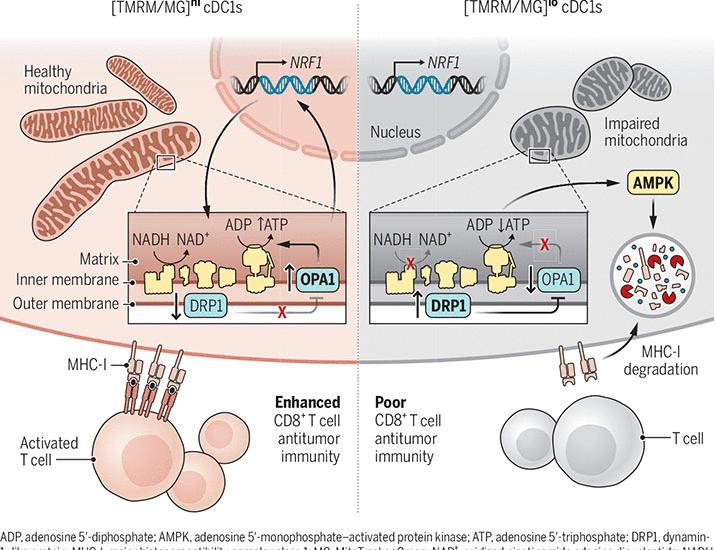
Recent research published this week in Science reveals that a specific subset of dendritic cells requires optimal mitochondrial fitness—the energy-producing capacity of the cell—to effectively trigger antitumor immune responses in mice. This discovery identifies a critical metabolic checkpoint that determines whether the immune system attacks or ignores malignant tumors.
For patients and clinicians, this represents a paradigm shift in how we view immunotherapy. We have long focused on the “instructions” we give the immune system (such as checkpoint inhibitors); however, this research suggests that if the cellular “engine” (the mitochondria) is failing, the instructions are irrelevant. By targeting the metabolic health of dendritic cells, we may unlock new ways to treat refractory cancers that currently evade the immune system.
In Plain English: The Clinical Takeaway
- The Energy Connection: Your immune cells need a specific type of “fuel” efficiency from their mitochondria to recognize and kill cancer cells.
- Not All Cells are Equal: Only a specific subset of dendritic cells (the scouts of the immune system) are the primary drivers of this mitochondrial-dependent response.
- Future Therapy: This opens the door for “metabolic priming,” where doctors might improve a patient’s cellular energy before administering immunotherapy to increase success rates.
The Metabolic Engine: How Mitochondrial Fitness Dictates Immune Surveillance
To understand this breakthrough, we must examine the mechanism of action—the specific biochemical process through which a drug or biological process produces its effect. In this instance, the researchers focused on dendritic cells (DCs), which act as the bridge between the innate and adaptive immune systems.

The study demonstrates that for these DCs to migrate to lymph nodes and activate T-cells, they require high levels of mitochondrial membrane potential. When mitochondria are dysfunctional, the DCs enter a state of metabolic exhaustion, rendering them unable to present tumor antigens (the “red flags” of cancer) to the rest of the immune system.
This process involves oxidative phosphorylation (the process where cells use oxygen to create ATP, or cellular energy). If this pathway is compromised, the immune system remains “blind” to the tumor, even if the tumor is highly immunogenic. This explains why some patients do not respond to current PD-1/PD-L1 inhibitors, as their dendritic cells may simply lack the energy to initiate the attack.
Bridging the Gap: From Murine Models to Global Clinical Application
Whereas the primary study was conducted in mice, the translational implications for human healthcare are significant. In the United States, the FDA is increasingly scrutinizing “metabolic biomarkers” to predict patient response to immunotherapy. In Europe, the EMA is exploring similar frameworks to reduce the administration of expensive, ineffective treatments to patients whose cellular metabolism cannot support the response.
The funding for this research typically stems from academic grants and National Institutes of Health (NIH) funding, though the translation into human trials often involves pharmaceutical partnerships. Transparency regarding funding is vital due to the fact that the development of “mitochondrial boosters” represents a multi-billion dollar market opportunity for biotech firms.
“The ability to modulate the metabolic state of the immune system represents the next frontier in oncology. We are moving from simply ‘unleashing’ the immune system to actively ‘powering’ it.”
The following table summarizes the comparative metabolic requirements of immune cells in healthy versus tumor-suppressed environments:
| Cellular State | Mitochondrial Activity | T-Cell Activation Rate | Clinical Outcome |
|---|---|---|---|
| Metabolically Fit DC | High ATP Production | Robust/Sustained | Tumor Regression |
| Metabolically Exhausted DC | Low Membrane Potential | Minimal/Absent | Tumor Progression |
| Checkpoint-Inhibited DC | Variable | Blocked by Ligands | Immune Evasion |
The Regulatory Horizon and Patient Access
The transition from mouse models to human application requires double-blind placebo-controlled trials—the gold standard of research where neither the patient nor the doctor knows who is receiving the treatment, eliminating bias. We are currently awaiting Phase I trials that will test whether mitochondrial antioxidants or metabolic precursors can “prime” the immune system in humans.
In the UK, the NHS may eventually integrate metabolic screening into the standard of care for oncology. If a patient’s dendritic cells show low mitochondrial fitness, clinicians could potentially administer a metabolic “booster” before starting chemotherapy or immunotherapy, significantly improving the statistical probability of a positive outcome.
Contraindications & When to Consult a Doctor
It is imperative to note that “mitochondrial support” should not be sought through unregulated supplements. Many over-the-counter “mitochondrial boosters” lack peer-reviewed evidence and can interfere with chemotherapy.
Consult your oncologist immediately if you experience:
- Unexplained fatigue that does not respond to rest (potential sign of systemic metabolic dysfunction).
- Rapid progression of a tumor despite adherence to immunotherapy.
- Severe adverse reactions to checkpoint inhibitors, such as colitis or pneumonitis.
Patients with pre-existing mitochondrial diseases or severe metabolic disorders may have contraindications (reasons why a specific treatment should not be used) regarding certain metabolic stimulants, as over-stimulating a damaged mitochondrion can lead to the production of harmful reactive oxygen species (ROS).
The Future of Immunometabolism
The discovery that mitochondria power the immune response against cancer moves us closer to a personalized medicine model. We are no longer looking at cancer as just a genetic mutation, but as a metabolic failure of the host’s defense system. By restoring the energy balance within the immune system, we can potentially turn “cold” tumors (those the immune system ignores) into “hot” tumors (those the immune system actively destroys).
References
- Science Magazine – Original Research Publication.
- PubMed – Mitochondrial Metabolism in Dendritic Cells.
- World Health Organization (WHO) – Global Cancer Research Frameworks.
- Centers for Disease Control and Prevention (CDC) – Public Health Oncology Statistics.