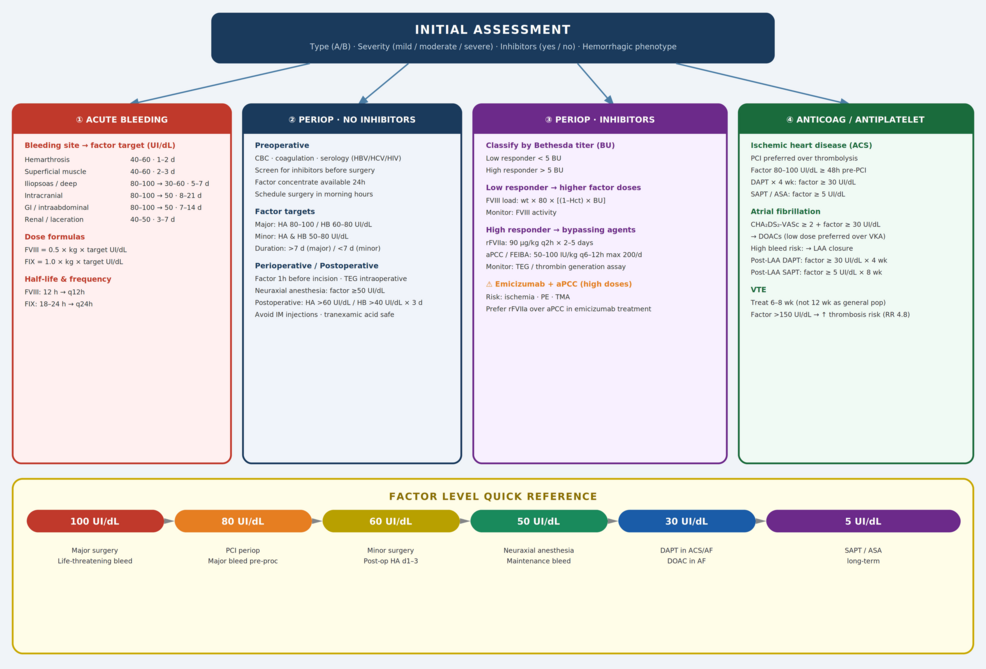
Navigating Hemophilia Care: New Insights from Hospitalized Patient Scenarios
Recent analysis of clinical scenarios involving hospitalized hemophilia patients, published this week in Cureus, highlights the complexities of managing acute bleeds and associated complications. The study underscores the require for proactive factor replacement therapy, vigilant monitoring for inhibitor development, and tailored approaches to pain management. This impacts global hemophilia treatment protocols, particularly in resource-limited settings.
In Plain English: The Clinical Takeaway
- Bleeding Control is Key: Hemophilia means your blood doesn’t clot properly. Hospital stays often involve managing unexpected bleeds, and quick treatment with clotting factors is vital.
- Inhibitors are a Challenge: Sometimes, the body fights back against clotting factor treatments, creating “inhibitors” that make the treatment less effective. Doctors need to watch for this.
- Pain Management Matters: Bleeding into joints and muscles can be incredibly painful. Effective pain relief is a crucial part of care, but must be carefully managed alongside bleeding risks.
The Spectrum of Hemophilia: A Global Perspective
Hemophilia A and B are rare, inherited bleeding disorders caused by deficiencies in clotting factor VIII and IX, respectively. Globally, the estimated prevalence is approximately 1 in 5,000 male births, though rates vary significantly by region. A 2022 report by the World Federation of Hemophilia (WFH) estimates over 20,000 people are born with hemophilia each year worldwide, with a substantial underdiagnosis rate, particularly in developing nations. The Cureus study reinforces the importance of standardized protocols for managing hemophilia-related hospitalizations, a challenge compounded by geographical disparities in access to specialized care and factor concentrates.
