INOVIO Pharmaceuticals, Inc. (Nasdaq: INO) announced plans this week for a public offering of its common stock, seeking to raise capital for the continued development and potential commercialization of its DNA medicines. This move comes as the biotechnology sector faces increased scrutiny regarding funding and clinical trial progress, particularly in the realm of infectious disease and oncology. The offering’s success will be closely watched by investors and the medical community alike.
The announcement by INOVIO isn’t simply a financial maneuver; it represents a critical juncture in the advancement of DNA medicine – a field promising targeted therapies with potentially fewer side effects than traditional approaches. While conventional vaccines and treatments often rely on introducing weakened or inactive pathogens, or on systemic chemotherapy, DNA medicines deliver genetic instructions directly to the body’s cells, prompting them to produce therapeutic proteins. This precision could revolutionize treatment for a range of conditions, but significant hurdles remain in demonstrating long-term efficacy and ensuring widespread accessibility.
In Plain English: The Clinical Takeaway
- DNA Medicines: Instead of giving you a drug, these therapies give your cells instructions to make their own medicine.
- Public Offering: INOVIO is selling more shares of its company to raise money for research and development. This doesn’t directly affect patients *yet*, but it could speed up the availability of new treatments.
- Clinical Trials are Key: The success of these therapies depends on proving they function safely and effectively in large groups of people.
Understanding INOVIO’s Technology: The Plasmid DNA Approach
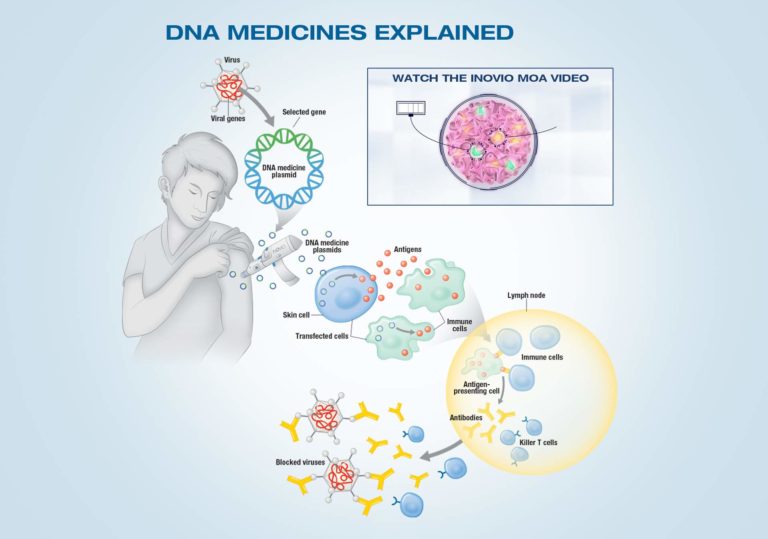
INOVIO’s core technology centers around its proprietary plasmid DNA delivery system. Plasmids are compact, circular DNA molecules that can carry genetic information. INOVIO uses these plasmids to deliver genes encoding for specific antigens – molecules that trigger an immune response – or therapeutic proteins. This is distinct from mRNA vaccines, like those developed by Pfizer and Moderna, which deliver messenger RNA instead of DNA. The key difference lies in how the genetic material interacts with the cell’s nucleus. DNA must enter the nucleus to be transcribed into RNA, while mRNA can be translated directly into protein in the cytoplasm. INOVIO utilizes a process called electroporation, employing brief electrical pulses to temporarily open pores in cell membranes, facilitating plasmid DNA entry. This mechanism of action is crucial for effective gene delivery and subsequent protein expression.

Clinical Trial Landscape and Regulatory Pathways
Currently, INOVIO is focusing its development efforts on several key areas, including infectious diseases like COVID-19 and cancers such as cervical dysplasia and soft tissue sarcoma. Their COVID-19 vaccine candidate, INO-4800, has undergone Phase 2 clinical trials, demonstrating an immune response, but faced challenges in achieving the same level of efficacy as mRNA vaccines. A Phase 3 trial was initiated but faced delays and ultimately did not meet primary endpoints as quickly as anticipated. The company is also exploring combinations with adjuvants – substances that enhance the immune response – to improve vaccine efficacy. The regulatory pathway for DNA medicines is complex. In the United States, the Food and Drug Administration (FDA) reviews these therapies under existing frameworks for biologics and vaccines, but may require additional data to assess long-term safety and efficacy. Similarly, in Europe, the European Medicines Agency (EMA) evaluates these products through a similar rigorous process.
Data Summary: INO-4800 Phase 2 Trial (Select Data)
| Parameter | Value |
|---|---|
| Total Participants (N) | 400 |
| Age Range | 18-85 years |
| Neutralizing Antibody Response (Geometric Mean Fold Rise) | 2.8x (after 2 doses) |
| T Cell Response (IFN-γ ELISpot) | Significant increase observed |
| Adverse Events (Grade 3 or higher) | <5% |
Geographical Impact and Access to Innovation
The potential impact of INOVIO’s technology extends beyond the United States and Europe. DNA medicines offer a potentially scalable and cost-effective solution for addressing infectious disease outbreaks in low- and middle-income countries. Still, challenges related to cold chain storage and distribution – maintaining the vaccines at specific temperatures – remain significant. The World Health Organization (WHO) is actively working to address these logistical hurdles and ensure equitable access to vaccines globally. The development of thermostable DNA vaccines – those that can withstand higher temperatures – is a critical area of research.
“The development of DNA vaccines represents a significant advancement in vaccine technology, offering the potential for rapid development and scalable manufacturing. However, ensuring equitable access and addressing logistical challenges in resource-limited settings are paramount.” – Dr. Kate O’Brien, Director of the WHO’s Department of Immunization, Vaccines and Biologicals (as stated in a 2024 WHO briefing).
Funding Transparency and Potential Biases
It’s crucial to acknowledge the funding sources behind INOVIO’s research. The company has received funding from the U.S. Department of Defense’s Defense Threat Reduction Agency (DTRA) for its vaccine development programs. While this funding supports important research, it also raises questions about potential biases in research priorities. Investors should be aware that the company’s stock price is subject to market volatility and speculation. A comprehensive understanding of the company’s financial situation and research pipeline is essential for informed investment decisions.
Contraindications & When to Consult a Doctor
While DNA medicines hold promise, they are not without potential risks. Individuals with a history of severe allergic reactions to vaccine components should avoid these therapies. Individuals with certain autoimmune disorders may experience exacerbation of their condition. Common side effects observed in clinical trials include injection site pain, muscle aches, and fatigue. If you experience any severe or persistent symptoms following vaccination or treatment with a DNA medicine, consult a doctor immediately. Specifically, seek medical attention if you develop difficulty breathing, swelling of the face or throat, or a high fever. Pregnant or breastfeeding individuals should discuss the potential risks and benefits with their healthcare provider before receiving these therapies.
Looking ahead, the success of INOVIO’s public offering and the continued progress of its clinical trials will be pivotal in determining the future of DNA medicine. While challenges remain, the potential benefits – targeted therapies, rapid development, and scalable manufacturing – are significant. Continued research, rigorous clinical evaluation, and transparent communication will be essential to unlock the full potential of this innovative technology and ensure its equitable access for all.
References
- “DNA Vaccines: A Review” – PubMed
- WHO Q&A on DNA Vaccines – World Health Organization
- Vaccine Development 101 – U.S. Food and Drug Administration
- Vaccine Development – European Medicines Agency
- “Electroporation-mediated DNA delivery: principles, applications and future perspectives” – Nature Reviews Genetics