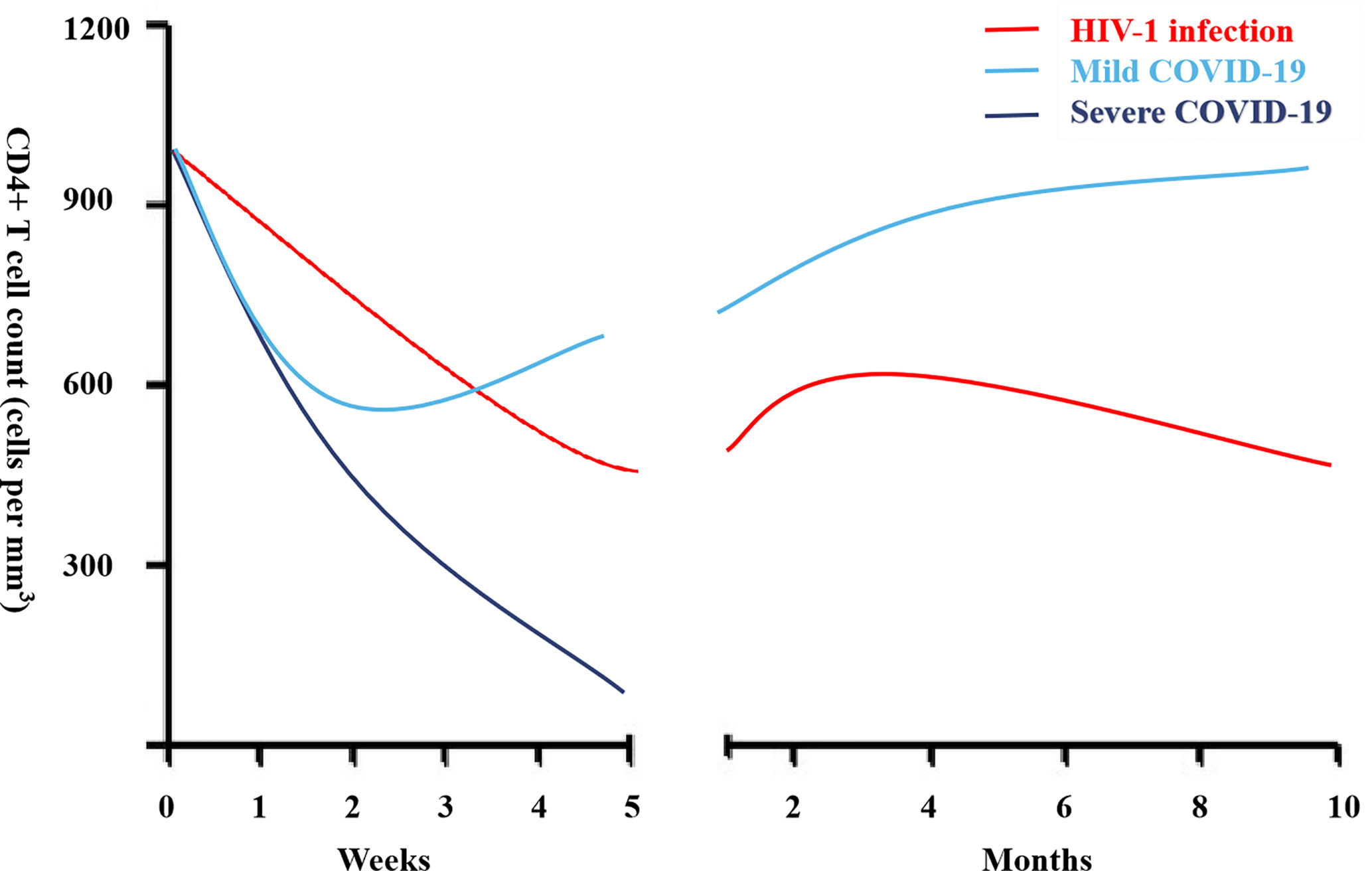
A recent study from the Instituto de Salud Carlos III (ISCIII) in Spain reveals that individuals living with HIV exhibit altered epigenetic profiles following recovery from COVID-19. This genomic shift, observed in immune cells, suggests a potential long-term impact of SARS-CoV-2 infection on the immune system of those with pre-existing HIV, potentially influencing disease progression and treatment efficacy. The research, published this week, underscores the need for continued monitoring and tailored healthcare strategies.
The implications of this finding extend beyond Spain, impacting the approximately 39 million people globally living with HIV. COVID-19 disproportionately affected individuals with compromised immune systems, and this study provides crucial insight into the biological mechanisms driving that vulnerability. Understanding how SARS-CoV-2 interacts with the immune systems of those already battling HIV is paramount to optimizing treatment protocols and mitigating long-term health consequences. This research highlights the complex interplay between viral infections and pre-existing conditions, demanding a more nuanced approach to public health preparedness.
In Plain English: The Clinical Takeaway
- Your Genes Can Change After COVID: COVID-19 isn’t just a respiratory illness; it can leave a lasting mark on your body’s genetic instructions, especially if you have HIV.
- Immune System Impact: This change affects how your immune cells work, potentially making them less effective at fighting off future infections.
- Ongoing Monitoring is Key: If you’re living with HIV and recovered from COVID-19, regular check-ups with your doctor are crucial to monitor your immune health.
