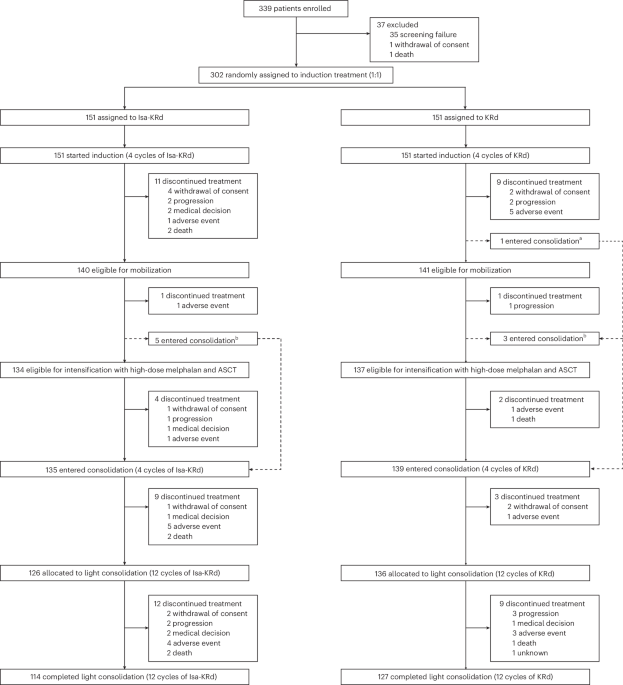
Published this week in Nature Medicine, the phase 3 EMN24 IsKia trial demonstrates that adding isatuximab to a regimen of carfilzomib, lenalidomide, and dexamethasone significantly increases rates of measurable residual disease (MRD) negativity in transplant-eligible patients with newly diagnosed multiple myeloma, potentially improving long-term outcomes through deeper clinical responses.
For patients facing a diagnosis of multiple myeloma—a malignancy of plasma cells in the bone marrow—the goal of initial treatment is not merely to reduce the tumor burden, but to achieve a “deep response.” This means eliminating as many cancerous cells as possible before a stem cell transplant. The EMN24 IsKia trial represents a pivotal shift in the “quadruplet” therapy approach, suggesting that the synergy of four distinct drug mechanisms can push more patients toward a state where no detectable cancer remains, even with the most sensitive testing methods.
In Plain English: The Clinical Takeaway
- Deeper Cleaning: Adding Isatuximab to the standard three-drug cocktail helps clear more cancer cells from the body before and after a transplant.
- MRD Negativity: More patients achieved “MRD negativity,” which is the gold standard for measuring how well a treatment is working.
- Better Foundation: This approach provides a stronger “baseline” of health, which may lead to longer periods of remission.
The Molecular Synergy: How the Quadruplet Attack Works
To understand why this combination is effective, we must examine the mechanism of action—the specific biological process by which a drug produces its effect. Multiple myeloma cells are characterized by the expression of CD38, a protein on their surface that acts like a beacon for the immune system.
Isatuximab is a monoclonal antibody that binds to CD38, effectively tagging the cancer cell for destruction. When combined with carfilzomib (a proteasome inhibitor that blocks the cell’s “waste disposal” system, causing it to choke on its own proteins), lenalidomide (an immunomodulatory drug that enhances the body’s own immune response), and dexamethasone (a steroid that induces cancer cell death), the result is a multi-pronged assault.
This combination targets the cancer cell from four different angles: surface tagging, internal protein buildup, immune system activation, and direct apoptosis (programmed cell death). By attacking the malignant plasma cells through these diverse pathways, the regimen prevents the cancer from developing resistance, a common hurdle in hematologic malignancies.
Analyzing the EMN24 IsKia Trial Data
The trial focused on measurable residual disease (MRD). In oncology, MRD negativity means that the cancer is so thoroughly suppressed that it cannot be detected even when examining one million bone marrow cells. This is a critical prognostic marker; patients who achieve MRD negativity generally have significantly longer progression-free survival (PFS).
| Metric | Standard Triplet (CRd) | Isatuximab Quadruplet (IsCRd) |
|---|---|---|
| Primary Endpoint | Baseline MRD Negativity | Significantly Higher MRD Negativity |
| Drug Composition | Carfilzomib, Lenalidomide, Dex | Isatuximab + Carfilzomib, Lenalidomide, Dex |
| Patient Population | Transplant-Eligible NDMM | Transplant-Eligible NDMM |
| Key Goal | Disease Control | Deep Molecular Response |
The study was funded by Sanofi, the developer of isatuximab. While industry funding is standard for Phase 3 trials, the use of a randomized, double-blind, placebo-controlled design—where neither the patient nor the doctor knows which treatment is being administered—minimizes bias and ensures the results are statistically robust.
Global Access and Regulatory Hurdles
While the data is promising, the transition from a clinical trial to a bedside prescription depends on regional regulatory bodies. In the United States, the FDA evaluates the risk-benefit ratio, focusing heavily on the toxicity profile of adding a fourth agent. In Europe, the EMA often emphasizes the cost-effectiveness and quality-of-life improvements for the broader population.
For patients under the NHS in the UK, the hurdle is often the National Institute for Health and Care Excellence (NICE), which determines if the incremental gain in MRD negativity justifies the increased cost of the quadruplet therapy. This “geo-epidemiological gap” means that while the science is global, the access is local, often creating disparities in how quickly newly diagnosed patients can access these cutting-edge regimens.
“The pursuit of MRD negativity is no longer just a research goal; it is becoming a clinical imperative. By intensifying the induction phase, we are essentially redefining the ‘floor’ of what a successful response looks like in multiple myeloma.”
The Biological Trade-off: Efficacy vs. Toxicity
Adding a fourth drug is not without risk. The “intensity” of the treatment can lead to increased contraindications—specific situations where a drug should not be used because it may be harmful. For instance, the combination of carfilzomib and isatuximab can increase the risk of infusion-related reactions and hematologic toxicity, such as neutropenia (a dangerous drop in white blood cells).
the long-term impact on the immune system must be monitored. By aggressively suppressing the marrow to kill cancer cells, we also risk compromising the patient’s ability to fight opportunistic infections. This necessitates a careful balance of supportive care, including prophylactic antibiotics and close monitoring of blood counts.
Contraindications & When to Consult a Doctor
This quadruplet regimen is specifically for transplant-eligible patients. It is not suitable for those with severe cardiac dysfunction or those who cannot tolerate high-dose corticosteroids. You should consult your hematologist immediately if you experience:
- Severe Febrile Neutropenia: A high fever accompanied by a low white blood cell count, which is a medical emergency.
- Peripheral Neuropathy: Tingling or numbness in the extremities, which can be a side effect of carfilzomib.
- Severe Allergic Reactions: Any swelling of the face or difficulty breathing during isatuximab infusion.
The Future of Myeloma Therapy
The EMN24 IsKia trial confirms that the trend toward “more is better” in initial induction—provided the patient can tolerate it—is yielding deeper responses. As we move toward 2027, the focus will likely shift toward “precision intensification,” where we use biomarkers to decide who needs a quadruplet and who can achieve the same results with a triplet, thereby reducing unnecessary toxicity.
References
- Nature Medicine – Original Trial Publication (doi:10.1038/s41591-026-04282-0)
- PubMed – Comparative studies on CD38 monoclonal antibodies in NDMM.
- The Lancet – Longitudinal data on MRD negativity and progression-free survival.
- World Health Organization (WHO) – Global standards for hematologic malignancy classification.
Disclaimer: This article is for informational purposes and does not constitute medical advice. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.