Canadian researchers are advancing precision oncology using biocompatible nanoparticles to target tumors directly. This approach minimizes systemic toxicity compared to traditional chemotherapy. Published this week, the findings suggest a shift toward guided therapeutic delivery systems. Patients may soon access treatments that spare healthy tissue while maximizing tumor eradication rates.
The evolution from cytotoxic scattershot approaches to ligand-targeted delivery represents a paradigm shift in oncology. Traditional chemotherapy relies on systemic distribution, affecting rapidly dividing cells indiscriminately. In contrast, the latest methodology utilizes surface-modified particles that recognize specific antigens on cancer cell membranes. This specificity reduces off-target effects, potentially lowering the incidence of severe adverse events like neutropenia and alopecia. For patients in Northern Ontario and beyond, this research signals a future where treatment tolerance improves significantly.
In Plain English: The Clinical Takeaway
- Targeted Delivery: Medicine travels directly to the cancer, sparing healthy organs.
- Reduced Side Effects: Patients may experience less nausea and hair loss than with standard chemo.
- Current Status: This technology is moving through clinical trials and is not yet standard care.
Mechanism of Action: Navigating the Vascular System
The core innovation lies in the pharmacokinetics of nanoparticle albumin-bound structures. These biocompatible particles are engineered to evade the immune system’s reticuloendothelial system, allowing prolonged circulation time. Once in the bloodstream, the particles exploit the enhanced permeability and retention effect inherent in tumor vasculature. Essentially, leaky blood vessels around tumors allow these particles to accumulate locally.
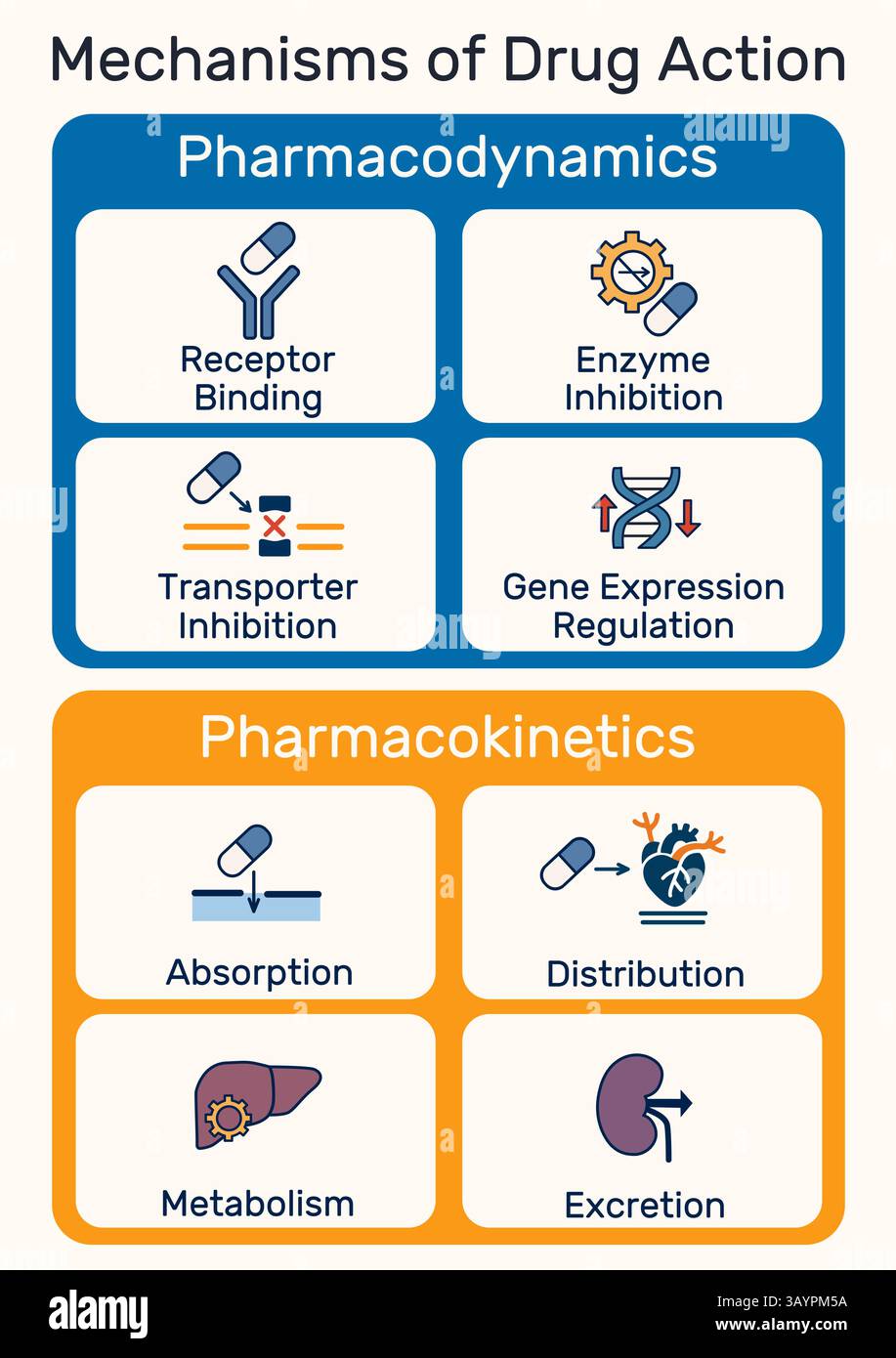
Upon reaching the target site, the particles release their payload through pH-dependent mechanisms or enzymatic triggers unique to the tumor microenvironment. This ensures the mechanism of action is localized. By concentrating the therapeutic agent within the malignancy, clinicians can potentially escalate dosages without increasing systemic toxicity. This precision is critical for treating aggressive carcinomas where previous limits were defined by patient tolerance rather than drug efficacy.
Regulatory Pathways and Patient Access in North America
While the research originates in Canada, regulatory approval requires harmonization across borders. Health Canada oversees domestic safety, but many patients look to the U.S. Food and Drug Administration (FDA) for broader access. The regulatory hurdle for nanomedicine is significant; agencies require rigorous data on long-term particle clearance to prevent accumulation in the liver or kidneys.
