Reality television personalities Zack Wickham and Jasmine Goode’s recent public acknowledgement of using glucagon-like peptide-1 (GLP-1) receptor agonists, most notably Ozempic, has ignited a debate surrounding the drug’s use beyond its approved indications for type 2 diabetes. This discussion centers on the ethical implications of utilizing a medication intended for a serious metabolic disorder for weight loss, and the resulting societal stigma faced by those who legitimately require it.
The increasing visibility of GLP-1 agonists in mainstream media, fueled by celebrity endorsements and social media trends, obscures a complex clinical reality. While these drugs demonstrate significant efficacy in promoting weight loss and improving glycemic control, their broader impact on public health, potential long-term effects, and equitable access remain critical areas of investigation. The conversation extends beyond individual choices, impacting healthcare systems globally and raising questions about responsible prescribing practices.
In Plain English: The Clinical Takeaway
- Ozempic isn’t just for diabetes: Originally designed to assist people with type 2 diabetes control their blood sugar, it also helps people lose weight.
- Side effects are real: While effective, these medications can cause nausea, vomiting, and other digestive issues. More serious, though rare, risks exist.
- Talk to your doctor: Don’t self-treat. A healthcare professional can determine if a GLP-1 agonist is appropriate for *your* specific health needs.
The Mechanism Behind GLP-1 Agonists: More Than Just Weight Loss
GLP-1 receptor agonists, including Ozempic (semaglutide), Wegovy (also semaglutide, but at a higher dose), and others like liraglutide (Victoza, Saxenda), mimic the effects of the naturally occurring incretin hormone, GLP-1. This hormone is released by the gut in response to food intake and plays a crucial role in regulating blood glucose levels. The mechanism of action involves several key processes: stimulating insulin secretion, suppressing glucagon secretion (a hormone that raises blood sugar), slowing gastric emptying (making you perceive fuller for longer), and reducing appetite. These combined effects lead to both improved glycemic control and weight loss.
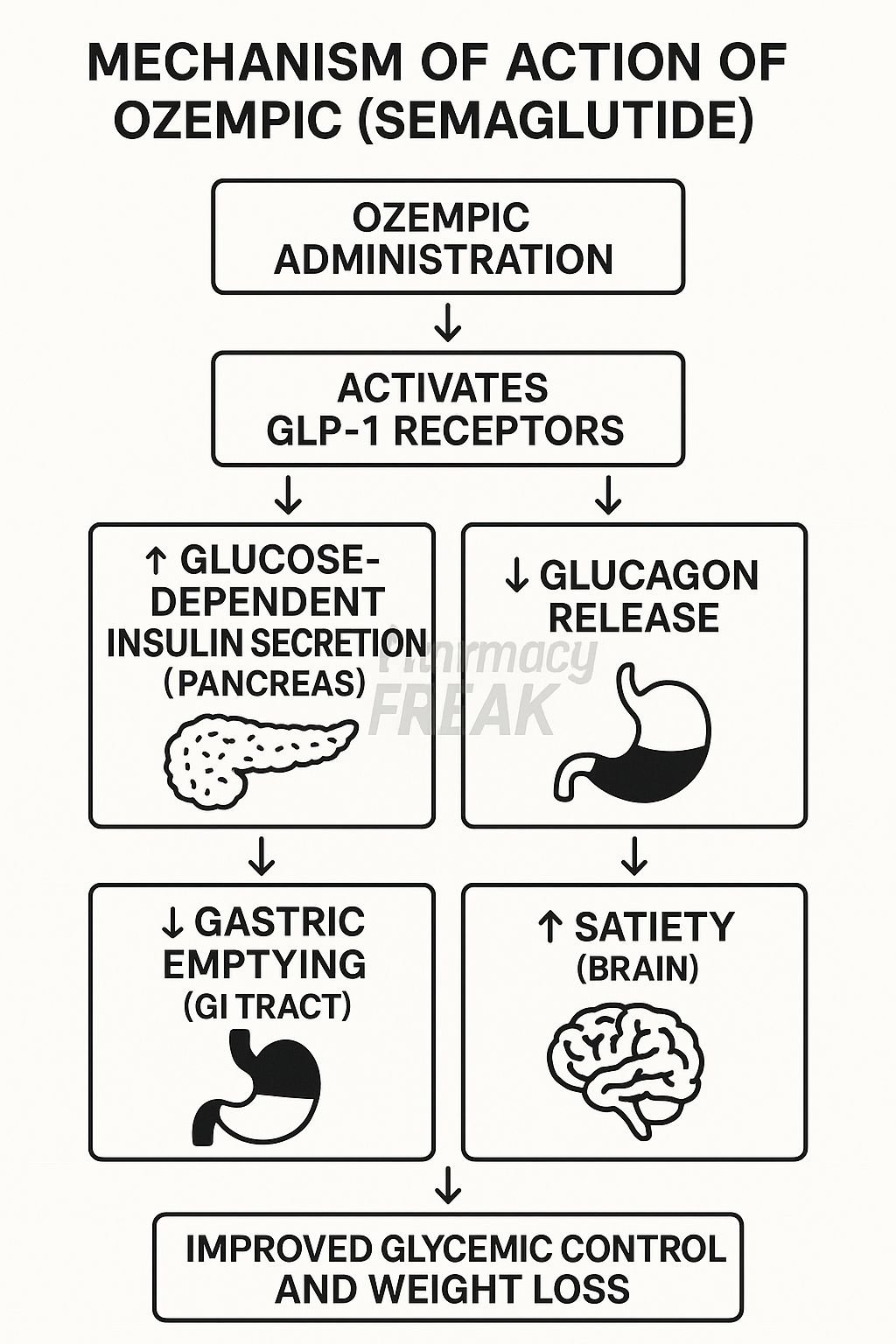
Clinical trials, such as the STEP 1 trial published in the New England Journal of Medicine in 2021, demonstrated that semaglutide 2.4 mg resulted in an average weight reduction of approximately 15% in individuals with obesity, but *without* type 2 diabetes. (NEJM, 2021). However, these benefits are not without risk. Common side effects include nausea, vomiting, diarrhea, and constipation. More concerning, though less frequent, are potential risks of pancreatitis, gallbladder disease, and, in pre-clinical studies, thyroid C-cell tumors in rodents – the clinical significance of which in humans is still under investigation.
