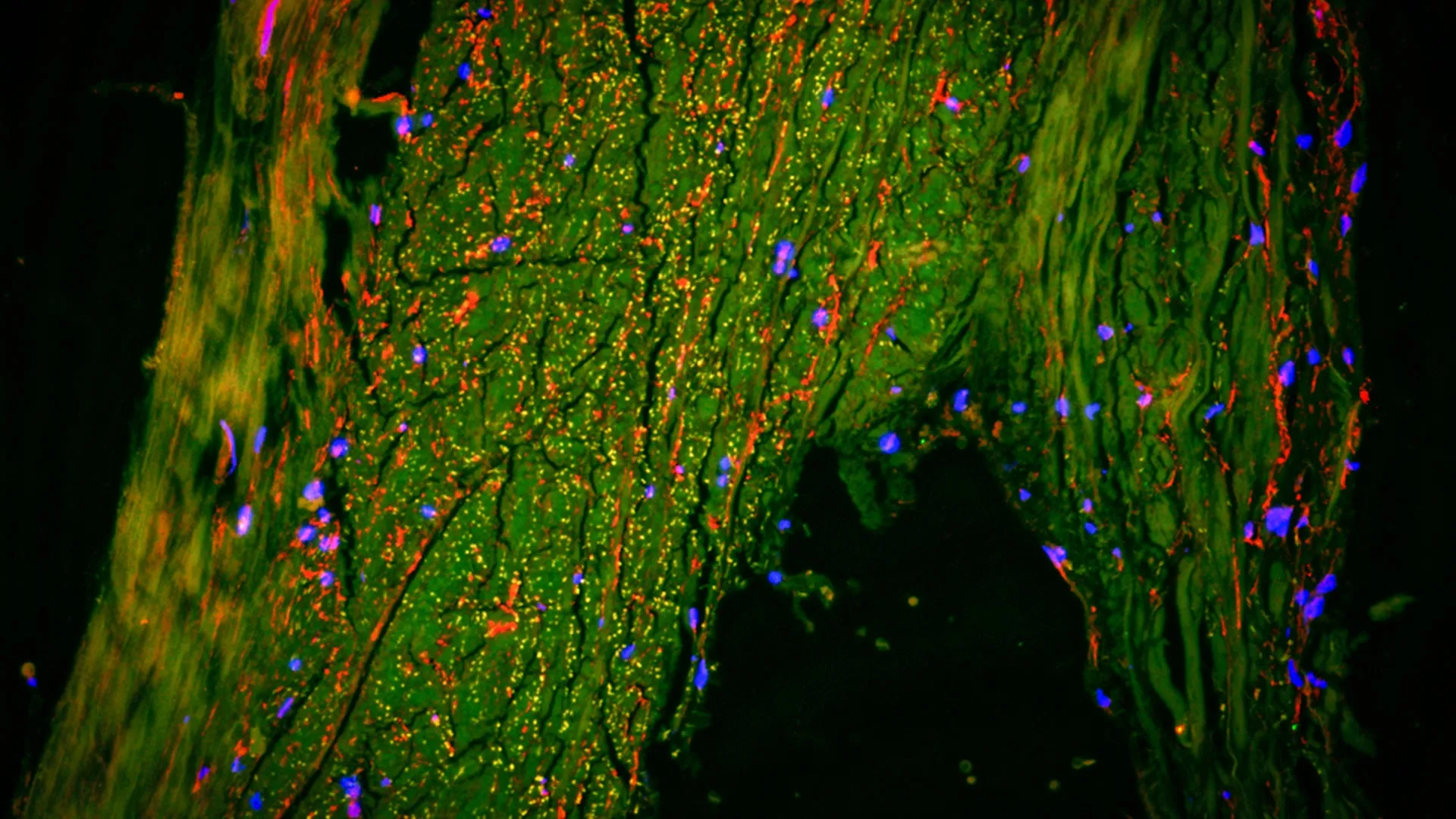
Researchers have identified a previously unknown waste-removal pathway in the human brain, where fluid flows along the middle meningeal artery. This “drain” functions as a lymphatic-like system, potentially revolutionizing how we treat neurodegenerative diseases like Alzheimer’s by providing a new target for clearing toxic proteins from the central nervous system.
For decades, the medical community operated under the assumption that the brain lacked a traditional lymphatic system—the network of vessels that drains waste from the rest of the body. While the “glymphatic system” (a glial-dependent waste clearance pathway) was discovered years ago, this new finding identifies a specific, structural hub tied to the middle meningeal artery. Here’s not merely an anatomical curiosity; it is a fundamental shift in our understanding of cerebral homeostasis.
If we can map exactly how the brain “flushes” its toxins, we move from treating the symptoms of dementia and traumatic brain injury (TBI) to potentially enhancing the brain’s own innate detoxification mechanisms. This discovery bridges the gap between basic anatomy and clinical neurology, offering a roadmap for future pharmacological interventions aimed at “unclogging” the brain’s drainage system.