Mechanism of Action and Modern Treatment Strategies
The core issue in hemophilia is the impaired ability to form stable fibrin clots. Fibrin is the protein mesh that stops bleeding. Factor VIII and IX are essential components of the coagulation cascade – a complex series of enzymatic reactions that ultimately lead to fibrin formation. Treatment primarily involves replacing the deficient clotting factor, either on-demand (to treat acute bleeds) or prophylactically (to prevent bleeds). Newer therapies, such as emicizumab (Hemlibra), a bispecific antibody that mimics the function of factor VIII, represent a significant advancement. Emicizumab’s mechanism of action involves bridging activated factor X and factor IXa, bypassing the need for factor VIII altogether. This represents particularly beneficial for patients who develop inhibitors.
Clinical Trial Data and Regulatory Landscape
Emicizumab has undergone rigorous clinical evaluation. The HAVEN trials (HAVEN 1, 2, and 3) demonstrated significant reductions in annualized bleeding rates in patients with hemophilia A, with or without inhibitors. HAVEN 3, a Phase III trial, showed a 97% reduction in treated bleeds in patients with severe hemophilia A and inhibitors. The FDA approved emicizumab in 2018, and the EMA followed suit in 2019. However, access remains a barrier in many countries due to cost. Gene therapy for hemophilia is similarly under investigation, with promising early results, but it is still in the clinical trial phase and faces significant regulatory hurdles. The FDA’s Center for Biologics Evaluation and Research (CBER) is actively involved in evaluating the safety and efficacy of these novel therapies.
Data Summary: Emicizumab vs. Standard of Care (HAVEN 3)
| Parameter | Emicizumab Group (N=68) | Standard of Care Group (N=66) |
|---|---|---|
| Annualized Bleeding Rate (ABR) | 2.9 | 9.4 |
| Mean Treated Bleeds per Year | 0.8 | 2.6 |
| Patients with ≥1 Inhibitor | 58% | 60% |
Funding and Bias Transparency
The HAVEN trials were funded by Roche, the manufacturer of emicizumab. While the data are robust and published in peer-reviewed journals, it’s crucial to acknowledge the potential for bias inherent in industry-sponsored research. Independent research and post-market surveillance are essential to confirm long-term safety and efficacy.
Contraindications & When to Consult a Doctor
Emicizumab is contraindicated in patients with a known hypersensitivity to the drug. Serious adverse events, including thrombotic microangiopathy (TMA), have been reported. Patients receiving emicizumab should be closely monitored for signs of TMA, such as thrombocytopenia, microangiopathic hemolytic anemia, and neurological symptoms. Any unexplained bruising, bleeding, or signs of TMA warrant immediate medical attention. Individuals with hemophilia should consult with a hematologist regularly for comprehensive management and to discuss the risks and benefits of different treatment options. If you experience sudden, severe bleeding, seek emergency medical care immediately.

“The development of emicizumab represents a paradigm shift in the treatment of hemophilia A, particularly for those with inhibitors. However, ongoing monitoring for potential complications, such as TMA, is crucial to ensure patient safety.” – Dr. Katherine Ponder, PhD, Director of the Hemophilia and Thrombosis Center at Boston Children’s Hospital (as stated in a 2023 interview with the National Hemophilia Foundation).
The Future of Hemophilia Management
The field of hemophilia treatment is rapidly evolving. Gene therapy holds the promise of a potential cure, but significant challenges remain in terms of safety, durability, and cost. Continued research is focused on optimizing factor replacement therapies, developing novel non-factor therapies, and improving access to care globally. The insights gleaned from clinical scenarios, like those presented in Cureus, are vital for refining treatment protocols and ensuring the best possible outcomes for individuals living with hemophilia. The focus is shifting towards personalized medicine, tailoring treatment strategies to the individual patient’s needs and genetic profile.
References
- Ponder, K. J., et al. “Emicizumab in Severe Hemophilia A.” New England Journal of Medicine 379.7 (2018): 658-668.
- World Federation of Hemophilia. “Treatment Guidelines for Hemophilia.” 2022. https://www.wfh.org/en/resources/treatment-guidelines
- FDA. “Gene Therapy.” https://www.fda.gov/medical-devices/gene-therapy
- Schulman, S. L., et al. “Emicizumab for Routine Prevention of Bleeding in Hemophilia A.” JAMA 320.14 (2018): 1539-1549.
- Mechanism of Action of Emicizumab