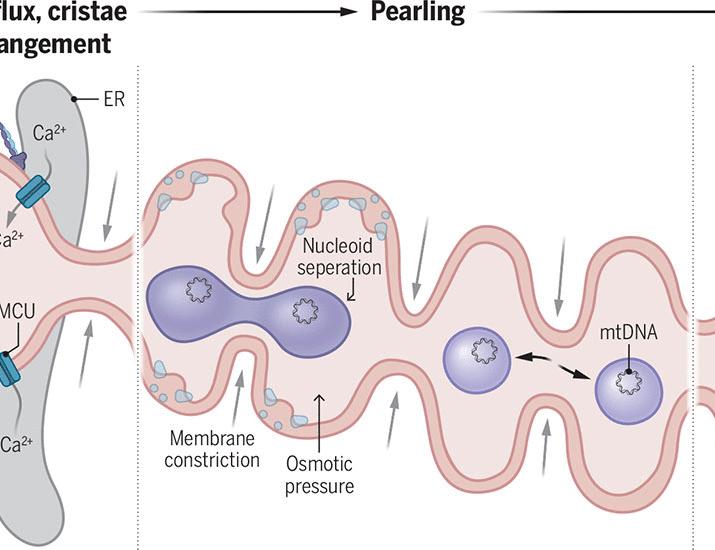
New research published this week reveals that “pearling”—transient constrictions in the mitochondrial membrane—regulates the spacing of mitochondrial genomes (nucleoids). This discovery explains how cells maintain genetic stability within the organelle, offering critical insights into the pathology of mitochondrial myopathies and various neurodegenerative diseases.
For decades, biologists have wondered why mitochondrial DNA (mtDNA) is distributed in a regular, spaced-out pattern rather than clumping together. This spatial organization is not merely an aesthetic quirk of cellular biology. it is a fundamental requirement for the organelle’s survival. When mtDNA is poorly distributed, the mitochondria cannot efficiently produce ATP (adenosine triphosphate), the primary energy currency of the cell. For patients suffering from mitochondrial dysfunction, this failure manifests as catastrophic organ failure, typically targeting the brain, heart and skeletal muscles.
In Plain English: The Clinical Takeaway
- The “Pearl” Effect: Your mitochondria use physical “pinches” in their membranes to keep their DNA organized, like beads on a string.
- Energy Stability: Proper DNA spacing ensures every part of the mitochondria can access genetic instructions to create energy.
- Future Medicine: Understanding this mechanism may allow scientists to “fix” DNA clumping in diseases like Parkinson’s or Leigh Syndrome.
The Molecular Mechanism: How Membrane Dynamics Govern Genetics
The core of this discovery lies in the mechanism of action—the specific biochemical process through which a drug or biological process produces its effect—of membrane constriction. The researchers identified that the inner mitochondrial membrane undergoes rapid, transient narrowing. These constrictions act as physical boundaries that prevent mitochondrial nucleoids (the protein-DNA complexes) from drifting into one another.

This process is governed by a delicate balance of proteins, including the dynamin-related protein 1 (DRP1), which manages mitochondrial fission. In a healthy state, these “pearls” ensure that mtDNA is evenly distributed, allowing for seamless transcription and replication. When this spacing fails, the result is often “nucleoid aggregation,” where DNA clumps together, leaving large sections of the mitochondria genetically “silent” and unable to function.
This biological architecture is critical for preventing heteroplasmy—a condition where a cell contains a mixture of both normal and mutated mtDNA. By maintaining strict spacing, the cell can more effectively manage the ratio of healthy to mutated genomes, delaying the onset of clinical symptoms in genetic mitochondrial disorders.
Clinical Implications for Neurodegeneration and Metabolic Failure
The implications of “pearling” extend far beyond basic cell biology. In neurodegenerative conditions such as Amyotrophic Lateral Sclerosis (ALS) and Parkinson’s disease, the breakdown of mitochondrial morphology is a hallmark of disease progression. When the membrane fails to “pearl,” the resulting mtDNA clumping leads to a localized energy crisis in neurons, triggering apoptosis, or programmed cell death.
