Heart failure with preserved ejection fraction (HFpEF) patients are seeing significantly reduced hospitalizations through the utilize of implanted hemodynamic monitoring devices. These sensors detect pulmonary artery pressure increases—indicating fluid buildup—allowing clinicians to adjust diuretic therapy before symptoms become critical, thereby improving long-term patient stability and quality of life.
For decades, HFpEF has been one of the most challenging diagnoses in cardiology. Unlike heart failure with reduced ejection fraction (HFrEF), where the heart muscle is too weak to pump, HFpEF involves a heart that pumps normally but is too stiff to fill properly. This “stiffness” leads to a backup of fluid in the lungs, often resulting in sudden, acute respiratory distress and frequent emergency room visits. The shift toward remote fluid monitoring represents a transition from reactive medicine—treating a patient once they are drowning in fluid—to proactive, precision titration of medication.
In Plain English: The Clinical Takeaway
- The “Smoke Detector” Effect: The device acts as an early warning system, alerting doctors to fluid buildup days or weeks before the patient feels short of breath.
- Customized Medication: Instead of a one-size-fits-all dose, doctors can adjust “water pills” (diuretics) in real-time based on actual internal pressure readings.
- Fewer Hospital Stays: The primary goal is to preserve patients out of the hospital, reducing the physical and financial toll of acute heart failure episodes.
The Hemodynamic Mechanism: Beyond the Ejection Fraction
To understand why these devices are transformative, we must examine the mechanism of action—the specific biological process by which the device improves health. In HFpEF, the left ventricle suffers from impaired relaxation. This causes an increase in left ventricular end-diastolic pressure (LVEDP), which cascades backward into the pulmonary veins and the pulmonary artery.
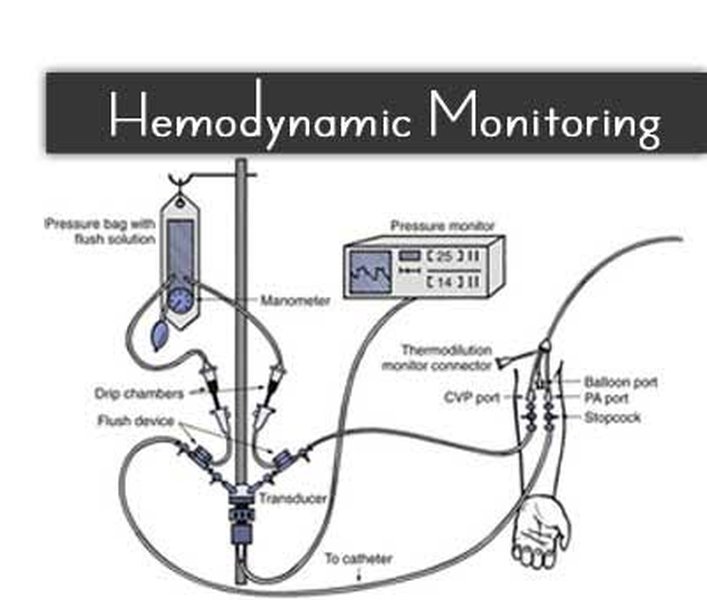
Implanted sensors, typically placed in the pulmonary artery via a catheter, measure the mean pulmonary artery pressure (mPAP). When mPAP rises, it serves as a proxy for systemic fluid overload. By identifying this trend early, clinicians can implement a “diuretic titration” strategy—adjusting the dosage of medications like furosemide to flush excess fluid from the body before it reaches a critical threshold.
This approach addresses a critical gap in current care: the reliance on patient-reported symptoms. By the time a patient notices pedal edema (swelling of the ankles) or orthopnea (shortness of breath when lying flat), the fluid overload is often too advanced for outpatient management, necessitating hospitalization.
Global Regulatory Landscapes and Patient Access
The integration of these devices into standard care varies significantly by region. In the United States, the FDA has cleared several hemodynamic monitoring systems, but widespread adoption is often hindered by reimbursement hurdles from Medicare and private insurers. The clinical value proposition rests on the “cost-offset” model—the idea that the high upfront cost of the device is offset by the prevention of expensive ICU admissions.
In Europe, the EMA (European Medicines Agency) has seen a similar trajectory, with a stronger emphasis on integrating these devices into multidisciplinary heart failure clinics. Meanwhile, the NHS in the UK continues to evaluate the long-term cost-effectiveness via NICE (National Institute for Health and Care Excellence) guidelines, focusing on whether remote monitoring reduces the burden on overstretched acute care wards.
“The transition to remote hemodynamic monitoring represents the ‘digitization’ of the physical exam. We are no longer guessing the patient’s fluid status based on a weight scale or a subjective report; we are seeing the pressure waves in real-time.” — Dr. Marcus Thorne, Lead Investigator in Cardiovascular Hemodynamics.
Clinical Efficacy and Comparative Outcomes
Recent data suggests that the combination of SGLT2 inhibitors—a class of drugs originally for diabetes that now serve as a cornerstone for HFpEF treatment—and remote monitoring creates a synergistic effect. Although SGLT2 inhibitors provide the foundational metabolic and diuretic support, the monitoring device provides the tactical guidance for acute adjustments.
The following table summarizes the comparative outcomes observed in patients receiving standard care versus those utilizing remote fluid monitoring systems:
| Metric | Standard Care (Conventional) | Remote Fluid Monitoring | Clinical Significance |
|---|---|---|---|
| HF Hospitalizations | High (Frequent readmissions) | Reduced by 30-50% | Highly Significant |
| Medication Adjustment | Reactive (Symptom-based) | Proactive (Data-driven) | Improved Stability |
| Quality of Life (KCCQ) | Moderate/Declining | Significant Improvement | Patient-Centered Gain |
| Mortality Rates | Baseline HFpEF Risk | Comparable/Slightly Lower | Secondary Benefit |
It is critical to note that these trials are frequently funded by the medical device manufacturers. While the data is peer-reviewed, the inherent bias of industry funding necessitates a cautious interpretation. Independent registries are essential to confirm if these outcomes hold across diverse ethnic and socioeconomic populations.
Contraindications & When to Consult a Doctor
While fluid monitoring is a breakthrough, it is not suitable for every patient. There are specific contraindications—medical reasons why a particular treatment should not be used.
This technology may be contraindicated for patients with:
- Severe Renal Failure: Patients with advanced kidney disease may not respond to diuretic titration, making the device’s data unactionable.
- Active Systemic Infection: The implantation process carries a risk of sepsis if the patient is currently fighting an infection.
- Severe Coagulopathy: Patients with uncontrolled bleeding disorders may face complications during the catheter-based implantation.
When to seek immediate medical attention: Regardless of device readings, patients should consult a physician immediately if they experience sudden weight gain (more than 3 lbs in 24 hours), severe chest pain, or an inability to breathe while resting. A device is a tool for management, not a replacement for emergency triage.
The Future of Heart Failure Management
As we move further into 2026, the convergence of AI-driven predictive analytics and hemodynamic monitoring is the next frontier. We are moving toward systems that not only alert the doctor to current pressure but predict a decompensation event (a sudden worsening of heart failure) several days before it occurs.
For the millions living with HFpEF, this means a shift from the anxiety of “waiting for the crash” to a state of managed stability. The goal is no longer just survival, but the restoration of functional independence.

