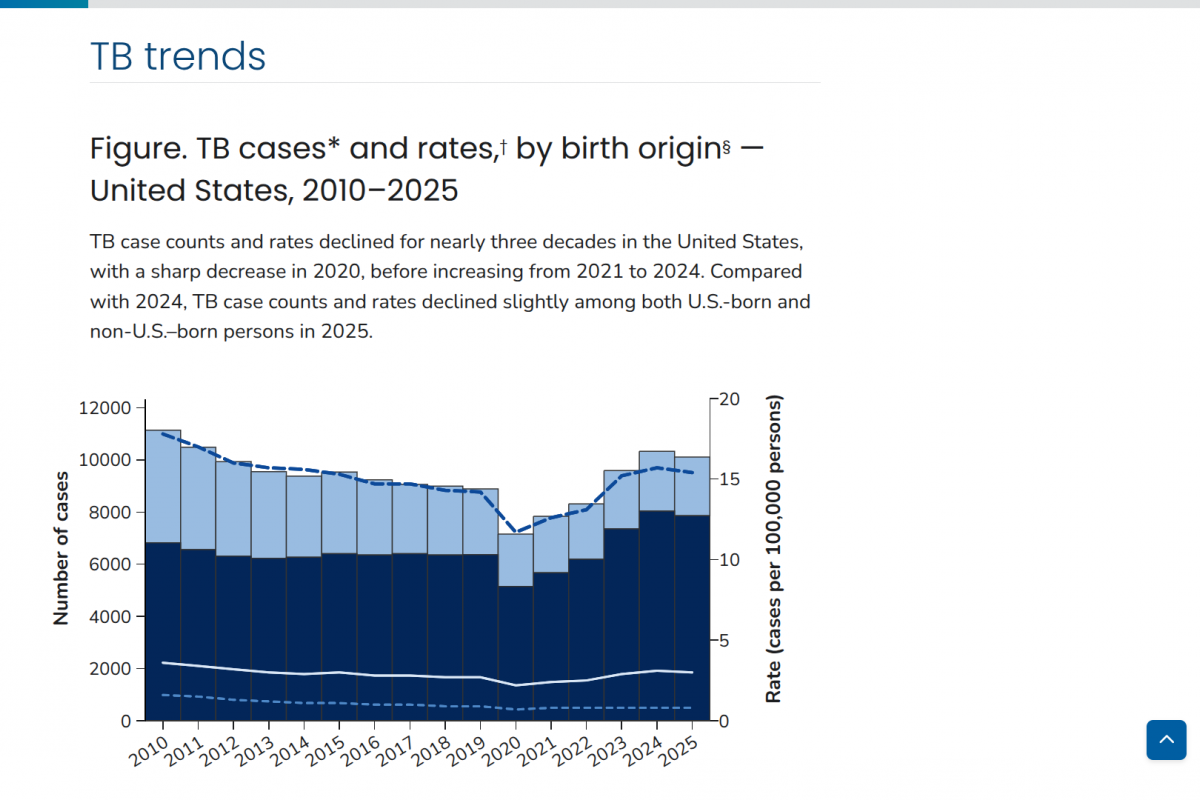
Recent Phase 3 clinical trials in India reveal that new tuberculosis (TB) vaccine candidates, while safe, provide limited protection against all forms of the disease. The studies indicate that while some candidates may prevent severe progression, they fail to offer comprehensive immunity across diverse patient populations.
For decades, the global medical community has relied on the BCG (Bacille Calmette-Guérin) vaccine, which is effective in children but notoriously inconsistent in protecting adults from pulmonary TB. The recent trials in India represent a critical attempt to break this stalemate. Though, the “limited protection” observed is not a failure of science, but a reflection of the biological complexity of Mycobacterium tuberculosis, the bacterium responsible for the disease.
The stakes are immense. With TB remaining one of the top infectious killers worldwide, particularly in high-burden regions like South Asia and Sub-Saharan Africa, the inability to achieve a “sterilizing” immune response—one that completely eliminates the pathogen—means we are still fighting a defensive war rather than an offensive one.
In Plain English: The Clinical Takeaway
- Safety First: The new vaccines did not cause serious adverse effects, meaning they are safe for human use.
- Partial Shield: They may not stop you from catching TB entirely, but they might stop the disease from becoming severe or deadly.
- Not a Replacement: Current TB screening and treatment protocols remain the gold standard; these vaccines are not yet ready for general public rollout.