For patients in the Sudbury region, access depends on trial enrollment or compassionate use programs until formal approval is granted. The European Medicines Agency (EMA) similarly evaluates these therapies under strict advanced therapy medicinal product regulations. This geo-epidemiological bridging means that even successful Canadian trials may take 12 to 18 months to translate into available prescriptions in local pharmacies. Insurance coverage also remains a variable, often requiring proof of cost-effectiveness compared to existing generics.
| Feature | Traditional Chemotherapy | Targeted Nanoparticle Therapy |
|---|---|---|
| Distribution | Systemic (Whole Body) | Localized (Tumor Site) |
| Primary Side Effect | Bone Marrow Suppression | Injection Site Reaction |
| Clearance | Renal/Hepatic | Reticuloendothelial System |
| Precision | Low (Cell Division Based) | High (Antigen Based) |
Funding Transparency and Research Integrity
Trust in medical innovation requires clear disclosure of financial interests. Much of the foundational work in Canadian nanomedicine is supported by the Canadian Institutes of Health Research (CIHR) alongside private biotechnology partnerships. It is vital for patients to understand that while academic institutions drive the science, commercial partners often hold the licensing rights for production.
Transparency regarding conflicts of interest ensures that clinical trial data remains unbiased. Independent verification of results is standard practice in peer-reviewed publications. Patients should look for studies published in high-impact journals where funding sources are explicitly declared in the disclosure section. This scrutiny protects against the promotion of unproven therapies driven solely by profit motives rather than patient outcomes.
“Precision medicine is not just about the drug; it is about the right drug for the right patient at the right time. We are moving toward a future where toxicity is no longer the price of survival.” — National Cancer Institute, Precision Medicine Initiative
This statement underscores the global commitment to reducing the burden of treatment. As we progress through 2026, the integration of genomic profiling with these delivery systems will further refine patient selection. Only those with specific molecular markers will receive these therapies, ensuring resources are allocated where efficacy is statistically probable.
Contraindications & When to Consult a Doctor
Despite the promise of targeted therapy, contraindications exist. Patients with severe hepatic impairment may struggle to clear the nanoparticle carriers, leading to potential toxicity. Those with known hypersensitivity to albumin or specific ligand coatings should avoid these trials. It is crucial to discuss your full medical history with an oncologist before considering experimental protocols.
Seek immediate medical intervention if you experience signs of an infusion reaction, such as difficulty breathing, swelling of the face, or severe rash. While these particles are biocompatible, immune responses can still occur. Do not discontinue standard care in favor of unproven treatments without explicit guidance from your care team. Always verify that any new therapy is part of a registered clinical trial.

