Region Kalmar län in Sweden is demonstrating a pathway toward eliminating hepatitis, particularly Hepatitis C, through a combination of advanced medical care, equitable access to treatment, and a harm reduction approach centered around syringe exchange programs. Dramatic reductions in cases – from 54 in 2015 to 11 in 2025 – position the region as a leader in achieving the World Health Organization’s (WHO) 2030 eradication goal.
The success in Kalmar län isn’t simply about pharmaceutical advancements; it’s a testament to a holistic public health strategy that prioritizes reaching vulnerable populations. This approach has significant implications for global hepatitis eradication efforts, particularly in regions facing similar challenges with access to care and harm reduction services. The region’s proactive expansion of these programs, coupled with increased screening, offers a replicable model for other healthcare systems worldwide.
In Plain English: The Clinical Takeaway
- Hepatitis C is curable: Modern medications can eliminate the virus in most people within 8-12 weeks.
- Harm reduction saves lives: Providing clean needles and healthcare access to people who inject drugs is crucial for preventing the spread of hepatitis C.
- Early detection is key: Screening programs, especially within vulnerable populations, are vital for identifying and treating infections before they cause serious liver damage.
The Biology of Hepatitis: Understanding the Viral Threat
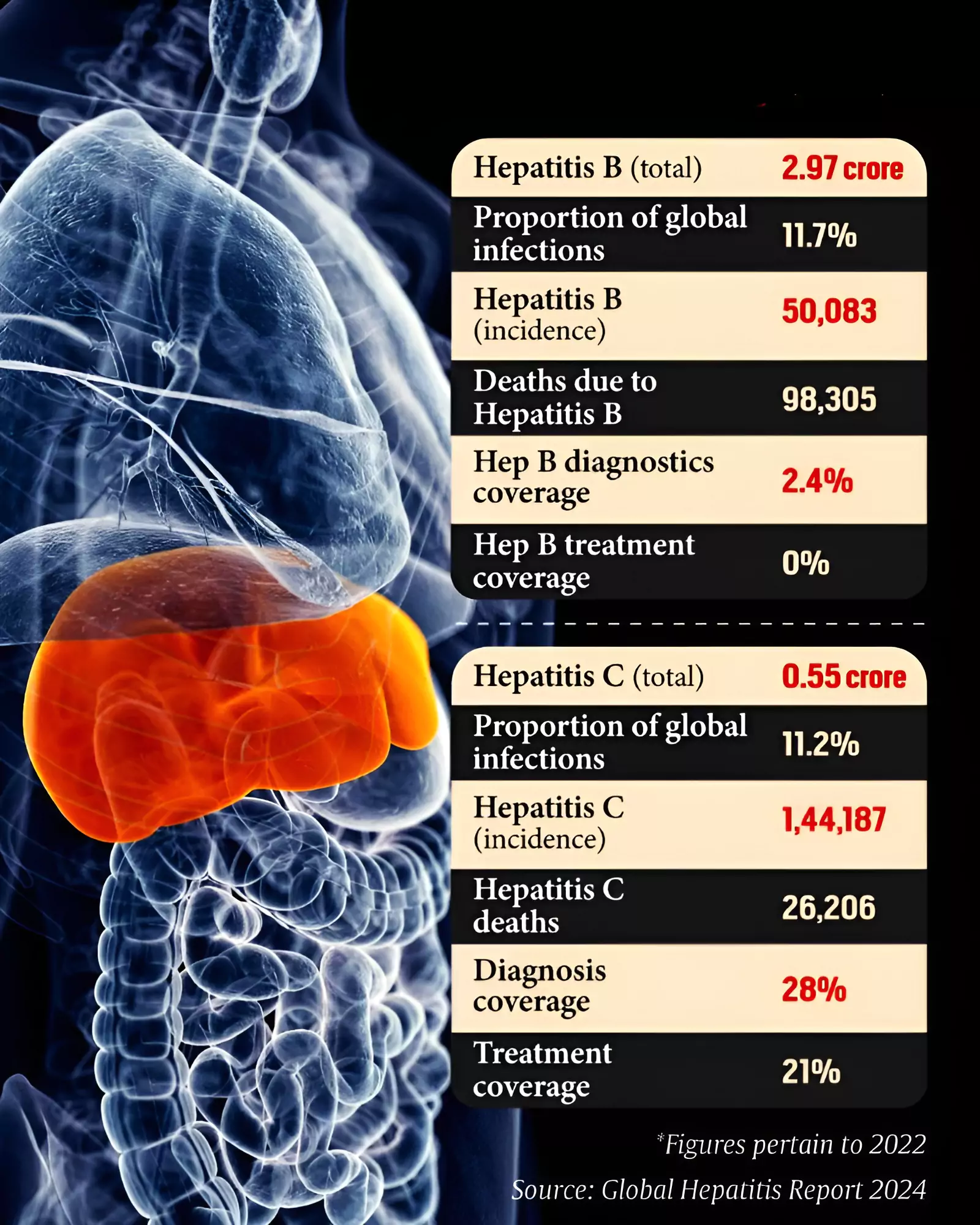
Hepatitis encompasses a group of inflammatory conditions affecting the liver, most commonly caused by viral infections. Hepatitis B (HBV) and Hepatitis C (HCV) are particularly concerning due to their potential for chronic infection, leading to cirrhosis, liver failure, and hepatocellular carcinoma. HCV, specifically, is an RNA virus belonging to the Flaviviridae family. Its mechanism of action involves hijacking the host cell’s machinery to replicate its genetic material, ultimately causing liver cell damage. The immune response to the virus, while attempting to clear the infection, also contributes to the inflammation and subsequent liver injury. Understanding the viral lifecycle is critical for developing targeted antiviral therapies. Direct-acting antivirals (DAAs) represent a major breakthrough, specifically targeting viral proteins essential for replication, such as NS3/4A protease, NS5A protein, and NS5B polymerase. [ 1 ]
Kalmar Län’s Strategy: A Multifaceted Approach
The dramatic decline in Hepatitis C cases in Kalmar län is directly attributable to a multi-pronged strategy. Central to this success is the region’s long-standing commitment to syringe exchange programs, initiated in 2012. These programs aren’t merely about providing sterile injection equipment; they serve as a crucial entry point to healthcare for individuals who may otherwise avoid traditional medical settings. Offering on-site screening for HCV and providing immediate access to DAAs dramatically increases treatment rates. The region is expanding screening initiatives within psychiatric facilities and opioid agonist therapy (LARO) clinics – recognizing these populations are at heightened risk. This proactive approach aligns with the WHO’s global strategy, which emphasizes prevention, diagnosis, treatment, and harm reduction. [ 2 ]
Global Context and the Role of Direct-Acting Antivirals
The efficacy of DAAs in treating Hepatitis C is remarkable. Phase III clinical trials have consistently demonstrated sustained virologic response (SVR) rates exceeding 95% in diverse patient populations. SVR, defined as undetectable HCV RNA 12 weeks after treatment completion, is generally considered a cure. However, access to these life-saving medications remains a significant barrier globally. The high cost of DAAs, particularly in low- and middle-income countries, has historically limited their availability. Generic versions of several DAAs are now becoming available, significantly reducing treatment costs and expanding access. The development of pan-genotypic DAAs, effective against all six major HCV genotypes, has further simplified treatment regimens.
