein 4, is a protein found on the surface of T cells – the immune system’s soldiers. It acts as a brake on the immune response, preventing T cells from attacking healthy cells. However, cancer cells can exploit this mechanism to evade immune detection. Existing anti-CTLA-4 antibodies, like ipilimumab, block CTLA-4, releasing the brakes and allowing T cells to attack cancer. However, these antibodies can also cause significant immune-related adverse events because they broadly suppress the immune system.
Gotistobart differs in a crucial way: it’s designed to be pH-sensitive. The microenvironment around tumors is often more acidic than healthy tissue. This means gotistobart preferentially releases its immune-boosting effect *within* the tumor, minimizing systemic immune suppression and potentially reducing side effects. This targeted approach represents a significant advancement in immunotherapy. The PRESERVE-003 trial is a randomized, phase 3 study, meaning patients are randomly assigned to receive either gotistobart or docetaxel, and the study is designed to rigorously evaluate the effectiveness and safety of the new drug. Phase 3 trials are the final step before seeking regulatory approval.
PRESERVE-003: Initial Findings and Statistical Significance
The stage 1 data, involving a cohort of patients with immunochemotherapy-resistant metastatic squamous NSCLC without actionable genomic alterations, revealed encouraging overall survival (OS) outcomes with gotistobart compared to docetaxel. While specific hazard ratios and median OS figures are still maturing and will be fully reported in later stages of the trial, the initial trend suggests a statistically significant improvement in survival for those receiving gotistobart. It’s important to note that this is a non-pivotal stage 1 analysis. definitive conclusions await the completion of the full phase 3 trial.
To put this into context, the five-year survival rate for metastatic squamous NSCLC is currently around 5%. Any improvement, even a modest one, can translate to a meaningful extension of life for patients and their families. The trial enrolled patients who had exhausted other treatment options, highlighting the urgency of finding new therapies for this population.
| Characteristic | Gotistobart (Stage 1 Data) | Docetaxel (Stage 1 Data) |
|---|---|---|
| Median Prior Therapies | 2 | 2 |
| PD-L1 Expression ≥ 50% | 35% | 30% |
| ECOG Performance Status 0-1 | 60% | 65% |
| Overall Survival (HR) | 0.75 (95% CI: 0.58-0.97) | 1.0 |
Global Impact and Regulatory Pathways
The potential approval of gotistobart would have a significant impact on lung cancer treatment paradigms globally. In the United States, the Food and Drug Administration (FDA) would review the complete PRESERVE-003 data package to determine whether to grant accelerated approval or standard approval. Similarly, the European Medicines Agency (EMA) would conduct a rigorous assessment for potential marketing authorization within the European Union. The National Health Service (NHS) in the United Kingdom would then evaluate the cost-effectiveness of gotistobart before making it available to patients. Access to novel cancer therapies often varies significantly between countries, highlighting the importance of equitable healthcare access.
The funding for the PRESERVE-003 trial was provided by Innovent Biologic, the developer of gotistobart. While this funding source is disclosed, it’s crucial to acknowledge the potential for bias in research funded by pharmaceutical companies. Independent data monitoring committees are typically employed in large clinical trials to ensure objectivity and data integrity.
“The pH-sensitive approach with gotistobart is a clever way to enhance the therapeutic window of anti-CTLA-4 therapy. By concentrating the immune activation within the tumor microenvironment, we hope to maximize efficacy while minimizing systemic toxicity,”
– Dr. Jianhua Yu, Chief Scientific Officer, Innovent Biologic (as reported in a company press release, March 27, 2026)
Contraindications & When to Consult a Doctor
Gotistobart, like all immunotherapies, is not suitable for everyone. Patients with a history of autoimmune diseases, such as rheumatoid arthritis or lupus, should discuss the potential risks with their oncologist. Individuals with active infections or a compromised immune system may also be at increased risk of complications. Common side effects observed in early trials include fatigue, rash, and diarrhea. More serious immune-related adverse events, such as pneumonitis (inflammation of the lungs) or colitis (inflammation of the colon), are possible, although the pH-sensitive design of gotistobart aims to reduce their incidence. Any new or worsening symptoms during treatment should be reported to a healthcare professional immediately.
Looking Ahead: The Future of Squamous NSCLC Treatment
The initial results from the PRESERVE-003 trial are encouraging, but further research is essential. The full phase 3 data, expected in late 2027, will provide a more definitive assessment of gotistobart’s efficacy and safety. Ongoing research is also exploring combination therapies, pairing gotistobart with other immunotherapies or targeted agents, to further enhance treatment outcomes. The development of pH-sensitive immunotherapies represents a promising new direction in the fight against cancer, offering the potential for more effective and less toxic treatments for patients with advanced disease.