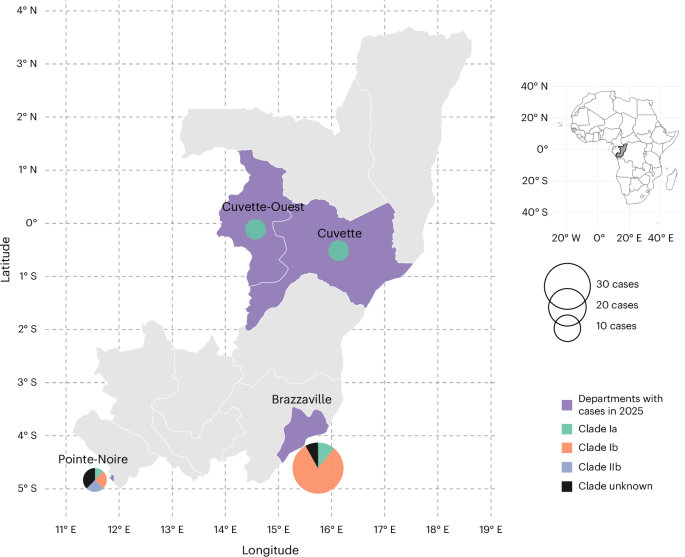
New genomic data confirms monkeypox virus clade IIb lineage A.2.2 in the Republic of the Congo. Co-circulation with clades Ia and Ib raises recombination concerns. Enhanced surveillance is critical to prevent potential viral evolution and manage public health risk globally.
This week’s publication in Nature Medicine signals a pivotal shift in the epidemiological landscape of orthopoxviruses. For patients and public health officials alike, the detection of clade IIb lineage A.2.2 outside its typical geographic stronghold indicates that viral boundaries are permeable. The core concern is not merely the presence of the virus, but the co-circulation of distinct clades within the same host population. When multiple lineages infect a single host, the biological mechanism of homologous recombination becomes possible. This process allows the virus to exchange genetic material, potentially creating a hybrid variant that combines the high transmissibility of clade IIb with the historical severity associated with clade I.
In Plain English: The Clinical Takeaway
- Viral Mixing Risk: Different strains of the virus are now circulating in the same region, which could theoretically allow them to swap genes and change behavior.
- Surveillance is Key: Health authorities are increasing genetic testing to catch any new variants early before they spread widely.
- Patient Action: Standard prevention methods remain effective; watch for unexplained rashes or fever and seek testing if exposed.
Understanding the Genomic Shift in Central Africa
Historically, the Republic of the Congo has been endemic for clade I (formerly Congo Basin clade), which carries a higher case fatality rate compared to clade II (formerly West African clade). The 2022 global outbreak was driven primarily by clade IIb. Finding lineage A.2.2—the specific sub-lineage responsible for the global outbreak—now established in Central Africa suggests sustained human-to-human transmission chains have bridged previous geographic gaps. This is not merely a travel-related case but indicates local establishment.

From a mechanism of action perspective, orthopoxviruses are large DNA viruses with complex genomes. While DNA viruses generally mutate slower than RNA viruses, the sheer volume of infections during the 2022-2025 period provided ample opportunity for adaptation. The concern regarding recombination events is grounded in virology precedent. If a patient is co-infected with clade Ia and clade IIb, the viral replication machinery may splice genetic segments from both parents. The resulting progeny could evade existing immunity or display altered tissue tropism, affecting how the virus targets human cells.
Global health security relies on genomic surveillance. The ability to sequence viral genomes in real-time allows researchers to track these mutations. While, capacity remains uneven. In many regions of Central Africa, sequencing infrastructure is limited, creating blind spots where viral evolution could occur undetected. This study underscores the urgent need to decentralize sequencing capabilities to match the virus’s mobility.
Geo-Epidemiological Bridging and Regulatory Impact
The implications extend beyond the Republic of the Congo. For regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), viral evolution poses a challenge to diagnostic and therapeutic efficacy. Current PCR diagnostics target conserved regions of the viral genome. If recombination alters these target regions, false negatives could occur. Similarly, antiviral treatments like tecovirimat target specific viral envelope proteins. Significant genomic shifts could theoretically reduce drug binding affinity, though current data suggests resilience.
