The public fascination with celebrity weight loss focuses on GLP-1 receptor agonists like Ozempic and Zepbound. While visual transformations spark speculation, these medications are clinical tools for treating type 2 diabetes and obesity, regulated by the FDA and EMA to manage metabolic dysfunction through hormonal regulation of appetite.
The discourse surrounding “celebrity transformations” is more than a tabloid curiosity; It’s a public health phenomenon. When high-profile individuals deny the use of GLP-1s while exhibiting rapid weight loss, it creates a dangerous “information gap” for the general public. This gap often leads to the off-label pursuit of powerful medications without medical supervision, masking the complex metabolic shifts and potential risks associated with these drugs.
In Plain English: The Clinical Takeaway
- Not a “Magic” Shot: These drugs mimic a hormone that tells your brain you’re full and slows down digestion.
- Medical Necessity: They are designed for people with chronic metabolic conditions, not for cosmetic weight loss.
- Long-term Commitment: Weight regain is common once the medication is stopped unless significant lifestyle changes are maintained.
The Molecular Mechanism: How GLP-1 Agonists Alter Metabolism
To understand why these drugs cause such visible changes, we must examine the mechanism of action—the specific biochemical process through which a drug produces its effect. Ozempic (semaglutide) and Mounjaro (tirzepatide) are GLP-1 receptor agonists. They mimic the glucagon-like peptide-1 hormone, which targets the hypothalamus in the brain to suppress appetite and delays gastric emptying (the speed at which food leaves the stomach).
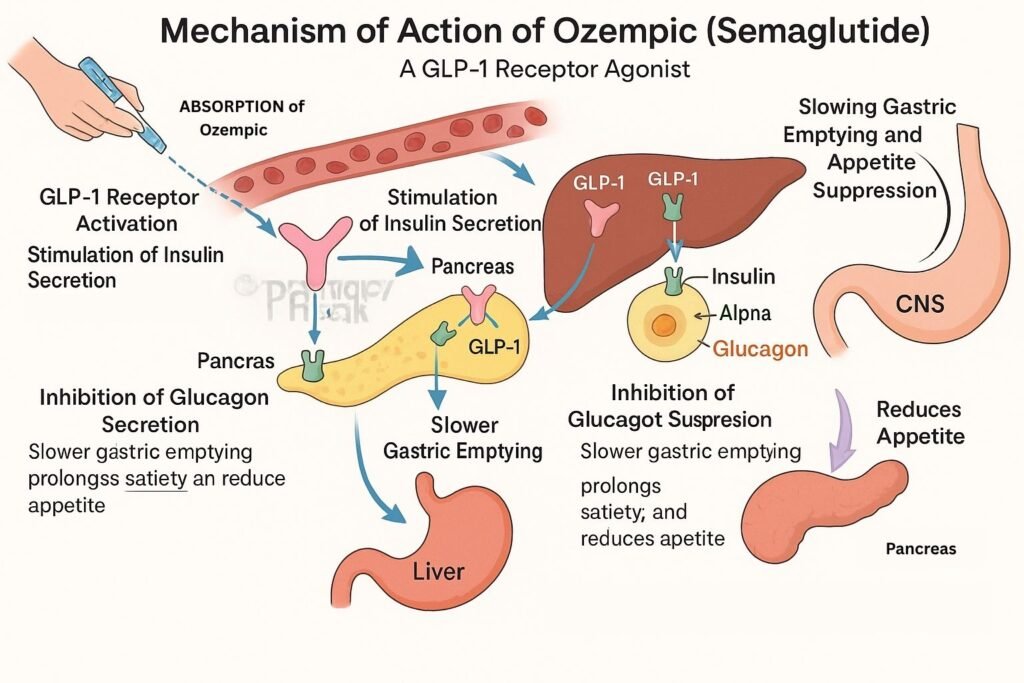
While semaglutide targets only the GLP-1 receptor, newer agents like tirzepatide are “dual agonists,” targeting both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. This synergy results in more significant reductions in adipose tissue (body fat) and improved glycemic control, which explains the more dramatic “before and after” transformations seen in recent months.
