Natera’s Signatera test, a circulating tumor DNA (ctDNA) assay, may allow select breast cancer patients—particularly those over 70—to safely avoid surgery. By detecting molecular residual disease (MRD), the test identifies patients with a negligible risk of recurrence, shifting the paradigm from standardized surgery to personalized, biology-driven care.
For decades, the clinical gold standard for breast cancer has been a combination of systemic therapy and surgical intervention to ensure all malignant tissue is removed. Yet, for elderly patients or those with significant comorbidities, the physiological toll of surgery can sometimes outweigh the oncological benefit. The emergence of “molecularly guided de-escalation” represents a pivotal shift in oncology, where the absence of detectable tumor DNA in the blood serves as a proxy for a “cure,” potentially sparing thousands of women from invasive procedures.
In Plain English: The Clinical Takeaway
- What it is: A “liquid biopsy” that looks for tiny fragments of cancer DNA circulating in your bloodstream.
- The Goal: To identify patients whose cancer is completely gone after initial treatment, meaning surgery may no longer be necessary to prevent recurrence.
- The Caveat: This is not a general screening tool. it is a specialized test for patients already diagnosed with cancer to determine the next steps in their treatment.
How Tumor-Informed ctDNA Redefines Surgical Necessity
To understand how Signatera functions, one must first understand the mechanism of action of circulating tumor DNA (ctDNA). When cancer cells undergo apoptosis (programmed cell death) or necrosis, they shed fragments of their genetic material into the plasma. Signatera is a “tumor-informed” assay, meaning it first sequences the patient’s specific tumor tissue to identify unique mutations. It then creates a personalized set of probes to search for those exact mutations in the blood.
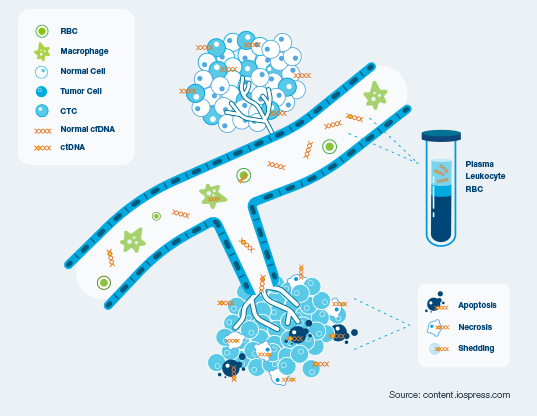
This process identifies Molecular Residual Disease (MRD)—the small number of cancer cells that remain in the body after primary treatment, which are too few to be seen on a CT scan or MRI. In the context of the recent breast cancer data, a “negative” MRD result indicates that the cancer is undetectable at a molecular level. For a 75-year-ancient patient with multiple health complications, this negative result suggests that the risk of the cancer returning is statistically low enough that the morbidity of surgery (such as infection or cardiovascular stress) becomes the primary risk factor.
The statistical significance of these findings lies in the high negative predictive value (NPV) of the test. When a test has a high NPV, it means that a negative result is highly reliable in ruling out the presence of the disease. By integrating peer-reviewed genomic data, clinicians can move away from “treating the stage” and start “treating the biology.”
Global Regulatory Hurdles and Patient Access
While the clinical data is promising, the transition from a study result to a standard of care depends heavily on regional healthcare infrastructure. In the United States, the FDA (Food and Drug Administration) regulates the clinical validity of such tests, but the primary hurdle is reimbursement from private insurers and Medicare. Without broad coverage, this technology remains a luxury for affluent patients.
In the United Kingdom, the NHS (National Health Service) typically requires a cost-effectiveness analysis from NICE (National Institute for Health and Care Excellence) before adopting liquid biopsies into routine care. For the NHS, the value proposition is clear: if a blood test can safely replace a surgical theater slot and a hospital stay, the systemic cost savings are immense. Similarly, the EMA (European Medicines Agency) focuses on the rigorous validation of the assay’s sensitivity to ensure that “false negatives” do not lead to undertreatment.
“The integration of ctDNA into the surgical decision-making process represents the next frontier of precision oncology. We are moving toward a future where the molecular signature of the patient dictates the scalpel, not the size of the tumor on an image.”
This shift is supported by broader epidemiological trends. As the global population ages, the prevalence of “geriatric oncology” increases. The need for non-invasive monitoring is no longer a convenience; it is a clinical necessity to maintain the quality of life in an aging patient cohort.
Comparative Analysis: Standard Care vs. MRD-Guided Care
| Metric | Standard Surgical Protocol | MRD-Guided Approach (Signatera) |
|---|---|---|
| Decision Driver | Tumor size & lymph node status | Presence of ctDNA in plasma |
| Patient Risk | Surgical complications/Anesthesia | Risk of a false negative result |
| Recovery Time | Weeks to months | Immediate (Blood draw only) |
| Primary Goal | Physical removal of mass | Molecular confirmation of eradication |
| Patient Cohort | Universal application | Targeted (e.g., Elderly, High-comorbidity) |
Funding Transparency and Potential Bias
It is imperative for patients and providers to recognize that the primary data supporting these findings are often generated or funded by Natera, the company that owns and commercializes the Signatera test. While the results have been presented in clinical settings, company-funded research can inherently lean toward highlighting efficacy. To maintain journalistic and medical integrity, these results must be validated by independent, third-party longitudinal studies—such as those published in The Lancet or JAMA—to ensure that skipping surgery does not lead to higher long-term recurrence rates in a statistically significant way.
Contraindications & When to Consult a Doctor
The ability to avoid surgery is not applicable to all breast cancer patients. This approach is generally contraindicated for:
- High-Risk Genetic Profiles: Patients with BRCA1 or BRCA2 mutations may still require prophylactic or therapeutic surgery regardless of ctDNA status due to the high risk of secondary primaries.
- Early-Stage Screening: Signatera is not a screening tool for the general population; using it to “rule out” cancer in a healthy person is clinically inappropriate and dangerous.
- Symptomatic Progression: If a patient has a negative ctDNA result but presents with a palpable lump or new imaging findings, the imaging and physical exam seize precedence over the blood test.
Patients should consult their oncology team immediately if they experience new breast lumps, unexplained skin changes, or persistent bone pain, regardless of their last liquid biopsy result.
The Future Trajectory of Liquid Biopsies
The valuation of Natera (NTRA) is currently tethered to the market’s belief that Signatera will become the “gatekeeper” for surgical intervention. If the medical community accepts ctDNA as a valid reason to skip surgery, the addressable market expands from “monitoring” to “diagnostic decision-making.”
However, the path forward requires a cautious, evidence-based approach. The goal of medicine is to maximize survival while minimizing suffering. If we can prove that a molecularly negative patient has the same 10-year survival rate without surgery as they would with it, we have achieved a triumph of precision medicine. Until then, these tests should be viewed as powerful adjuncts to—not replacements for—the multidisciplinary tumor board’s expertise.

