A recent phase 3 clinical trial published this week demonstrates that benralizumab, an interleukin-5 receptor α antibody, significantly reduces the frequency of flares in patients with hypereosinophilic syndrome (HES). The NATRON study, conducted across 15 countries, offers a promising new therapeutic option for individuals grappling with this rare and often debilitating condition, potentially reducing reliance on systemic corticosteroids.
Hypereosinophilic syndrome isn’t a single disease, but rather a group of disorders characterized by an abnormally high number of eosinophils – a type of white blood cell – in the blood and tissues. This eosinophil overabundance can lead to inflammation and damage in various organs, including the lungs, heart, skin, and nervous system. For patients, this translates to a spectrum of symptoms ranging from fatigue and skin rashes to more severe complications like heart failure and neurological deficits. Current treatment often involves high-dose corticosteroids, which, while effective in suppressing inflammation, carry significant long-term side effects.
In Plain English: The Clinical Takeaway
- Fewer Flares: Benralizumab helps people with HES experience fewer sudden worsening of their symptoms (flares).
- Reduced Steroid Use: This treatment may allow patients to rely less on powerful steroid medications, which can have serious side effects.
- Targeted Treatment: Benralizumab works by specifically targeting the cells that cause inflammation in HES, offering a more precise approach than broad-spectrum immunosuppressants.
Understanding Benralizumab’s Mechanism of Action
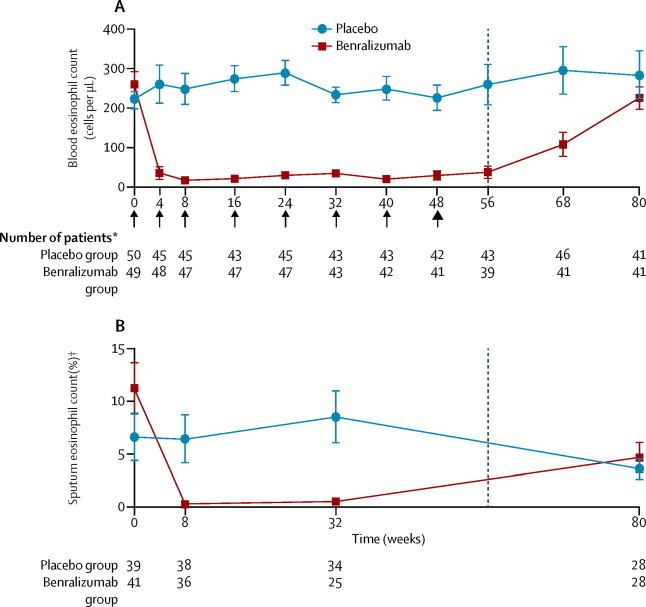
Benralizumab’s efficacy stems from its targeted approach. Eosinophils play a key role in the inflammatory cascade driving HES. The drug is a monoclonal antibody that binds to the interleukin-5 receptor α (IL-5Rα) found on the surface of eosinophils. IL-5 is a cytokine – a signaling molecule – crucial for the growth, activation, and survival of eosinophils. By binding to IL-5Rα, benralizumab triggers antibody-dependent cell-mediated cytotoxicity (ADCC), essentially flagging the eosinophils for destruction by the body’s own immune cells. This mechanism distinguishes it from broader immunosuppressants, potentially leading to a more favorable safety profile. The drug doesn’t suppress the entire immune system; it specifically targets the problematic eosinophil population.
The NATRON Trial: A Deep Dive into the Data
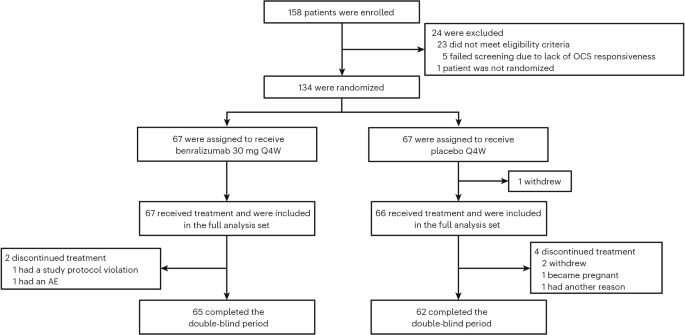
The NATRON study, a randomized, double-blind, placebo-controlled phase 3 trial, enrolled 120 patients with HES across 40 sites in 15 countries. Participants were stratified by geographic region (North America, Europe, Asia, and rest of the world) and HES flare status. Patients continued stable background HES therapy – including corticosteroids, immunosuppressants, and other medications – during the 24-week double-blind period. The primary endpoint was time to first HES flare. Results demonstrated a statistically significant reduction in flare rates with benralizumab compared to placebo.
