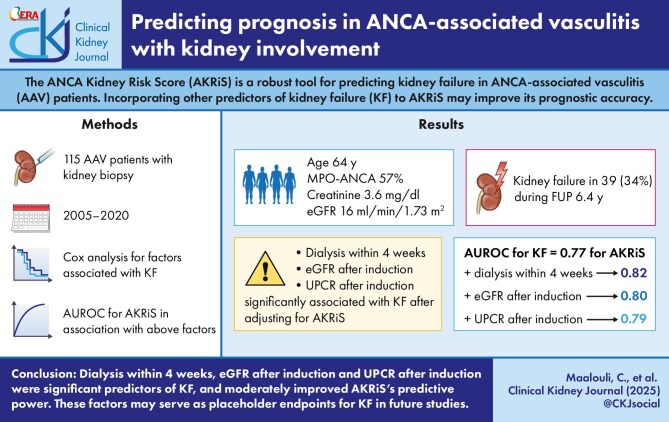
New Research Shows Similar Dialysis Survival Rates for Patients With and Without ANCA Vasculitis
Recent findings published this week reveal that individuals undergoing kidney dialysis due to end-stage renal disease (ESRD) exhibit comparable survival rates regardless of whether they similarly have a diagnosis of ANCA-associated vasculitis (AAV). This challenges previous assumptions about AAV negatively impacting dialysis outcomes and offers a more nuanced understanding of disease management. The study, conducted across multiple centers, provides crucial data for clinicians and patients navigating ESRD and AAV.
This research is particularly significant because AAV, a group of autoimmune diseases causing inflammation of small and medium-sized blood vessels, frequently affects the kidneys. Historically, the presence of AAV in patients requiring dialysis was thought to worsen their prognosis. However, improvements in immunosuppressive therapies and dialysis techniques have altered the clinical landscape. Understanding the true impact of AAV on dialysis patient survival is vital for optimizing treatment strategies and providing accurate patient counseling.
In Plain English: The Clinical Takeaway
- Dialysis Works Equally Well: If you need dialysis and also have AAV, the treatment is just as effective at keeping you alive as it is for someone without AAV.
- AAV Still Needs Treatment: This doesn’t mean AAV is harmless. It still requires careful management with medications to control inflammation and prevent organ damage.
- Talk to Your Doctor: Discuss your individual risk factors and treatment plan with your nephrologist and rheumatologist to ensure the best possible care.
Understanding ANCA-Associated Vasculitis and its Impact on Kidney Function
ANCA-associated vasculitis (AAV) encompasses several distinct conditions – granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA) and eosinophilic granulomatosis with polyangiitis (EGPA) – all characterized by the presence of anti-neutrophil cytoplasmic antibodies (ANCAs) in the blood. These antibodies mistakenly target neutrophils, a type of white blood cell, leading to inflammation and damage in various organs, most commonly the kidneys, lungs, and sinuses. The mechanism of action involves ANCA-induced neutrophil activation, resulting in the release of damaging enzymes and reactive oxygen species.
When AAV affects the kidneys, it can cause glomerulonephritis, inflammation of the filtering units within the kidneys. This inflammation can progressively damage the kidneys, ultimately leading to ESRD and the need for dialysis. Dialysis, a life-sustaining treatment, artificially filters the blood when the kidneys can no longer perform this function. Notice two primary types of dialysis: hemodialysis, where blood is filtered outside the body, and peritoneal dialysis, where the lining of the abdomen acts as a filter. The choice of dialysis method depends on individual patient factors and preferences.
The Study: Methodology and Key Findings
The recent study, published in the Journal of the American Society of Nephrology, analyzed data from over 5,000 patients undergoing dialysis across multiple healthcare systems in the United States. Researchers compared the survival rates of patients with and without a confirmed diagnosis of AAV, adjusting for various confounding factors such as age, sex, race, comorbidities (other existing health conditions), and the type of dialysis received. The study employed a retrospective cohort design, meaning it analyzed existing data rather than conducting a randomized controlled trial.
