A next-generation yellow fever vaccine has demonstrated safety and efficacy comparable to the current gold-standard 17D vaccine in mid-stage clinical trials. This development aims to expand global vaccine production and improve accessibility in endemic regions of Africa and South America, where the viral hemorrhagic fever remains a significant threat.
For decades, the world has relied almost exclusively on the 17D vaccine—a live-attenuated virus (a weakened version of the virus that triggers an immune response without causing disease). Although highly effective, the 17D strain faces production bottlenecks and specific safety concerns for certain populations. The emergence of a comparable alternative is not merely a pharmaceutical redundancy; it is a strategic necessity for global health security. By diversifying the available vaccine candidates, we reduce the risk of supply chain collapses and provide a fallback should the primary strain face manufacturing hurdles.
In Plain English: The Clinical Takeaway
- Same Protection: The new vaccine works as well as the current shot at preventing yellow fever.
- Safety First: Early trial data shows no increase in severe side effects compared to the existing vaccine.
- Better Access: This new version may be easier to produce in large quantities, meaning more people in high-risk areas can receive protected.
The Mechanism of Action: Moving Beyond 17D
To understand why this new candidate is significant, we must examine the mechanism of action—the specific biochemical process through which a drug or vaccine produces its effect. The current 17D vaccine utilizes a live-attenuated virus that mimics a natural infection to “train” the immune system. This triggers the production of neutralizing antibodies and T-cell responses that recognize and destroy the yellow fever virus upon exposure.
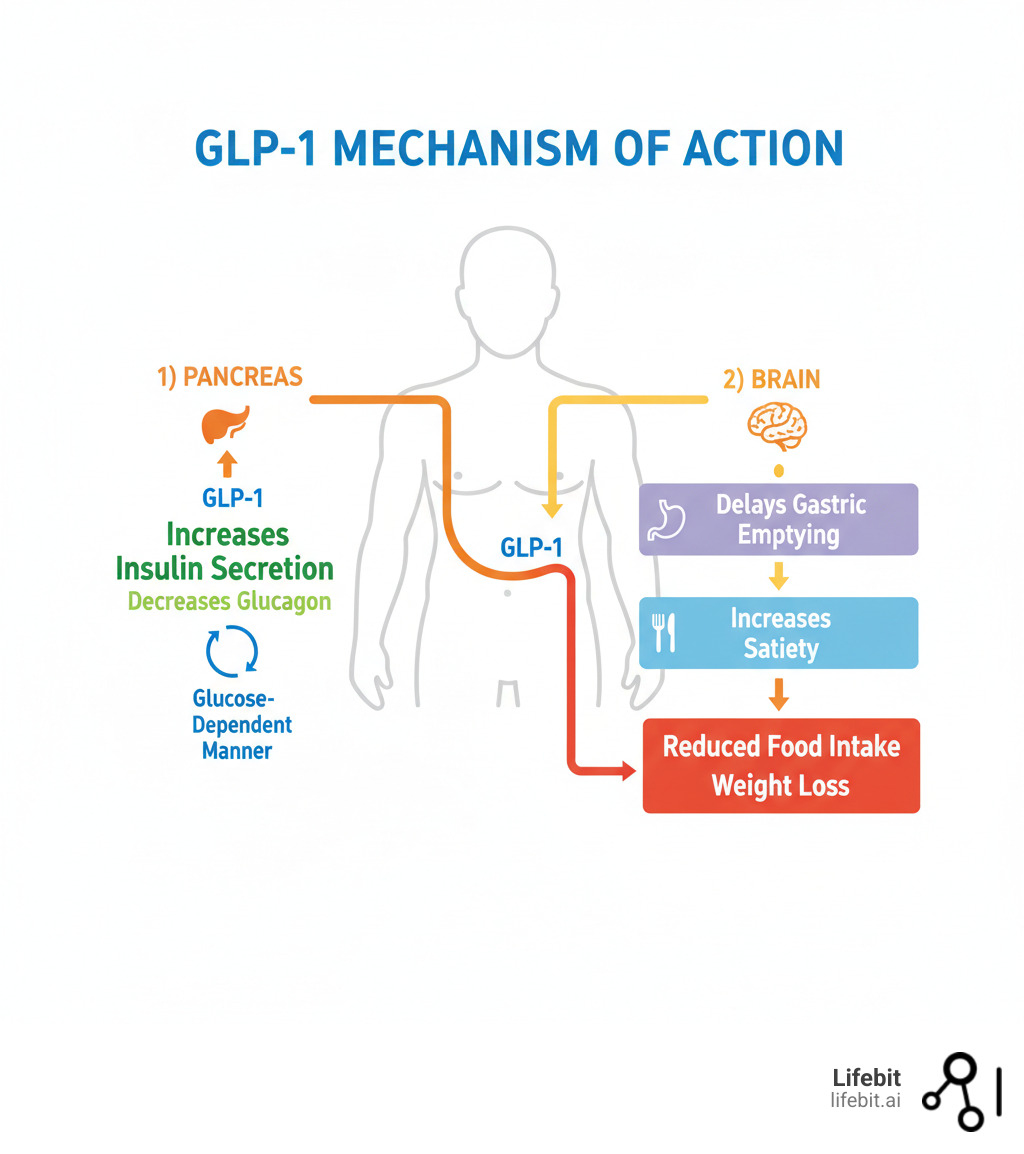
The next-generation candidate focuses on optimizing the stability of the viral antigen (the part of the virus the immune system recognizes). By refining the genetic sequence, researchers aim to maintain high immunogenicity—the ability to provoke an immune response—while potentially reducing the risk of vaccine-associated adverse events. This is particularly critical for individuals with compromised immune systems who cannot safely receive a live-attenuated vaccine.