“The clinical efficacy of dual GLP-1/GIP agonism represents a paradigm shift in metabolic medicine, moving us closer to the weight loss percentages previously only achievable through bariatric surgery.” — Dr. Ania Hsia, Endocrinologist and Metabolic Specialist.
Global Access and the Regulatory Divide
The accessibility of these medications varies wildly by geography, creating a “medical divide.” In the United States, the FDA has approved several of these agents, but high costs and insurance hurdles often limit access to those with significant comorbidities. In contrast, the European Medicines Agency (EMA) maintains strict guidelines that prioritize the treatment of obesity as a chronic disease rather than a lifestyle choice.
In the UK, the NHS has implemented tiered access, ensuring that those with the highest BMI and most severe health risks receive priority. This systemic difference means that while a celebrity in Los Angeles may access these drugs via a “wellness clinic” with minimal oversight, a patient in London or Berlin must navigate a more rigorous clinical pathway based on evidence-based necessity.
Comparative Clinical Efficacy and Side Effects
It is critical to distinguish between the different molecules currently dominating the market. The following data is derived from Phase III clinical trials, including the SURMOUNT and STEP programs, which were funded primarily by the pharmaceutical manufacturers (Eli Lilly and Novo Nordisk). This funding transparency is essential, as industry-sponsored trials can sometimes emphasize efficacy over long-term adverse effects.
| Medication | Primary Target | Avg. Weight Loss (%) | Common Side Effects | Regulatory Status |
|---|---|---|---|---|
| Semaglutide (Ozempic/Wegovy) | GLP-1 | 10% – 15% | Nausea, Vomiting, Constipation | FDA/EMA Approved |
| Tirzepatide (Mounjaro/Zepbound) | GLP-1 & GIP | 15% – 22% | GI Distress, Diarrhea | FDA/EMA Approved |
| Liraglutide (Saxenda) | GLP-1 | 5% – 8% | Nausea, Low Blood Sugar | FDA/EMA Approved |
The Psychological Toll of the “Ozempic Face” Phenomenon
The “side-by-side” photos often highlight a hollowed appearance in the cheeks and jawline, colloquially termed “Ozempic Face.” Clinically, this is not a side effect of the drug itself, but a result of rapid adipose tissue depletion. When fat is lost rapidly from the subcutaneous layers of the face, the skin loses its structural support, leading to premature sagging and wrinkles.
This phenomenon underscores the danger of using these medications for cosmetic purposes. Rapid weight loss can lead to muscle wasting (sarcopenia) if not paired with high-protein nutrition and resistance training. The public’s obsession with the visual result ignores the metabolic cost of losing lean muscle mass alongside fat.
Contraindications & When to Consult a Doctor
These medications are not safe for everyone. You must consult a licensed physician if you have a history of medullary thyroid carcinoma (a specific type of thyroid cancer) or if you have a family history of this condition, as GLP-1s are strictly contraindicated in these cases.
Seek immediate medical attention if you experience:
- Severe, persistent abdominal pain (which may indicate pancreatitis).
- Frequent vomiting that leads to dehydration.
- Signs of gallbladder inflammation, such as right upper quadrant pain.
- Sudden changes in vision or severe hypoglycemia (low blood sugar), especially if taking insulin.
The Path Forward: Beyond the Trend
As we move through 2026, the medical community is shifting focus from “weight loss” to “metabolic health.” The goal is no longer just a lower number on the scale, but the improvement of cardiovascular outcomes and the reversal of fatty liver disease. The celebrity trend has brought visibility to these drugs, but it has also obscured the necessity of a comprehensive, multidisciplinary approach to health.
True wellness cannot be injected. While GLP-1s are revolutionary tools for those with clinical needs, they are supplements to—not replacements for—nutritional literacy and physical activity. The future of obesity treatment lies in personalized medicine, where genetics, lifestyle, and pharmacology intersect to treat the patient, not the image.
References
- PubMed: National Library of Medicine (Clinical Trials on GLP-1 Agonists)
- The Lancet: Global Obesity and Metabolic Health Studies
- JAMA: Comparative Efficacy of Tirzepatide vs Semaglutide
- World Health Organization: Guidelines on Obesity Management
- Centers for Disease Control and Prevention (CDC): Adult Obesity Statistics