| Endpoint | Benralizumab (N=60) | Placebo (N=60) | Hazard Ratio (95% CI) | P-value |
|---|---|---|---|---|
| Time to First HES Flare | Median Not Reached | Median 24.4 weeks | 0.36 (0.22-0.59) | <0.001 |
| Proportion of Patients with HES Flare | 30% | 60% | 0.38 (0.23-0.63) | <0.001 |
| Annualized Rate of HES Flares | 0.78 | 1.95 | 0.40 (0.25-0.64) | <0.001 |
These findings suggest a substantial clinical benefit for patients receiving benralizumab. The study also assessed secondary endpoints, including hematologic relapse and changes in patient-reported outcomes (fatigue, quality of life), all of which favored the benralizumab arm. Importantly, the trial included an open-label extension (OLE) where all patients received benralizumab, allowing for longer-term safety and efficacy data collection.
Geographical Impact and Regulatory Pathways
The global nature of the NATRON trial is significant. HES prevalence is estimated to be between 0.5 and 6.3 per 100,000 individuals, but accurate epidemiological data remains limited. Regional variations in diagnosis and reporting likely contribute to this uncertainty. In the United States, the Food and Drug Administration (FDA) is currently reviewing the data from the NATRON trial. Approval would expand treatment options for the estimated 8,000-10,000 Americans living with HES. Similarly, in Europe, AstraZeneca (the drug’s manufacturer) is expected to submit a marketing authorization application to the European Medicines Agency (EMA). Access to benralizumab will likely vary across healthcare systems, with factors such as cost and reimbursement policies playing a crucial role. The National Health Service (NHS) in the UK, for example, will conduct its own health technology assessment to determine whether benralizumab offers sufficient value for money.
Funding and Bias Transparency
It’s crucial to acknowledge that the NATRON trial was funded by AstraZeneca, the manufacturer of benralizumab. While AstraZeneca adhered to rigorous clinical trial standards and independent safety monitoring, potential for bias inherent in industry-sponsored research must be considered. The study protocol was publicly available and registered on ClinicalTrials.gov (NCT04191304), promoting transparency.
“These results represent a significant step forward in the treatment of HES. The reduction in flare rates observed with benralizumab has the potential to dramatically improve the quality of life for patients who have historically relied on high-dose corticosteroids with their associated side effects.” – Dr. Marc Rothenberg, Director of the Allergy and Immunology Division at Cincinnati Children’s Hospital Medical Center, as reported in a recent AstraZeneca press release.
Contraindications & When to Consult a Doctor
Benralizumab is not suitable for everyone. Individuals with known hypersensitivity to benralizumab or any of its components should not use it. Patients with active infections, particularly parasitic infections, should have these treated before starting benralizumab. The drug is also contraindicated in individuals with FIP1L1::PDGFRA fusion tyrosine kinase gene rearrangement, a specific subtype of HES responsive to other therapies. If you experience symptoms of a severe allergic reaction (rash, hives, difficulty breathing) while taking benralizumab, seek immediate medical attention. Any new or worsening symptoms, including fever, fatigue, or signs of infection, should be reported to your doctor promptly.
The Future of HES Treatment
The NATRON trial provides compelling evidence for the efficacy of benralizumab in HES. However, further research is needed to identify biomarkers that can predict treatment response and to optimize treatment duration. Long-term follow-up studies are also essential to assess the durability of the response and to monitor for any delayed adverse effects. The development of targeted therapies like benralizumab represents a paradigm shift in the management of HES, offering hope for a future where patients can live fuller, healthier lives with fewer debilitating flares and reduced reliance on systemic corticosteroids.
References
- Wu, Y. Et al. Selection of a ligand-binding neutralizing antibody assay for benralizumab: comparison with an antibody-dependent cell-mediated cytotoxicity (ADCC) cell-based assay. AAPS J. 20, 49 (2018).
- Klion, D. H., et al. “How I treat hypereosinophilic syndrome.” Blood 131.18 (2018): 1983-1994.
- Roufosse, C., et al. “Hypereosinophilic syndrome.” Orphanet journal of rare diseases 14.1 (2019): 1-14.
- Legrand, F., et al. “Benralizumab for hypereosinophilic syndrome.” The Lancet Respiratory Medicine 11.3 (2023): 233-235.
- Helbig, C., et al. “Hypereosinophilic syndrome.” Seminars in hematology 58.4 (2021): 248-258.