Travel medicine protocols may require updates. Clinicians in non-endemic countries must maintain a high index of suspicion for patients presenting with vesicular pustular rashes, regardless of travel history to traditional endemic zones. The World Health Organization (WHO) continues to emphasize that border closures are ineffective against established transmission. Instead, the focus remains on rapid identification and isolation.
Funding for this critical surveillance often comes from coalitions such as the Coalition for Epidemic Preparedness Innovations (CEPI) and national institutes of health. Transparency in funding is vital to ensure data independence. Public health research of this magnitude typically relies on multi-national grants to support the logistics of sample collection and bioinformatics analysis in resource-limited settings.
“Genomic surveillance is our early warning system. When we see clades mixing, we must assume the virus is testing its evolutionary options. Our job is to ensure public health measures evolve faster than the pathogen.”
— WHO Mpox Technical Advisory Group, Public Statement on Orthopoxvirus Surveillance
Clinical Comparisons and Risk Stratification
To understand the stakes, one must differentiate the clinical profiles of the circulating clades. Clade I has historically been associated with more severe disease outcomes, including higher rates of hospitalization and mortality, particularly among children and immunocompromised individuals. Clade IIb, while highly transmissible, generally presents with lower mortality rates in healthy adults. The table below summarizes the current understanding of these differences based on cumulative epidemiological data.
| Feature | Clade I (Congo Basin) | Clade IIb (Global Outbreak) |
|---|---|---|
| Historical Fatality Rate | Up to 10% (untreated) | <1% (in high-resource settings) |
| Primary Transmission | Zoonotic & Household | Close Contact & Sexual Networks |
| Incubation Period | 7-14 Days | 6-13 Days |
| Severity Markers | Higher fever, lymphadenopathy | Localized lesions, systemic symptoms vary |
This data highlights why the co-circulation in the Republic of the Congo is monitored so closely. If a recombinant virus emerges with the transmissibility of IIb and the virulence of I, the global impact could be significant. However, it is crucial to maintain objective statistical probability. Recombination is a possibility, not a certainty. Most co-infections do not result in viable recombinant offspring due to biological constraints.
Contraindications & When to Consult a Doctor
Patients should not panic, but vigilance is required. There are no specific contraindications for general public health measures, but certain groups face higher risks. Individuals who are immunocompromised, such as those undergoing chemotherapy or living with untreated HIV, should be particularly cautious. The smallpox vaccine (MVA-BN) is generally safe but may have reduced efficacy in severely immunosuppressed patients.
Consult a healthcare provider immediately if you develop a new, unexplained rash, especially if accompanied by fever, chills, or swollen lymph nodes. This is critical if you have had close contact with a confirmed case or have traveled to regions with known active transmission. Do not attempt to self-diagnose using online images, as many dermatological conditions mimic orthopoxvirus lesions. Early isolation prevents household transmission, which remains the most common vector for spread outside of specific network clusters.
The Path Forward: Surveillance and Stability
The detection of lineage A.2.2 in the Republic of the Congo is a testament to improved detection capabilities rather than necessarily a sudden viral explosion. It confirms that the virus has not disappeared but has integrated into endemic regions. The scientific community must now focus on longitudinal studies to monitor whether these co-circulating lineages remain stable or begin to merge genetically.
For the public, the guidance remains consistent with established protocols: hygiene, avoidance of close contact with symptomatic individuals, and vaccination for high-risk groups. The medical community’s role is to ensure that diagnostic tools remain updated against evolving genomic sequences. By maintaining rigorous surveillance without succumbing to alarmism, we can manage the risk of recombination while protecting global health security.
References
- Nature Medicine: Evidence of monkeypox virus clade IIb lineage A.2.2 in the Republic of the Congo
- Centers for Disease Control and Prevention: Mpox (Monkeypox) Information
- World Health Organization: Mpox Global Task Force Updates
- PubMed Central: Orthopoxvirus Genomic Surveillance Studies
- The Lancet Infectious Diseases: Clinical Severity of Mpox Clades