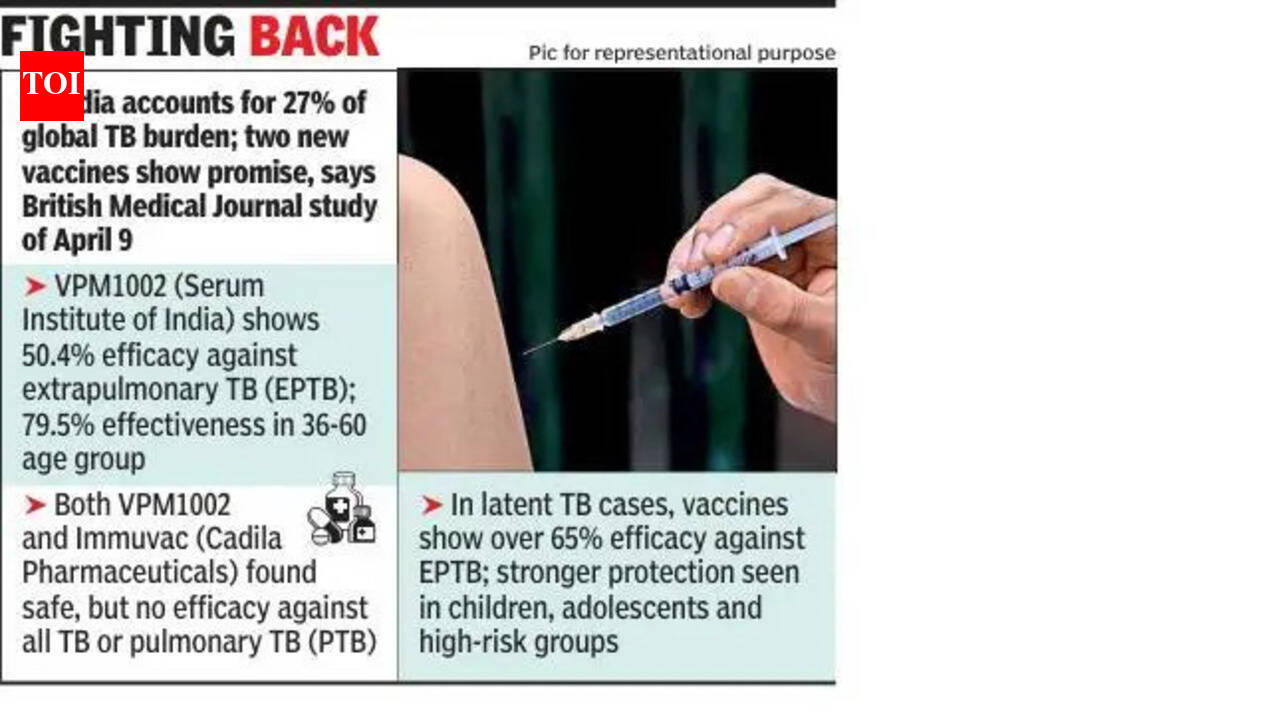
New Phase 3 trial results from India indicate that two novel tuberculosis (TB) vaccine candidates are safe and significantly reduce the risk of extrapulmonary TB, particularly in children. While overall protection against pulmonary TB remains limited, these results mark a critical step toward supplementing the century-vintage BCG vaccine.
For over a hundred years, the Bacillus Calmette-Guérin (BCG) vaccine has been the global standard for TB prevention. While it is remarkably effective at preventing severe miliary tuberculosis—a disseminated form of the disease—in infants, its efficacy in preventing the pulmonary (lung) form of TB in adults is notoriously inconsistent, ranging from 0% to 80% depending on the region. This “efficacy gap” is why TB remains one of the world’s deadliest infectious killers.
The recent data emerging from a massive trial involving nearly 13,000 participants represents a pivotal shift. We are no longer looking for a single “silver bullet” vaccine to replace BCG, but rather a secondary immunization—a booster or a complementary shot—that targets the specific pathways the bacteria use to hide in the body. By halving the risk of extrapulmonary TB, these candidates address a critical vulnerability in pediatric populations where TB often attacks the lymph nodes, bones, or central nervous system.
In Plain English: The Clinical Takeaway
- Not a BCG Replacement: These new vaccines are designed to work with the existing BCG shot, not replace it.
- Targeted Protection: They are particularly effective at stopping TB from spreading to organs outside the lungs (extrapulmonary TB), which is a major win for child health.
- Safety First: The Phase 3 trials confirm the vaccines are safe for human use, though they don’t yet stop all forms of lung infections.
