Researchers at Heinrich Heine University Düsseldorf have mapped human lymph node immune cell organization, revealing why lymphoma disrupts tissue architecture. Published in Nature Cancer, the study identifies stromal cell reprogramming as the driver, offering new targets for stabilizing tissue structure and improving survival rates in aggressive B-cell lymphomas.
This breakthrough shifts the therapeutic paradigm from targeting malignant cells alone to preserving the “scaffolding” of the immune system. For patients facing diffuse large B-cell lymphoma (DLBCL), where tissue structure completely collapses, understanding this mechanism is critical. The loss of spatial organization is not merely a symptom of tumor growth but an active process driven by inflammatory signaling. By identifying the specific chemokine networks involved, clinicians may soon intervene to maintain lymph node integrity, potentially slowing disease progression and enhancing the efficacy of existing immunotherapies.
In Plain English: The Clinical Takeaway
- Architecture Matters: Lymph nodes require a specific structure to fight cancer; lymphoma destroys this structure actively, not just by crowding it out.
- New Drug Targets: Future treatments may focus on protecting support cells (stroma) rather than just killing cancer cells.
- Prognosis Indicator: The level of structural loss in a biopsy could soon predict how aggressive the disease will be.
The Mechanism of Stromal Reprogramming
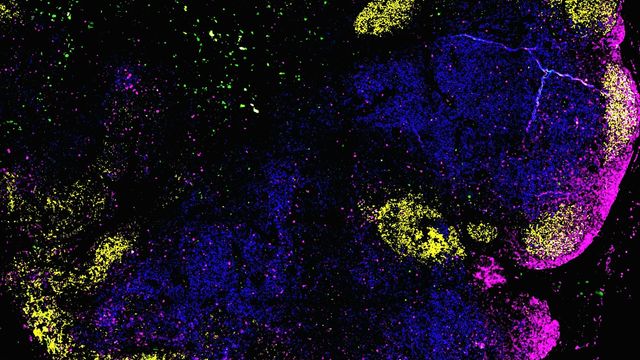
The study elucidates a specific mechanism of action wherein stromal cells, traditionally viewed as passive support structures, act as active architects. In a healthy immune response, stromal cells release chemokines—signaling proteins that guide B cells and T cells to their designated zones. However, in malignant lymphomas, T cells within the tumor microenvironment produce pro-inflammatory signaling molecules called interferons.
These interferons trigger a fundamental change in the stromal cells. Instead of producing structural chemokines that maintain organization, the stromal cells start producing inflammatory chemokines. This creates a vicious cycle: inflammatory signals attract more inflammatory cells, further degrading the tissue architecture. In slow-growing follicular lymphoma (FL), this shift is partial, preserving some spatial separation. In aggressive DLBCL, the regulatory signals are largely lost, leading to a total collapse of tissue structure. This distinction is vital for double-blind placebo-controlled trials future targeting these pathways, as patient stratification must account for baseline tissue integrity.
Regulatory Pathways and Global Patient Access
Translating these findings from the laboratory to the clinic requires navigating complex regulatory frameworks. In the United States, the Food and Drug Administration (FDA) would classify stromal-stabilizing agents as novel biologics or small molecules depending on the delivery mechanism. Current approval pathways for lymphoma treatments focus heavily on overall survival (OS) and progression-free survival (PFS). A therapy aimed at preserving tissue architecture might show benefit in delaying transformation from indolent to aggressive disease, a metric that requires long-term longitudinal study results.
In Europe, the European Medicines Agency (EMA) may prioritize these therapies under orphan drug designation if targeted at specific lymphoma subtypes. Access within the National Health Service (NHS) in the UK would depend on cost-effectiveness analyses by NICE, weighing the high cost of novel microenvironment modulators against standard chemotherapy regimens. Patients should anticipate that while the science is published in 2026, clinical availability may lag by 3 to 5 years pending Phase III trial completion.
| Lymphoma Subtype | Tissue Architecture Status | Chemokine Network | Prognostic Implication |
|---|---|---|---|
| Follicular Lymphoma (FL) | Partially Preserved | Shifted Ratios | Slower Progression |
| Diffuse Large B-Cell (DLBCL) | Collapsed | Lost Regulatory Signals | Aggressive Course |
Funding Transparency and Research Integrity
Scientific trust relies on clear disclosure of funding sources. This multicenter study was conducted through a consortium including the German Cancer Research Center (DKFZ) and the European Molecular Biology Laboratory (EMBL). Research of this magnitude typically relies on public funding grants, such as those from the German Research Foundation (DFG) or EU Horizon programs, ensuring that the primary motive remains public health advancement rather than commercial gain. Transparency in funding is essential to rule out conflicts of interest that might bias the interpretation of stromal cell behavior.
The broader scientific community recognizes the significance of the tumor microenvironment. As noted in public educational materials from the National Cancer Institute, “The tumor microenvironment plays a critical role in cancer progression and response to therapy.” This study provides the molecular evidence to support that broader consensus, moving from observation to mechanistic understanding.
“Understanding the microenvironment is key to unlocking the next generation of cancer therapies. We are moving beyond just targeting the tumor cell to targeting the ecosystem that allows it to thrive.” — Public Position Statement, American Society of Hematology.
Contraindications & When to Consult a Doctor
While this research points to future therapies, current patients must rely on established treatment protocols. There are no immediate contraindications for patients based on this study alone, as stromal-targeting drugs are not yet commercially available. However, patients should consult a hematologist-oncologist immediately if they experience persistent swollen lymph nodes, unexplained fever, night sweats, or weight loss. These “B symptoms” indicate active disease requiring intervention.
Patients should not attempt to modulate their immune system through supplements based on this news. Interferon pathways are complex; unsupervised manipulation could theoretically exacerbate inflammation. Always verify treatment plans through peer-reviewed clinical guidelines rather than preliminary research reports.