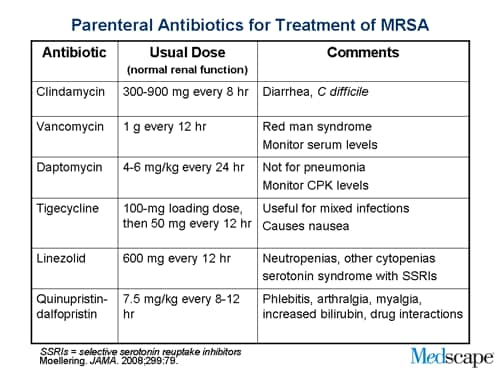
Recent correspondence regarding community-acquired pneumonia (CAP) highlights the evolving landscape of treatment options for methicillin-resistant Staphylococcus aureus (MRSA). While modern therapies are emerging, experts emphasize that broad-spectrum MRSA coverage should remain reserved for patients exhibiting specific risk factors or confirmed infection, avoiding unnecessary antibiotic apply and mitigating the rise of antimicrobial resistance.
The increasing prevalence of MRSA in CAP, once considered primarily a hospital-acquired infection, presents a significant challenge to clinicians globally. Overuse of antibiotics, particularly broad-spectrum agents, has driven the selection and spread of MRSA strains within communities. This necessitates a nuanced approach to empirical therapy – treatment initiated before definitive pathogen identification – balancing the need for effective coverage against the potential harms of widespread antibiotic resistance. The debate isn’t about *if* we treat MRSA, but *when* and *how* judiciously.
In Plain English: The Clinical Takeaway
- Not Everyone Needs Strong Antibiotics: If you have pneumonia, your doctor won’t automatically provide you antibiotics that fight MRSA. These stronger drugs are reserved for people at higher risk.
- Risk Factors Matter: Being recently hospitalized, having a weakened immune system, or having previously confirmed MRSA infection increases your risk and may warrant broader antibiotic coverage.
- Early Diagnosis is Key: Rapid diagnostic tests are becoming more common, helping doctors quickly identify the specific cause of pneumonia and tailor treatment accordingly.
The Expanding Arsenal Against MRSA: Beyond Vancomycin
For decades, vancomycin has been the mainstay of MRSA treatment. However, rising rates of vancomycin-intermediate Staphylococcus aureus (VISA) and vancomycin-resistant Staphylococcus aureus (VRSA) – though still relatively uncommon – are prompting the development and adoption of alternative agents. These include newer beta-lactams like ceftaroline, daptomycin, linezolid, and telavancin. Each possesses a unique mechanism of action, targeting different aspects of bacterial cell wall synthesis or protein production. Ceftaroline, for example, exhibits a high affinity for penicillin-binding protein 2a (PBP2a), a modified penicillin-binding protein responsible for methicillin resistance. Daptomycin, a lipopeptide, inserts into the bacterial cell membrane, causing depolarization and ultimately cell death.
Geographical Variations and Regulatory Pathways
The adoption of these newer agents varies significantly across geographical regions. In the United States, the Food and Drug Administration (FDA) has approved all four alternatives, but access can be limited by formulary restrictions and cost. The European Medicines Agency (EMA) has similarly approved these drugs, but national healthcare systems, like the National Health Service (NHS) in the UK, employ health technology assessments to determine cost-effectiveness and reimbursement policies. Antimicrobial resistance action plans are central to EMA’s strategy. Epidemiological data reveals a higher prevalence of community-acquired MRSA in certain regions, such as the Southeastern United States and parts of Eastern Europe, necessitating more aggressive empirical treatment strategies in those areas. A 2023 CDC report indicated a 37% increase in invasive MRSA infections among adults aged 65 years and older between 2017 and 2022, highlighting the growing public health concern.
Funding and Bias Considerations
It’s crucial to acknowledge potential biases inherent in clinical research. Many studies evaluating newer anti-MRSA agents are funded, directly or indirectly, by pharmaceutical companies. While this doesn’t automatically invalidate the findings, it necessitates careful scrutiny of study design, data analysis, and reporting. For instance, a significant portion of the clinical trials supporting ceftaroline’s approval were funded by Forest Pharmaceuticals (now part of Allergan). Transparency regarding funding sources is paramount to maintaining public trust and ensuring evidence-based clinical practice. The ClinicalTrials.gov database provides a valuable resource for identifying funding sources and study protocols.
“The challenge isn’t simply developing new antibiotics, but ensuring their responsible use. We need to move towards a model of ‘antimicrobial stewardship’ where antibiotics are prescribed only when truly necessary, and the right drug is used for the right infection, at the right dose, and for the right duration.”
Clinical Trial Data and Efficacy Profiles
The efficacy of alternative anti-MRSA agents has been evaluated in numerous randomized, double-blind, placebo-controlled clinical trials. Linezolid, for example, demonstrated non-inferiority to vancomycin in the treatment of complicated skin and skin structure infections caused by MRSA. However, linezolid is associated with a risk of thrombocytopenia (low platelet count) and peripheral neuropathy, limiting its long-term use. Daptomycin has shown excellent efficacy in treating bacteremia (bloodstream infection) caused by MRSA, but This proves not effective against pneumonia due to inactivation by pulmonary surfactant. Ceftaroline has demonstrated comparable efficacy to vancomycin in treating CAP caused by MRSA, with a potentially lower risk of nephrotoxicity (kidney damage).
| Agent | Mechanism of Action | Common Side Effects | FDA Approval (CAP) |
|---|---|---|---|
| Vancomycin | Inhibits cell wall synthesis | Nephrotoxicity, Red Man Syndrome | Yes |
| Linezolid | Inhibits protein synthesis | Thrombocytopenia, Peripheral Neuropathy | No |
| Daptomycin | Causes cell membrane depolarization | Myopathy, Elevated Creatine Kinase | No |
| Ceftaroline | Binds to PBP2a | Diarrhea, Rash | Yes |
Contraindications & When to Consult a Doctor
Individuals with known allergies to any of the beta-lactam antibiotics should avoid ceftaroline. Patients with pre-existing thrombocytopenia or peripheral neuropathy should use linezolid with caution. Daptomycin is contraindicated in patients with active myopathy (muscle disease). If you experience symptoms of pneumonia – fever, cough, shortness of breath, chest pain – seek immediate medical attention. Do not self-treat with antibiotics. Any signs of allergic reaction (rash, hives, difficulty breathing) after starting an antibiotic require immediate medical intervention.
The future of MRSA treatment in CAP lies in a combination of strategies: judicious antibiotic use guided by rapid diagnostics, the development of novel antimicrobial agents, and the implementation of robust infection control measures. Continued surveillance of antimicrobial resistance patterns and investment in research are essential to staying ahead of this evolving threat. The focus must shift from simply treating infections to preventing them in the first place.
References
- Corey GR, et al. Ceftaroline for Community-Acquired Bacterial Pneumonia. N Engl J Med. 2020;382(2):129-139.
- Centers for Disease Control and Prevention. Community-Associated MRSA.
- European Medicines Agency. Antimicrobial Resistance Action Plan.
- ClinicalTrials.gov
- World Health Organization. Antimicrobial Resistance.