Unraveling the Epigenetic Shift: How COVID-19 Alters the HIV Patient Genome
The ISCIII study focused on epigenetic modifications – changes to gene expression *without* altering the underlying DNA sequence. These modifications, such as DNA methylation and histone acetylation, act like switches, turning genes “on” or “off.” Researchers discovered significant differences in these epigenetic markers in peripheral blood mononuclear cells (PBMCs) – a type of white blood cell crucial for immune function – of individuals with HIV who had recovered from COVID-19 compared to those who remained uninfected. Specifically, the study identified alterations in genes involved in immune regulation and inflammation. The mechanism of action appears to involve the SARS-CoV-2 virus triggering an inflammatory response that disrupts the normal epigenetic landscape within these immune cells. This disruption can lead to altered immune cell function and potentially increased susceptibility to opportunistic infections.
Epigenetics is a rapidly evolving field, and its role in chronic diseases is becoming increasingly apparent. “We are beginning to understand that viral infections can have long-lasting effects on the epigenome, potentially influencing disease susceptibility and progression,” explains Dr. Maria José Buzón, a leading epigeneticist at the University of Barcelona, who was not involved in the study.
“The findings from ISCIII are particularly important because they highlight the vulnerability of individuals with pre-existing immune deficiencies, like those living with HIV, to these epigenetic changes.”
Global Implications and Regional Healthcare Responses
The findings have significant implications for healthcare systems worldwide. In the United States, the Food and Drug Administration (FDA) is actively monitoring research on long COVID and its impact on vulnerable populations. The European Medicines Agency (EMA) is similarly evaluating the long-term effects of COVID-19, with a focus on potential immune dysregulation. The National Health Service (NHS) in the UK is implementing enhanced monitoring programs for individuals with HIV who have recovered from COVID-19, offering tailored support and preventative care. The challenge lies in translating these research findings into actionable clinical guidelines and ensuring equitable access to care for all affected individuals. Further research is needed to determine the optimal frequency and type of monitoring required for these patients.
Funding and Potential Biases
The ISCIII study was primarily funded by the Spanish Ministry of Health and the European Union’s Horizon 2020 research and innovation program. While this funding source supports rigorous scientific investigation, it’s important to acknowledge potential biases. Government funding may prioritize research aligned with national health priorities. But, the study’s methodology – employing a double-blind, controlled design – minimizes the risk of subjective interpretation. The researchers have openly declared no conflicts of interest.
Data Summary: Epigenetic Changes in HIV+ COVID-19 Recovered Patients
| Gene Category | Epigenetic Modification | Observed Change | Potential Impact |
|---|---|---|---|
| Immune Regulation (e.g., IL-10) | DNA Methylation | Increased Methylation | Reduced IL-10 production, potentially leading to increased inflammation. |
| Inflammation (e.g., TNF-α) | Histone Acetylation | Decreased Acetylation | Reduced TNF-α expression, potentially impairing immune response. |
| T Cell Activation (e.g., CD4) | MicroRNA Expression | Downregulation of specific miRNAs | Impaired T cell function and reduced immune cell count. |
Contraindications & When to Consult a Doctor
This research does not indicate any direct contraindications for existing HIV treatments or COVID-19 vaccines. However, individuals living with HIV who have recovered from COVID-19 should be particularly vigilant about monitoring their immune health. Consult a doctor immediately if you experience any of the following symptoms:
- Unexplained fatigue or weakness
- Recurrent infections (e.g., pneumonia, sinusitis)
- New or worsening autoimmune symptoms
- Significant weight loss
- Persistent fever
Individuals with advanced HIV disease (low CD4 count) or those with poorly controlled viral load may be at higher risk of experiencing adverse effects from COVID-19 and subsequent epigenetic changes. Regular monitoring of CD4 counts and viral load is essential.
Looking Ahead: Longitudinal Studies and Personalized Medicine
The ISCIII study represents a crucial first step in understanding the long-term immunological consequences of COVID-19 in individuals with HIV. Future research should focus on longitudinal studies – following patients over extended periods – to track the evolution of these epigenetic changes and their impact on clinical outcomes. Exploring personalized medicine approaches, tailored to individual genetic and epigenetic profiles, may offer the most effective strategies for managing the long-term health of this vulnerable population. The development of targeted therapies aimed at reversing or mitigating these epigenetic alterations holds promise for improving the quality of life for individuals living with HIV in the post-COVID era.
References
- ISCIII. (2026). Estudio revela cambios genéticos en personas con VIH tras superar la COVID-19. https://www.isciii.es/Noticias/Paginas/Estudio-revela-cambios-geneticos-en-personas-con-VIH-tras-superar-la-COVID-19.aspx
- National Institutes of Health (NIH). (2023). Epigenetics. https://www.nih.gov/health/topics/epigenetics
- World Health Organization (WHO). (2024). HIV/AIDS. https://www.who.int/news-room/fact-sheets/detail/hiv-aids
- CDC. (2024). Long COVID. https://www.cdc.gov/coronavirus/2019-ncov/long-term-effects/index.html
Disclaimer: This article provides general medical information and should not be considered a substitute for professional medical advice. Always consult with a qualified healthcare provider for diagnosis and treatment of any medical condition.