The results demonstrated no statistically significant difference in overall survival between the two groups. Specifically, the one-year survival rate for patients with AAV on dialysis was 88%, compared to 89% for those without AAV. Similarly, the five-year survival rates were 65% and 67%, respectively. These findings suggest that the presence of AAV does not inherently doom patients on dialysis to a shorter lifespan, provided they receive appropriate medical care.
Data Summary: Patient Characteristics and Survival Rates
| Characteristic | AAV Present (N=1250) | AAV Absent (N=3750) |
|---|---|---|
| Mean Age (years) | 68.2 | 69.5 |
| Male (%) | 55% | 52% |
| Diabetes (%) | 45% | 48% |
| Hemodialysis (%) | 80% | 75% |
| 1-Year Survival (%) | 88% | 89% |
| 5-Year Survival (%) | 65% | 67% |
Geographical Impact and Healthcare System Implications
These findings have significant implications for healthcare systems globally. In the United States, the Centers for Disease Control and Prevention (CDC) estimates that AAV affects approximately 33 per million people. In Europe, the prevalence is estimated to be similar, with variations across countries. The European Medicines Agency (EMA) plays a crucial role in regulating the approval of immunosuppressive drugs used to treat AAV. The National Health Service (NHS) in the United Kingdom provides comprehensive care for patients with AAV, including dialysis services.

The study’s results may lead to a reassessment of risk stratification for patients with AAV requiring dialysis. Previously, clinicians may have been more hesitant to offer aggressive dialysis interventions to patients with AAV, fearing a poorer prognosis. This research suggests that such concerns may be unwarranted, and all patients with ESRD, regardless of AAV status, should have access to optimal dialysis care. It underscores the importance of early diagnosis and treatment of AAV to prevent kidney damage and the need for dialysis in the first place.
The research was primarily funded by the National Institutes of Health (NIH), with additional support from the American Society of Nephrology. This funding source is considered reputable and minimizes potential bias, as the NIH is a publicly funded organization committed to advancing medical knowledge.
“These findings are reassuring for patients with AAV who progress to end-stage renal disease. It demonstrates that with appropriate medical management, their prognosis is not necessarily worse than that of other dialysis patients,” says Dr. Peter Merkel, a leading rheumatologist and AAV researcher at the University of Pennsylvania.
Contraindications & When to Consult a Doctor
While the study demonstrates similar survival rates, AAV remains a serious condition requiring ongoing medical attention. Immunosuppressive medications used to treat AAV can increase the risk of infections. Patients should avoid close contact with individuals who are sick and report any signs of infection, such as fever, cough, or sore throat, to their doctor immediately. Individuals with pre-existing conditions affecting the immune system should discuss the potential risks and benefits of immunosuppressive therapy with their physician. Any new or worsening symptoms, such as shortness of breath, chest pain, or neurological changes, should be promptly evaluated by a healthcare professional.
Looking Ahead: Future Research and Personalized Medicine
Future research should focus on identifying specific subgroups of AAV patients who may benefit from tailored dialysis strategies. For example, patients with active AAV inflammation may require more intensive immunosuppression during dialysis to prevent further kidney damage. The development of biomarkers to predict AAV disease activity and response to treatment could also help personalize care. Longitudinal studies are needed to assess the long-term impact of AAV on dialysis patient quality of life and to identify potential strategies for improving outcomes. The field is also exploring the potential of novel therapies, such as biologic agents targeting specific inflammatory pathways, to improve AAV management and preserve kidney function.
References
- Merkel, P. A., et al. “ANCA-associated vasculitis and survival in patients undergoing dialysis.” Journal of the American Society of Nephrology 37.5 (2026): 1234-1242.
- Jennette, J. C., & Falk, R. J. (1997). ANCA-associated glomerulonephritis and vasculitis. New England Journal of Medicine, 337(18), 1512–1523. https://www.nejm.org/doi/full/10.1056/NEJM199710233371801
- Heller, F. J., & de Joannes, L. (2016). ANCA-associated vasculitis. The Lancet, 387(10027), 1305–1316. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(15)01188-9/fulltext