A German federal institute has confirmed the death of an infant linked to a vitamin supplement. The tragedy highlights critical gaps in supplement regulation and the dangers of fat-soluble vitamin toxicity in neonates, prompting urgent warnings regarding pediatric dosage and product purity across the European Union.
This fatality is not an isolated incident of misfortune but a systemic failure in how we categorize “nutraceuticals”—products that sit in a regulatory gray zone between food and medicine. For parents, the distinction is academic; for a neonate, whose hepatic (liver) and renal (kidney) systems are still maturing, the difference between a therapeutic dose and a lethal one is razor-thin. This case underscores a global public health crisis: the proliferation of unregulated supplements that bypass the rigorous pre-market clinical trials required for pharmaceuticals.
In Plain English: The Clinical Takeaway
- Not all vitamins are safe: While water-soluble vitamins (like C) are flushed out through urine, fat-soluble vitamins (A, D, E, K) store in the body and can reach toxic levels.
- Infant vulnerability: Babies cannot process high doses of supplements; their livers cannot detoxify these substances as efficiently as adults.
- Medical supervision is mandatory: Never administer any supplement to an infant—even “natural” ones—without a prescription or direct guidance from a pediatrician.
The Pathophysiology of Fat-Soluble Toxicity
To understand how a vitamin can become a toxin, we must examine the mechanism of action—the specific biochemical process through which a substance produces its effect in the body. Most lethal vitamin overdoses in infants involve fat-soluble vitamins, particularly Vitamin A (Retinol) or Vitamin D (Calciferol).
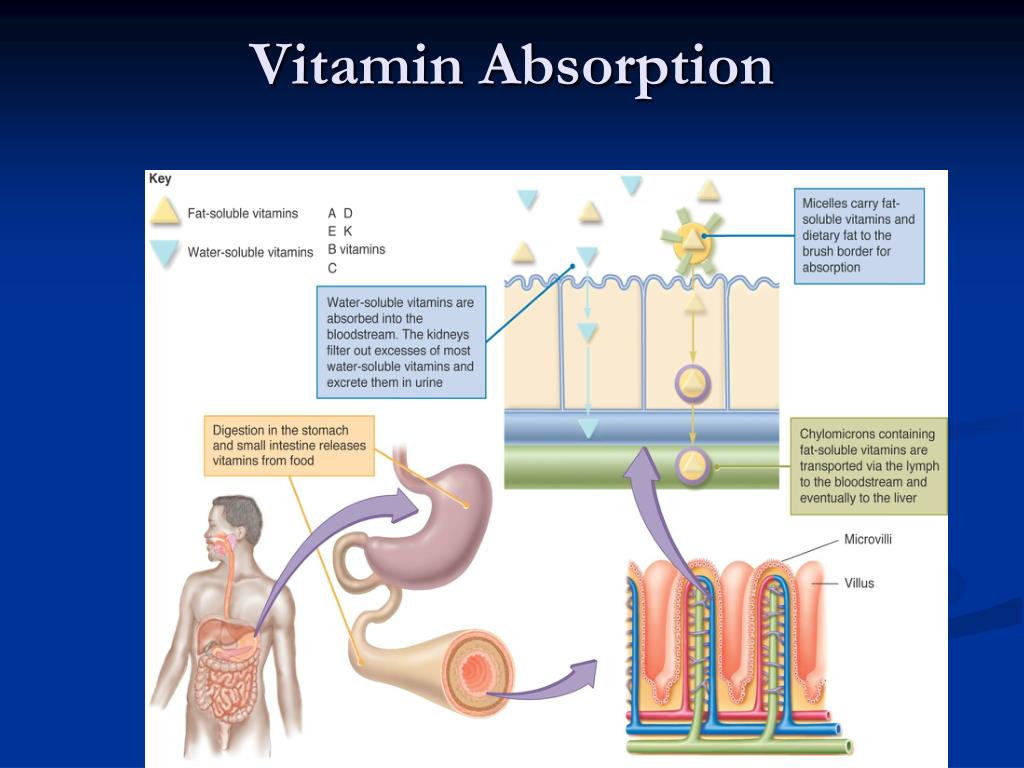
Unlike water-soluble vitamins, which the body excretes easily, fat-soluble vitamins are sequestered in the liver and adipose (fat) tissue. In a neonate, an overdose leads to hypervitaminosis, a state of vitamin toxicity. When Vitamin A levels spike, it can lead to increased intracranial pressure, causing the fontanelle (the soft spot on a baby’s head) to bulge and potentially causing permanent neurological damage or respiratory failure.
