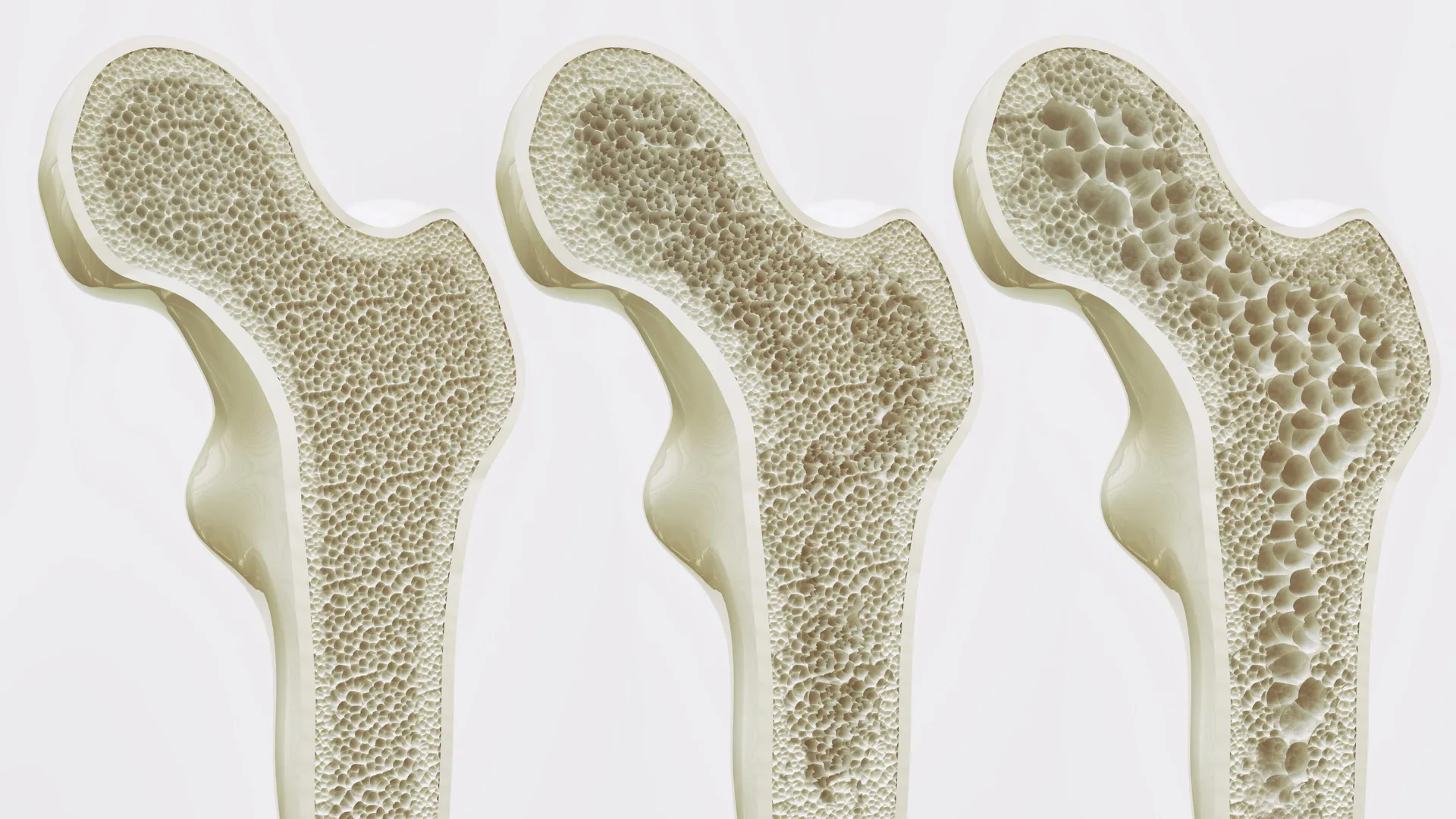
Researchers have identified the GPR133 receptor as a critical regulator of bone density. By utilizing a novel compound, AP503, scientists successfully increased bone mass and reversed osteoporosis-like degradation in murine models, potentially paving the way for human therapies that rebuild skeletal integrity rather than simply slowing bone loss.
For the millions globally living with osteoporosis—a systemic skeletal disorder characterized by compromised bone strength—this discovery represents a paradigm shift. Current gold-standard treatments, such as bisphosphonates, primarily act as antiresorptives; they stop bone from breaking down but do not effectively “build” new bone. The activation of GPR133 suggests a transition toward anabolic therapy, where the body is chemically signaled to actively restore its own structural architecture.
In Plain English: The Clinical Takeaway
- Beyond Prevention: Unlike current drugs that only slow bone loss, this new approach aims to actually rebuild lost bone density.
- The Target: Scientists found a “switch” (GPR133) in the body that, when flipped, tells the body to strengthen bones.
