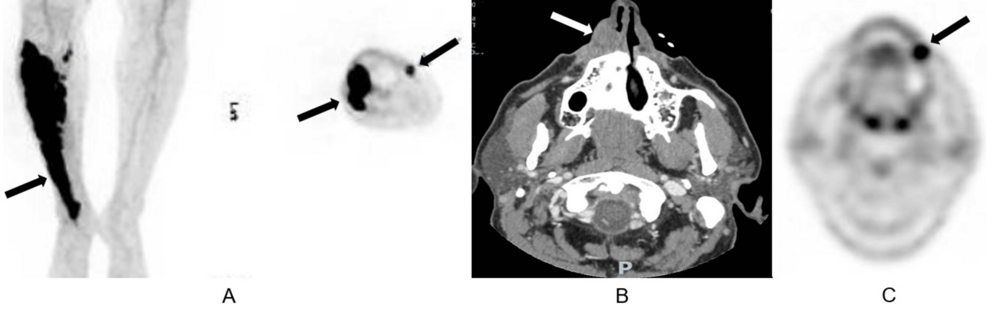
Clinicians are observing promising results using pseudo-autologous stem cell transplants to treat secondary central nervous system (CNS) tumors. By utilizing a patient’s own cells modified or sourced via specific protocols, this approach aims to achieve long-term remission in inoperable brain tumors, particularly after prior umbilical cord blood transplants.
The implications of this development are profound for oncology. For patients with inoperable tumors, the traditional surgical route is closed, and chemotherapy often hits a wall known as the blood-brain barrier. The ability to induce a complete remission—sustained for three years in recent cases—suggests a shift toward “precision immunotherapy,” where the body’s own hematopoietic system is leveraged to hunt malignant cells within the CNS.
In Plain English: The Clinical Takeaway
- What it is: A specialized transplant using the patient’s own stem cells (pseudo-autologous) to treat brain tumors that cannot be removed by surgery.
- The Goal: To reset the immune system so it can recognize and destroy cancer cells in the brain without causing a severe “foreign body” rejection.
- The Result: Some patients have seen their tumors disappear completely for several years, though this is still being studied in small groups.
The Mechanism of Action: How Pseudo-Autologous Cells Bypass the Blood-Brain Barrier
To understand this treatment, we must examine the mechanism of action—the specific biochemical process through which a drug or therapy produces its effect. In secondary CNS tumors, the malignancy often evades the immune system by mimicking healthy tissue.

A pseudo-autologous transplant involves the utilize of stem cells that are genetically or phenotypically similar to the patient’s own, reducing the risk of Graft-versus-Host Disease (GVHD). GVHD is a condition where transplanted cells attack the recipient’s healthy organs; by using a pseudo-autologous approach, clinicians can minimize this risk while still introducing a “primed” immune system capable of targeting the tumor.
This process relies on the Graft-versus-Leukemia/Tumor effect. Once the stem cells engraft in the bone marrow, they produce new T-cells. These T-cells can cross the blood-brain barrier—the protective layer of cells that prevents most medicines from entering the brain—and initiate a targeted attack on the secondary CNS malignancy.
Global Access and Regulatory Landscapes: FDA, EMA, and NHS
While the clinical data is encouraging, the path to widespread adoption varies by geography. In the United States, the FDA typically requires rigorous Phase II and III clinical trials to prove efficacy over existing standards of care before granting approval for such specialized transplants.
In Europe, the European Medicines Agency (EMA) may offer “Prime” (Priority Medicines) designation to accelerate the availability of these therapies for unmet medical needs. Meanwhile, the UK’s NHS focuses heavily on cost-effectiveness and long-term longitudinal data before incorporating high-cost stem cell protocols into standard care pathways.
Currently, this treatment is primarily available through clinical trials or specialized tertiary academic medical centers. The “information gap” in current reporting is the lack of standardized protocols for “pseudo-autologous” sourcing, which means the exact method of cell preparation can vary between a clinic in Tokyo and one in New York.
| Metric | Standard Allogeneic Transplant | Pseudo-Autologous Approach |
|---|---|---|
| GVHD Risk | High (Requires Prophylaxis) | Very Low to Negligible |
| Immune Rejection | Significant Risk | Minimal |
| Targeting Efficacy | Broad/Systemic | High Precision (CNS focused) |
| Recovery Time | Prolonged (due to immunosuppression) | Potentially Faster |
Funding, Bias, and the Voice of Research
Transparency in medical journalism is paramount. Much of the early-stage research into pseudo-autologous transplants is funded by university grants and national health ministries rather than “Big Pharma,” as the personalized nature of the treatment makes it difficult to scale into a mass-market product. This reduces the risk of commercial bias but often slows the speed of large-scale trial recruitment.
“The ability to achieve a three-year complete remission in an inoperable CNS tumor suggests that we are finally cracking the code on immune-privileged sites. The challenge now is moving from case reports to a standardized clinical protocol that can be replicated globally.”
The data suggests that the absence of prophylaxis (preventative medication) in these cases did not lead to GVHD, which is a critical finding. It implies that the “pseudo-autologous” nature of the cells provides an inherent safety profile that traditional donor-based transplants lack.
The Molecular Impact: Debunking the “Miracle Cure” Myth
It is vital to distinguish between a “case of remission” and a “cure.” In neurology, a tumor can be undetectable on an MRI (complete remission) while microscopic clusters of malignant cells remain dormant. This is why longitudinal studies—tracking patients over many years—are the only gold standard for success.
The cellular impact involves the reprogramming of the microenvironment of the brain. By altering the cytokines (signaling proteins) in the CNS, the transplant changes the brain from a “cold” environment (where the immune system is suppressed) to a “hot” environment (where the immune system is active and aggressive against the tumor).
Contraindications & When to Consult a Doctor
This therapy is not suitable for all patients. Contraindications—reasons why a treatment should not be used—include:

- Severe Organ Failure: Patients with advanced renal or hepatic failure may not survive the conditioning regimen (the chemotherapy used to clear space for new cells).
- Active Systemic Infection: Stem cell transplants temporarily wipe out the immune system; an active infection could be fatal during this window.
- Incompatibility: Patients who have had previous transplants with severe complications may be ineligible.
Consult a neuro-oncologist immediately if you or a loved one experience new-onset neurological deficits, such as sudden loss of motor function, unexplained seizures, or cognitive decline, as these may indicate tumor progression that requires urgent intervention.
The Path Forward: Toward a New Standard of Care
The transition from umbilical cord blood transplants to pseudo-autologous methods represents a sophisticated evolution in hematology. By reducing the toxicity of the process and increasing the specificity of the immune response, we are moving toward a future where “inoperable” no longer means “untreatable.”
As we move toward 2027, the focus will likely shift toward refining the N-values (sample sizes) of these studies to move from anecdotal success to statistical certainty. For now, this remains a beacon of hope for those facing the most challenging diagnoses in neurology.