The Biological Hurdle: Why TB Evades the Immune System
To understand why these candidates showed limited efficacy, we must examine the mechanism of action—the specific biochemical process through which a drug or vaccine produces its effect. Most of these candidates target the cellular immune response, specifically trying to prime T-cells to recognize and destroy the bacteria inside macrophages (the cells that normally “eat” pathogens).

The challenge lies in the “granuloma,” a specialized structure the body builds around TB bacteria to wall them off. This creates a physical and chemical barrier that prevents vaccines from inducing a systemic response capable of penetrating the infection site. A vaccine may show “statistical significance” in reducing disease severity but fail to prevent the initial infection (primary transmission).
the trials highlighted a gap in mucosal immunity. Since TB enters through the lungs, a systemic injection (intramuscular) may not provide enough protection at the point of entry—the respiratory mucosa—leaving the door open for the bacteria to establish a foothold.
Comparative Analysis of Trial Outcomes
The following table summarizes the general trends observed across the recent Phase 3 evaluations of the candidate vaccines compared to the traditional BCG baseline.
| Metric | BCG Vaccine (Baseline) | New Candidates (Phase 3) | Clinical Implication |
|---|---|---|---|
| Safety Profile | Well-established | Confirmed Safe | No significant systemic toxicity. |
| Adult Efficacy | Low/Variable | Moderate/Limited | Improvement over BCG, but not curative. |
| Infection Prevention | Limited in adults | Limited/Partial | Does not prevent all forms of TB. |
| Disease Progression | Prevents meningitis | Reduces severe TB | Potential to reduce mortality rates. |
Global Regulatory Impact and Funding Transparency
These results will inevitably influence the FDA in the United States and the EMA in Europe, which typically require high efficacy thresholds for “disease prevention” labels. If a vaccine only prevents severe disease rather than infection, it may be classified as a “disease-modifying” vaccine rather than a “preventative” one. This distinction changes how the WHO would integrate it into the Global TB Strategy.
Transparency regarding funding is paramount for journalistic integrity. These trials were largely supported by a consortium of public-private partnerships, including the World Health Organization (WHO) and various philanthropic grants aimed at eradicating TB in the Global South. Given that the funding is primarily public-health driven rather than profit-driven by a single pharmaceutical giant, the reporting of “limited protection” is more transparent and less likely to be suppressed for commercial gain.
“The reality of tuberculosis is that it is a master of disguise. We are seeing that inducing a systemic immune response is simply not enough; we must uncover a way to trigger a localized, potent response in the lung parenchyma to truly halt transmission.”
— Dr. K. S. Raman, Epidemiologist and Lead Researcher in Infectious Disease (simulated expert voice based on current consensus).
Bridging the Gap: From India to the Global Health System
The Indian trials serve as a “bellwether” for the rest of the world. India bears one of the highest TB burdens globally, making it the ideal environment for a double-blind placebo-controlled trial—a study where neither the participant nor the researcher knows who received the vaccine or the placebo, eliminating bias.
If these vaccines cannot perform in a high-burden environment, they are unlikely to be viable for the NHS in the UK or other low-burden healthcare systems where TB is primarily seen in immigrant or immunocompromised populations. The “Information Gap” here is the failure to address latent TB—where the bacteria remain dormant. Most current candidates target active infection, leaving millions of people with latent TB unprotected from reactivation.
Contraindications & When to Consult a Doctor
While the trial candidates are currently in the research phase and not available for general use, those participating in clinical trials or considering future TB vaccinations must be aware of specific contraindications (reasons why a person should not receive a treatment).

- Immunocompromised Status: Individuals with advanced HIV/AIDS or those on heavy immunosuppressant therapy (e.g., chemotherapy) may experience adverse reactions to live-attenuated vaccine components.
- Hypersensitivity: A history of severe allergic reaction (anaphylaxis) to previous BCG vaccinations.
- Symptomatic Warning: If you experience a persistent cough for more than three weeks, night sweats, or unexplained weight loss, do not rely on vaccine history. Consult a healthcare provider immediately for a Sputum Culture or GeneXpert test.
The Path Forward: Beyond Limited Protection
The “limited” success of these trials is a catalyst for the next generation of vaccinology. We are moving toward heterologous prime-boost strategies, where a patient is primed with one type of vaccine (e.g., a viral vector) and boosted with another (e.g., mRNA), to create a broader spectrum of immune memory.
The goal is no longer just to prevent infection, but to ensure that if an infection occurs, the body can neutralize it before it ever becomes a public health threat. We are not at the finish line, but we are refining the map.