The Cellular Shield: How New Antigens Target Extrapulmonary TB
To understand why these vaccines display promise, we must examine their mechanism of action—the specific biological process through which a drug or vaccine produces its effect. While BCG uses a weakened strain of bovine tuberculosis, these new candidates utilize specific antigens. Antigens are molecular “red flags” that train the immune system to recognize and attack a specific pathogen.

These vaccines aim to stimulate a robust T-cell response. T-cells are a type of white blood cell that acts as the “special forces” of the immune system, capable of hunting down and destroying cells already infected by Mycobacterium tuberculosis. By targeting antigens that are highly conserved across different strains of TB, the vaccines prime the immune system to intercept the bacteria before they can migrate from the lungs into the bloodstream and settle in other organs.
The reduction in extrapulmonary TB is statistically significant because this form of the disease is often harder to diagnose and treat. When TB moves beyond the lungs, it can cause meningitis or skeletal tuberculosis, which often lead to permanent disability in children. By suppressing this migration, the vaccines provide a critical layer of neuro-protection and systemic stability.
Analyzing the Efficacy Gap: Pulmonary vs. Extrapulmonary Outcomes
The trial data presents a nuanced reality: the vaccines are highly effective against extrapulmonary TB but show “limited protection” against pulmonary TB. In clinical terms, Which means the vaccines may not prevent the initial infection of the lungs, but they may prevent the disease from becoming systemic.
This distinction is vital. Pulmonary TB is the primary driver of transmission; if a patient cannot clear the bacteria from their lungs, they continue to shed the pathogen into the air. While these vaccines are a triumph for individual patient outcomes (especially for children), they may not yet be the tool that ends the global TB epidemic by stopping transmission.
| Feature | BCG Vaccine (Standard) | New Vaccine Candidates |
|---|---|---|
| Primary Goal | Prevent severe childhood TB | Supplement BCG / Prevent systemic spread |
| Pulmonary Efficacy | Highly variable in adults | Limited/Partial protection |
| Extrapulmonary Efficacy | Moderate | Significant reduction (approx. 50%) |
| Safety Profile | Established/Safe | Phase 3 Confirmed Safe |
Regulatory Hurdles and the Path to Global Distribution
The transition from a successful Phase 3 trial to a vial in a clinic involves rigorous regulatory scrutiny. In India, the Drugs Controller General of India (DCGI) oversees this process. Still, for these vaccines to reach the global south, they require WHO Prequalification. This is a service provided by the World Health Organization to ensure that medicines meet global standards of quality, safety, and efficacy.
The funding for these trials—often a collaboration between the Indian government, the Wellcome Trust, and the Bill & Melinda Gates Foundation—highlights the necessity of public-private partnerships. Because TB disproportionately affects low-income populations, there is little “market incentive” for traditional big pharma to invest. These vaccines are developed under “social impact” models to ensure affordability.
“The challenge with tuberculosis has always been the bacteria’s ability to evade the immune system by hiding inside macrophages. Any vaccine that can significantly reduce the systemic spread of the disease is a victory for public health, even if we are still searching for the perfect pulmonary shield.”
From a geo-epidemiological perspective, the impact will be felt most acutely in Southeast Asia and Africa. In these regions, the burden of childhood TB is highest. Integrating these vaccines into existing Expanded Programs on Immunization (EPI) would allow healthcare workers to provide a “dual-shield” approach: BCG at birth and the new candidate as a booster in early childhood.
Contraindications & When to Consult a Doctor
While the Phase 3 trials confirm a high safety profile, vaccines are not universal. Certain individuals must exercise caution:
- Severe Immunocompromise: Patients with advanced HIV/AIDS or those undergoing aggressive chemotherapy may not mount an effective immune response and could potentially react poorly to live-attenuated components.
- Hypersensitivity: Individuals with a known severe allergic reaction (anaphylaxis) to any component of the vaccine formulation should avoid it.
- Active TB Infection: Vaccines are preventative, not curative. If a patient is already symptomatic, they require a course of antibiotics (such as Rifampicin and Isoniazid), not a vaccine.
Consult a physician immediately if you or your child exhibit: A persistent cough lasting more than three weeks, unexplained weight loss, night sweats, or swelling in the lymph nodes of the neck.
The Road Ahead: Toward a Multivalent Strategy
The medical community is moving toward a “multivalent” strategy—using a cocktail of different vaccines to attack the bacteria from multiple angles. The success of these two candidates proves that we can target extrapulmonary TB. The next frontier is refining the mucosal immunity in the lungs to stop the infection at the point of entry.
We must remain objective: these vaccines are not a cure, nor are they a total replacement for the rigorous screening and treatment protocols currently in place. However, for a child in a high-burden area, a 50% reduction in the risk of TB attacking their brain or bones is not just a statistic—it is a life-altering clinical victory.