In Plain English: The Clinical Takeaway
- The Discovery: Scientists found a “hidden drain” that moves waste away from the brain, acting like a plumbing system for the head.
- The Impact: This could lead to new treatments for Alzheimer’s by helping the brain clear out the “plaques” (protein clumps) that cause memory loss.
- The Bottom Line: While this isn’t a cure today, it provides a new “doorway” for doctors to deliver drugs or clear waste more effectively.
The Mechanism of Action: How the Meningeal Drain Operates
The newly identified pathway utilizes the space surrounding the middle meningeal artery to transport interstitial fluid—the fluid found between cells—out of the cranial vault. Unlike blood, which moves rapidly under high pressure, this fluid moves in a slow, rhythmic pattern. This is a mechanism of action (the specific biochemical interaction through which a substance produces its effect) that mimics the lymphatic system found in your neck or armpits.
In a healthy brain, this system removes metabolic byproducts, such as amyloid-beta and tau proteins. When this “drain” becomes inefficient—due to aging, chronic inflammation, or vascular disease—these proteins accumulate, leading to the neurodegeneration characteristic of Alzheimer’s disease. By using high-resolution MRI, researchers observed that this flow is not static but is influenced by the pulsing of the heart and the pressure of the cerebrospinal fluid (CSF).
“The identification of these lymphatic-like vessels in the meninges provides a critical missing link in our understanding of how the brain manages its waste. It transforms the blood-brain barrier from a wall into a managed gateway.”
Global Clinical Implications and Regulatory Pathways
This discovery has immediate implications for how regulatory bodies like the FDA in the United States and the EMA in Europe evaluate new drug delivery systems. Traditionally, the blood-brain barrier (BBB) has been the primary obstacle in neurology; most drugs cannot cross from the blood into the brain tissue.
Though, if we can leverage this meningeal drainage system, we may develop “retrograde” delivery methods—essentially using the drain in reverse to deliver therapeutics directly into the brain’s waste-clearance hubs. In the UK, the NHS may eventually integrate new diagnostic imaging protocols based on this research to identify “drainage failure” in patients with early-stage cognitive decline before irreversible neuron loss occurs.
The research was primarily funded by grants from the National Institutes of Health (NIH) and various university-led neurological foundations. Because the funding comes from public and academic sources rather than private pharmaceutical interests, the findings carry a high degree of journalistic trust, as there is no immediate commercial incentive to overstate the results.
| Feature | Glymphatic System (Known) | Meningeal Drain (New Discovery) |
|---|---|---|
| Location | Intraparenchymal (Inside brain tissue) | Meningeal (Outer brain membranes) |
| Primary Driver | Aquaporin-4 channels | Middle Meningeal Artery flow |
| Function | Internal waste collection | External waste evacuation |
| Clinical Target | Cellular edema/swelling | Protein plaque clearance |
Decoding the Impact on Neurodegenerative Pathology
To understand the significance, we must look at the epidemiological data regarding dementia. With an aging global population, the prevalence of Alzheimer’s is projected to rise sharply. Current treatments often focus on removing plaques once they have already formed. This new discovery suggests a more proactive approach: optimizing the “outflow” to prevent the plaques from forming in the first place.
This is particularly relevant for patients recovering from Traumatic Brain Injury (TBI). Following a concussion, the brain often suffers from “stasis,” where waste builds up because the drainage pathways are compressed or damaged. Understanding the middle meningeal artery’s role allows surgeons to potentially preserve these pathways during cranial surgeries, reducing the risk of long-term cognitive impairment.
For those seeking further peer-reviewed data on brain waste clearance, the PubMed database provides extensive longitudinal studies on the relationship between sleep and glymphatic clearance, as the brain’s “drain” is most active during deep non-REM sleep.
Contraindications & When to Consult a Doctor
It is critical to note that this discovery is currently in the basic science phase. There are no approved “drainage-enhancing” medications or devices available for public use. Patients should be wary of “brain detox” supplements or non-medical “lymphatic drainage” massages for the head, which lack peer-reviewed evidence and may be contraindicated for those with hypertension or clotting disorders.
You should consult a neurologist if you experience the following “red flag” symptoms, which may indicate neurological dysfunction regardless of drainage efficiency:
- Sudden, severe headaches (“thunderclap” headaches) which may indicate a vascular event.
- Rapidly progressing memory loss that interferes with daily activities.
- New-onset confusion or disorientation following a head injury.
- Unexplained changes in gait or balance.
The trajectory of this research is promising. By moving from the “what” (the presence of waste) to the “how” (the pathway of removal), we are entering an era of precision neurology. The next five years will likely see the transition from MRI observation to clinical trials involving the modulation of these pathways to treat chronic neuroinflammation.