The toxicity is often exacerbated by bioavailability—the proportion of the nutrient that enters the circulation and is able to have an active effect. In contaminated or poorly formulated supplements, the concentration of the active ingredient may be significantly higher than what is listed on the label, leading to accidental acute poisoning.
“The tragedy of pediatric supplement toxicity often lies in the ‘dosage gap.’ Because supplements are not held to the same Solid Manufacturing Practices (GMP) as pharmaceuticals, a single drop can contain ten times the intended dose, which is catastrophic for a three-kilogram infant.” — Dr. Elena Rossi, Senior Toxicologist and Pediatric Epidemiologist.
Regulatory Divergence: BfArM, EMA, and the FDA
The confirmation of this death by the Bundesinstitut für Arzneimittel und Medizinprodukte (BfArM) in Germany highlights a fragmented global regulatory landscape. In Europe, the European Medicines Agency (EMA) provides overarching guidelines, but the enforcement of “food supplement” safety often falls to national bodies.
In the United States, the FDA operates under the Dietary Supplement Health and Education Act (DSHEA), which essentially treats supplements as food. This means the FDA does not approve supplements for safety or efficacy before they hit the market; they only intervene after a product is proven unsafe. This “post-market surveillance” model is inherently reactive, meaning the “signal” of a dangerous product is often only detected after patient harm has occurred.
The German case demonstrates that even in regions with stricter oversight, the boundary between a “food supplement” and a “medicinal product” remains porous. When a product is marketed as a supplement, it avoids the double-blind placebo-controlled trials—the gold standard of research where neither the patient nor the doctor knows who is receiving the treatment—that would normally uncover lethal side effects in vulnerable populations.
| Vitamin Type | Storage Site | Excretion Method | Toxicity Risk (Infants) |
|---|---|---|---|
| Water-Soluble (B, C) | Minimal/Blood | Renal (Urine) | Low (Generally safe) |
| Fat-Soluble (A, D, E, K) | Liver/Adipose | Biliary/Fecal | High (Cumulative toxicity) |
Funding, Bias, and the Nutraceutical Industry
A critical component of this public health failure is the funding of nutritional research. A vast majority of studies touting the benefits of pediatric supplements are funded by the manufacturers themselves. This creates a significant publication bias, where positive results are highlighted and adverse events are minimized or omitted from the final report.
The industry lobbies heavily to preserve supplements categorized as food, as this removes the requirement for expensive Phase I, II, and III clinical trials. By avoiding these phases, companies can bring products to market in months rather than years, but they do so without a comprehensive understanding of the contraindications—specific conditions or factors that make a treatment inadvisable for a particular patient.
Contraindications & When to Consult a Doctor
Parents must be vigilant. Vitamin supplements are contraindicated for infants who are receiving fortified formula or are exclusively breastfed unless a clinical deficiency has been diagnosed via blood test. You should seek immediate emergency medical intervention if an infant shows the following signs after supplement administration:
- Extreme Lethargy: Unusual sleepiness or inability to wake for feedings.
- Hypercalcemia Symptoms: Excessive thirst, vomiting, or constipation (common in Vitamin D toxicity).
- Neurological Distress: A bulging soft spot (fontanelle) on the skull or unexplained irritability.
- Respiratory Depression: Shallow or irregular breathing patterns.
The Path Forward: Precision Pediatric Nutrition
This week’s regulatory confirmation serves as a grim reminder that “natural” does not mean “safe.” The medical community must push for a reclassification of high-dose pediatric supplements, moving them from the “food” category to the “pharmaceutical” category. This would mandate strict dosage verification and rigorous safety testing in neonatal cohorts.
Until legislative changes occur, the burden of safety falls on the prescribing physician and the vigilant parent. The goal must be precision nutrition—providing exactly what the infant needs based on clinical data, rather than relying on the marketing claims of an unregulated industry.