Global Access and Regulatory Landscape: A Patchwork of Policies
The availability and regulation of GLP-1 agonists vary significantly across the globe. In the United States, the Food and Drug Administration (FDA) has approved semaglutide for both type 2 diabetes (Ozempic) and obesity (Wegovy). However, supply chain issues and high demand have led to intermittent shortages, impacting access for patients with diabetes. The European Medicines Agency (EMA) has similarly approved these medications, but national healthcare systems, like the National Health Service (NHS) in the United Kingdom, are grappling with cost-effectiveness considerations and establishing clear guidelines for prescribing.
The NHS, for example, initially restricted access to Wegovy to a limited number of specialist weight management services due to budgetary constraints. This highlights a critical issue: the affordability and equitable distribution of these potentially life-changing medications.
“The challenge isn’t just about the drugs themselves, but about building comprehensive weight management programs that integrate medication with lifestyle interventions – diet, exercise, and behavioral therapy. Simply prescribing a medication without addressing these underlying factors is unlikely to yield sustainable results.”
Dr. Emily Carter, PhD, Epidemiologist, University of Oxford
Funding, Bias, and the Pursuit of Objective Data
It’s crucial to acknowledge the funding sources behind the research on GLP-1 agonists. The majority of clinical trials are funded by pharmaceutical companies, such as Novo Nordisk (the manufacturer of Ozempic and Wegovy). While these companies adhere to rigorous regulatory standards, potential for bias always exists. Independent research, funded by governmental agencies like the National Institutes of Health (NIH) in the US, is essential to corroborate findings and provide a more comprehensive understanding of the long-term effects of these medications.
Transparency regarding funding sources is paramount. The ClinicalTrials.gov database provides information on trial sponsors, protocols, and results, allowing for greater scrutiny, and accountability.
Long-Term Effects and Ongoing Research
While short-term clinical trials have demonstrated the efficacy and safety of GLP-1 agonists, the long-term effects remain largely unknown. Ongoing longitudinal studies are investigating the potential impact on cardiovascular health, kidney function, and the risk of rare but serious adverse events. Researchers are also exploring the potential of GLP-1 agonists for other conditions, such as Alzheimer’s disease and non-alcoholic steatohepatitis (NASH).
| Drug | Indication | Average Weight Loss (%) | Common Side Effects | Serious Adverse Events (Reported Frequency) |
|---|---|---|---|---|
| Ozempic (Semaglutide) | Type 2 Diabetes | 5-10% | Nausea, Vomiting, Diarrhea | Pancreatitis (rare), Gallbladder Disease (rare) |
| Wegovy (Semaglutide) | Obesity | 15-18% | Nausea, Vomiting, Diarrhea | Pancreatitis (rare), Gallbladder Disease (rare) |
| Liraglutide (Saxenda) | Obesity | 8-10% | Nausea, Vomiting, Diarrhea | Pancreatitis (rare) |
Contraindications & When to Consult a Doctor
GLP-1 agonists are not suitable for everyone. Individuals with a personal or family history of medullary thyroid cancer, multiple endocrine neoplasia syndrome type 2 (MEN 2), or pancreatitis should avoid these medications. Pregnant or breastfeeding women should also not use GLP-1 agonists.
Consult a doctor immediately if you experience severe abdominal pain, persistent nausea or vomiting, signs of an allergic reaction (rash, itching, swelling), or any other concerning symptoms while taking a GLP-1 agonist. Self-treating with these medications is strongly discouraged.
The debate surrounding celebrity use of Ozempic and similar drugs underscores a broader societal challenge: the complex relationship between weight, health, and body image. While these medications can be valuable tools for managing obesity and type 2 diabetes, they are not a quick fix and should be used responsibly under the guidance of a healthcare professional. Future research and policy decisions must prioritize equitable access, long-term safety, and a holistic approach to health that addresses the underlying causes of obesity and metabolic disease.
References
- Frias, J. P., et al. “Semaglutide 2.4 mg Once Weekly versus Placebo in Adults with Overweight or Obesity.” New England Journal of Medicine 385.16 (2021): 1401-1412.
- American Diabetes Association. “Pharmacologic Approaches to Glycemic Control.” Diabetes Care 45.Supplement 1 (2022): S125-S143.
- EMA (European Medicines Agency). https://www.ema.europa.eu/
- FDA (Food and Drug Administration). https://www.fda.gov/
- National Institutes of Health (NIH). https://www.nih.gov/