- Current Stage: This is early-stage research (animal models); it is not yet available for human use and requires clinical trials.
The Molecular Mechanism: How AP503 Activates GPR133
To understand this breakthrough, we must examine the mechanism of action—the specific biochemical process through which a drug produces its effect. GPR133 is a G protein-coupled receptor, a class of proteins that act as molecular switches on the surface of cells.

In the skeletal system, the balance between osteoblasts (cells that build bone) and osteoclasts (cells that break down bone) is delicate. In osteoporosis, osteoclast activity dominates. The compound AP503 acts as an agonist for GPR133, meaning it binds to the receptor and triggers a signaling cascade that favors osteoblast activity and inhibits excessive resorption.
This process effectively alters the bone remodeling cycle. By enhancing the mineralization process—the deposition of calcium and phosphate into the bone matrix—AP503 doesn’t just stop the “leak” of minerals; it refills the reservoir. This is fundamentally different from the mechanism of World Health Organization recognized standard therapies that focus on inhibiting the RANKL pathway to stop bone erosion.
From Murine Models to Global Regulatory Hurdles
While the results in mice are statistically significant, the leap to human application involves rigorous translational medicine. The research must now move from in vivo animal studies to Phase I clinical trials, where the primary focus is safety and pharmacokinetics (how the drug is absorbed and excreted by the human body).
For patients in the United States, the path to access depends on FDA approval, which requires evidence of efficacy and safety across diverse demographics. In Europe, the European Medicines Agency (EMA) will scrutinize the drug’s benefit-risk ratio, particularly regarding potential off-target effects, as G protein-coupled receptors are expressed in various tissues throughout the body.
The funding for this specific line of inquiry often stems from academic grants and biotechnology venture capital. Transparency in funding is vital; when research is funded by pharmaceutical entities, there is a risk of “publication bias,” where negative results are suppressed. However, the current findings on GPR133 have been peer-reviewed and published in high-impact journals, suggesting a high level of scientific integrity.
| Treatment Approach | Primary Action | Effect on Bone Mass | Current Clinical Status |
|---|---|---|---|
| Bisphosphonates | Antiresorptive (Stops loss) | Maintenance | FDA/EMA Approved |
| Teriparatide | Anabolic (Builds bone) | Increase | Approved (Limited use) |
| AP503 (GPR133) | Targeted Receptor Activation | Regeneration/Increase | Pre-clinical (Animal) |
Addressing the Information Gap: Epidemiological Impact
The global burden of osteoporosis is staggering. According to data from the PubMed indexed literature on skeletal health, fragility fractures are increasing globally due to an aging population. In the UK, the NHS manages thousands of hip fractures annually, each resulting in significant morbidity and healthcare costs.

“The identification of GPR133 provides a novel pharmacological target that could decouple the relationship between aging and bone loss, potentially reducing the incidence of fragility fractures by providing a more potent anabolic stimulus than current options.”
The “Information Gap” in early reporting often ignores the contraindications—reasons why a person should not receive a treatment. For instance, any drug that aggressively stimulates bone growth must be screened for osteosarcoma risk (bone cancer). If a compound over-activates bone growth pathways, it could theoretically trigger malignant cellular proliferation.
Contraindications & When to Consult a Doctor
While AP503 is not yet a prescription medication, patients currently managing bone health should be aware of the following risks associated with potent bone-altering therapies:
- Hypercalcemia: Excessive calcium levels in the blood can lead to kidney stones and cardiac arrhythmias.
- Osteosarcoma History: Patients with a history of bone malignancies must avoid potent anabolic agents unless strictly monitored.
- Renal Impairment: Those with severe kidney disease may struggle to clear these compounds, leading to toxicity.
Consult your physician immediately if: You experience a sudden onset of severe back pain (potential vertebral fracture), unexplained calcium deposits in soft tissues, or sudden changes in kidney function markers.
The Future of Skeletal Longevity
The discovery of the GPR133 receptor is not a “miracle cure,” but it is a significant scientific milestone. The transition from “stopping the rot” to “restoring the foundation” is the goal of modern orthopedics. As we move toward 2027 and beyond, the focus will shift to whether AP503 can maintain its efficacy in humans without causing systemic side effects.
For now, the most evidence-based approach to bone health remains a combination of weight-bearing exercise, adequate Vitamin D and Calcium intake and adherence to prescribed medications. We are standing on the threshold of a new era in regenerative medicine, where “strong bones for life” may move from a hopeful headline to a clinical reality.
References
- The Lancet – Endocrine and Metabolic Research Archives
- Journal of the American Medical Association (JAMA) – Clinical Trials Database
- Centers for Disease Control and Prevention (CDC) – Osteoporosis Prevalence Data
- PubMed / National Library of Medicine – G Protein-Coupled Receptor Studies