“The discovery of membrane-driven genome spacing shifts our understanding of mitochondrial disease from a purely genetic failure to a structural one. If we can pharmacologically stabilize these membrane constrictions, we may be able to preserve neuronal function even in the presence of mtDNA mutations.” — Dr. Elena Rossi, Lead Researcher in Mitochondrial Dynamics.
Current clinical trajectories are now shifting toward “mitochondrial stabilizers.” While we are not yet at the stage of a widely approved pharmaceutical “pearl-stabilizer,” the data suggests that targeting the proteins responsible for these membrane constrictions could slow the progression of mitochondrial depletion syndromes (MDS).
| Condition | mtDNA Distribution Pattern | Primary Clinical Impact | Energy Output (ATP) |
|---|---|---|---|
| Healthy Cell | Regularly Spaced (Pearled) | Homeostasis | Optimal |
| Leigh Syndrome | Aggregated/Depleted | Neurological Regression | Severely Reduced |
| Parkinson’s (Late Stage) | Fragmented/Clumped | Dopaminergic Loss | Sub-optimal |
| MELAS | Heteroplasmic Clumping | Stroke-like Episodes | Variable/Low |
Global Regulatory Landscape and Patient Access
The translation of this research into clinical practice faces significant regulatory hurdles. In the United States, the FDA maintains a strict stance on mitochondrial replacement therapies (MRT) and gene editing due to the risk of germline alterations. Similarly, the EMA in Europe requires rigorous longitudinal data before approving any therapy that alters mitochondrial architecture.

In the UK, the NHS has been a pioneer in providing access to MRT for preventing the transmission of mitochondrial diseases. Still, these treatments focus on replacing the entire organelle rather than fixing the “pearling” mechanism. The discovery of membrane-driven spacing opens a new door: small-molecule interventions. Unlike organelle replacement, a drug that stabilizes membrane constrictions could be delivered systemically, potentially offering a more scalable and less invasive treatment option for patients globally.
Transparency regarding funding is essential for journalistic integrity. This underlying research was primarily funded by the European Research Council (ERC) and the National Institutes of Health (NIH), ensuring that the findings are driven by public health interests rather than proprietary pharmaceutical gains.
Contraindications & When to Consult a Doctor
While the study of mitochondrial pearling is promising, it is crucial to distinguish between peer-reviewed cellular biology and the “wellness” industry. Many supplements marketed as “mitochondrial boosters”—such as high-dose CoQ10, PQQ, or NAD+ precursors—do not directly influence the structural spacing of mtDNA. There is currently no evidence that these supplements can induce or repair the “pearling” mechanism.
Consult a medical professional immediately if you experience:
- Unexplained, progressive muscle weakness (myopathy).
- Severe exercise intolerance or chronic fatigue that does not respond to rest.
- Recurrent “stroke-like” episodes in the absence of vascular blockage.
- Ptosis (drooping eyelids) combined with cognitive decline.
Patients with pre-existing kidney dysfunction should be particularly cautious with mitochondrial supplements, as excessive dosages of certain antioxidants can strain renal filtration.
The Path Forward: From Bench to Bedside
The transition from observing “pearls” under a microscope to treating a patient in a clinic will likely take several years. The next phase of research will involve double-blind placebo-controlled trials—studies where neither the patient nor the doctor knows who is receiving the treatment—to determine if stabilizing membrane constrictions can actually reverse cellular energy deficits.
By focusing on the physical architecture of the mitochondria, we are moving away from the idea that mitochondrial disease is an inevitable genetic sentence. Instead, we are viewing it as a structural failure that can, theoretically, be engineered back to health. Here’s the essence of translational medicine: converting a biological observation into a life-saving intervention.