The trials utilized a double-blind placebo-controlled design, which is the gold standard of clinical research. In this setup, neither the participants nor the researchers know who received the new vaccine or the control, eliminating observer bias and ensuring that the results are statistically significant rather than coincidental.
Comparing Clinical Efficacy and Trial Data
The mid-stage trials focused on “seroconversion” rates—the point at which a patient develops detectable antibodies in their blood. The data indicates that the new candidate matches the 17D vaccine in inducing a robust immune response. Below is a summary of the comparative performance based on available trial metrics.
| Metric | Current 17D Vaccine | Next-Gen Candidate | Clinical Significance |
|---|---|---|---|
| Efficacy (Seroconversion) | >99% | Comparable (High) | Equivalent protection levels |
| Safety Profile | Established / Rare AE | Comparable / Low AE | No new safety signals detected |
| Production Scalability | Limited/Specialized | Potentially Higher | Reduced global shortage risk |
| Trial Phase | Marketed/Approved | Mid-Stage (Phase II) | Requires Phase III confirmation |
while the “comparable efficacy” is promising, the vaccine must still undergo large-scale Phase III trials to confirm these results across diverse genetic populations. This process ensures that the vaccine remains effective across different ethnicities and age groups, particularly in the tropical regions where yellow fever is endemic.
Geo-Epidemiological Impact and Regulatory Pathways
The impact of this vaccine extends beyond the laboratory. In the United States, the FDA (Food and Drug Administration) manages vaccine approval, but yellow fever is primarily a concern for travelers. Still, for the CDC and the World Health Organization (WHO), the focus is on the “Yellow Fever Belt” in Sub-Saharan Africa and South America.
Current production is concentrated in a few facilities globally. If a manufacturing failure occurs at one of these sites, global supplies plummet. A new, scalable vaccine candidate allows for a decentralized manufacturing model. Which means countries in South America or Africa could potentially produce their own supplies, bypassing the logistical nightmares of cold-chain transport (the requirement to keep vaccines refrigerated from factory to patient).
Regarding funding and transparency, much of the research into next-generation flavivirus vaccines is supported by a mix of public-private partnerships, including the Coalition for Epidemic Preparedness Innovations (CEPI) and national health institutes. This funding structure is designed to prioritize public health over profit, ensuring that the resulting vaccines are affordable for low-income nations.
“The diversification of our vaccine portfolio against yellow fever is not just a scientific achievement, but a critical pillar of global health security. Reducing our reliance on a single strain ensures that we can respond to outbreaks with agility, and scale.”
Contraindications & When to Consult a Doctor
While the new vaccine is promising, it is not for everyone. Live-attenuated vaccines carry specific contraindications—medical reasons why a particular treatment should not be used because it may be harmful to the patient.

- Immunocompromised Individuals: People with HIV/AIDS, those undergoing chemotherapy, or patients on high-dose corticosteroids should avoid live vaccines as they may trigger a systemic infection.
- Severe Allergies: Anyone with a known severe allergy to eggs or gelatin should consult an allergist, as these components are often used in vaccine cultivation.
- Age Constraints: The vaccine is generally not recommended for infants under 6 months of age.
- Thymus Disorders: Individuals with a history of thymus gland issues (e.g., myasthenia gravis) are at higher risk for adverse reactions.
When to seek immediate care: If you experience symptoms of anaphylaxis (swelling of the throat, difficulty breathing) or signs of vaccine-associated neurologic events (severe headache, confusion, or focal weakness) following a vaccination, seek emergency medical attention immediately.
The Path Forward: From Trial to Triage
The transition from a mid-stage trial to global distribution is a rigorous journey. The next step involves large-scale efficacy trials to ensure the vaccine provides long-term immunity. We must remain objective: “comparable” does not mean “superior.” The goal here is not to replace a working vaccine, but to create a redundant, robust system of protection.
As we move toward 2027, the focus will shift toward regulatory harmonization between the EMA (European Medicines Agency) and the WHO to ensure that once approved, the vaccine can be deployed rapidly. For the global citizen, this means more reliable travel protections and, more importantly, a significant reduction in preventable deaths in the world’s most vulnerable regions.