| Drug Regimen | SVR Rate (%) | Treatment Duration (Weeks) | Common Side Effects |
|---|---|---|---|
| Sofosbuvir/Velpatasvir | 95-98 | 12 | Fatigue, Headache |
| Glecaprevir/Pibrentasvir | 92-95 | 8-16 | Fatigue, Nausea |
| Sofosbuvir/Ribavirin (Genotype 1) | 75-85 | 24 | Anemia, Rash |
Funding for the development of DAAs has primarily come from pharmaceutical companies, including Gilead Sciences and AbbVie. While these companies have invested heavily in research and development, concerns have been raised regarding drug pricing and access. Transparency regarding clinical trial data and funding sources is crucial for maintaining public trust and ensuring equitable access to these medications.
Expanding the Fight: Hepatitis B and Future Challenges
While Kalmar län is making significant strides against Hepatitis C, the region is also intensifying its efforts to combat Hepatitis B. Hepatitis B is a DNA virus that, like HCV, can cause chronic liver disease. Unlike HCV, however, a highly effective vaccine exists for HBV, making prevention a cornerstone of control efforts. However, a significant proportion of the global population remains unvaccinated, and chronic HBV infection remains a major public health concern. Increased awareness campaigns and expanded vaccination programs are essential for reducing the incidence of HBV. Treatment for chronic HBV typically involves antiviral medications that suppress viral replication, but a functional cure – complete elimination of the virus – remains elusive. [ 3 ]
Contraindications & When to Consult a Doctor
DAAs for Hepatitis C are generally well-tolerated, but certain contraindications and potential side effects should be considered. Individuals with severe renal impairment may require dose adjustments. Drug interactions are also possible, particularly with certain medications used to treat HIV or other chronic conditions. While rare, serious adverse events, such as liver decompensation, have been reported. Patients should consult a doctor immediately if they experience symptoms such as jaundice, abdominal pain, or unexplained fatigue during or after treatment. Individuals with pre-existing liver disease should be closely monitored throughout treatment. For Hepatitis B, vaccination is contraindicated in individuals with severe allergic reactions to vaccine components.
The success of Kalmar län demonstrates that hepatitis eradication is achievable. However, sustained progress requires a continued commitment to innovation, equitable access to care, and a holistic public health approach. As Dr. Meg Doherty, Director of the WHO’s Global HIV, Hepatitis and STI Programmes, stated in a recent interview, “Eliminating viral hepatitis requires a coordinated, multi-sectoral response, with strong political commitment and adequate resources.” [ 4 ] The region’s proactive expansion of harm reduction services and screening programs serves as a model for other healthcare systems striving to achieve the WHO’s ambitious 2030 goal.
References
- 1. Pawlotsky, Y. M. (2019). Direct-acting antiviral agents for hepatitis C virus infection. Nature Reviews Gastroenterology & Hepatology, 16(1), 47–62.
- 2. World Health Organization. (n.d.). Hepatitis C.
- 3. Centers for Disease Control and Prevention. (n.d.). Hepatitis B.
- 4. World Health Organization. (2023). WHO releases new hepatitis data revealing progress but urgent need for action.
- 5. European Medicines Agency. (n.d.). Pharmacovigilance.
