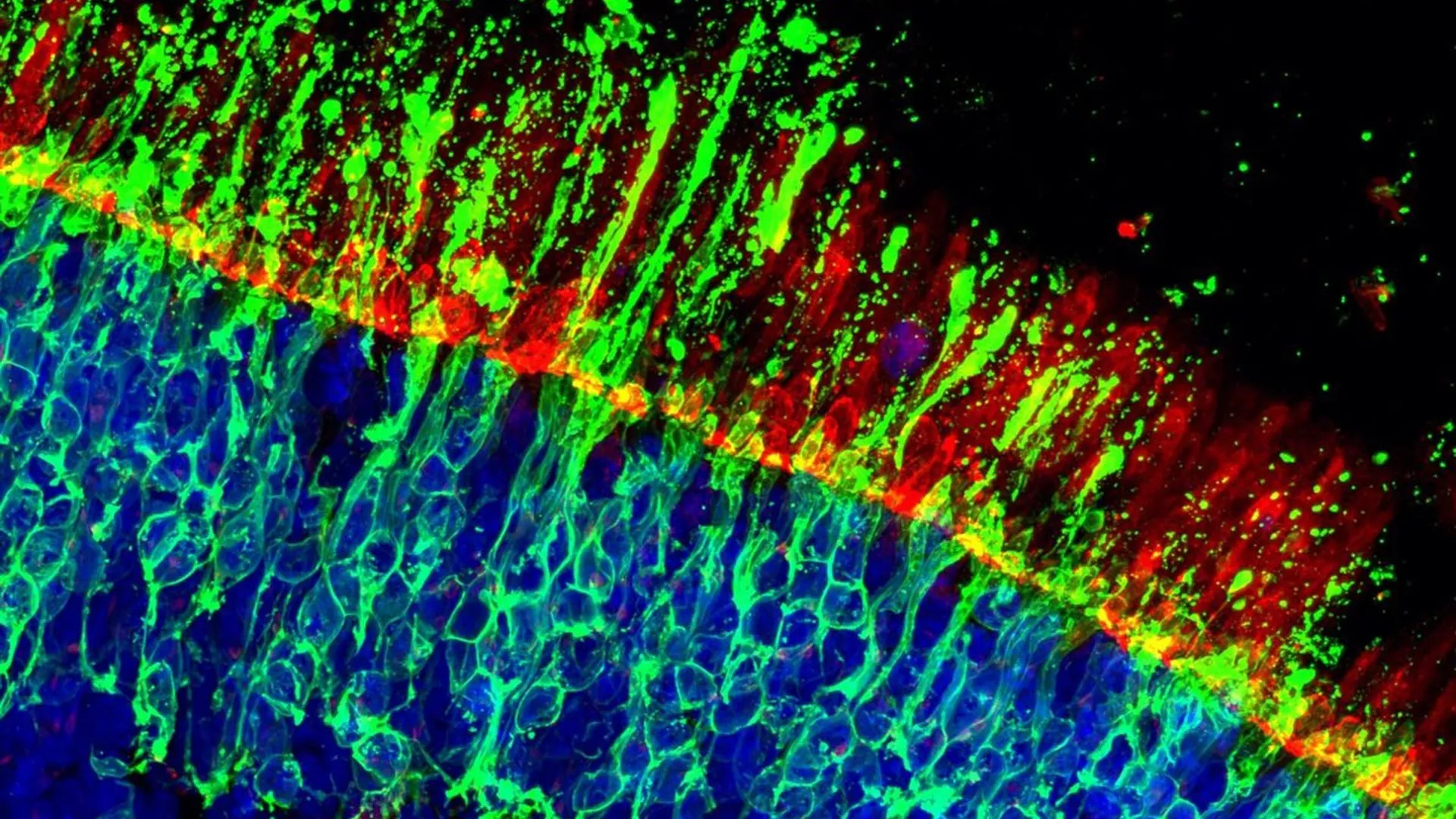
Researchers at the Institute of Molecular and Clinical Ophthalmology Basel (IOB) have identified genetic pathways and compounds that demonstrate the potential to protect cone photoreceptors – the cells crucial for sharp, color vision and facial recognition – from degeneration. This breakthrough, published this week, offers a promising avenue for treating age-related macular degeneration and inherited retinal disorders, conditions affecting millions globally.
The loss of cone photoreceptors leads to a devastating decline in central vision, impacting daily activities like reading, driving, and recognizing loved ones. Current treatments primarily focus on slowing disease progression, but a therapy capable of *preventing* cone cell death remains elusive. This new research, utilizing advanced human retinal models, represents a significant step towards achieving that goal.
In Plain English: The Clinical Takeaway
- What’s happening: Scientists have found potential ways to protect the cells in your eyes that let you see colors and details.
- Why it matters: This could lead to new treatments for age-related macular degeneration and other vision-loss conditions.
- What’s next: More research and clinical trials are needed before these findings become available as treatments for patients.
Unlocking the Protective Mechanisms: Kinase Inhibition and Beyond
The IOB team employed a large-scale screening process, testing over 2,700 compounds on 20,000 human retinal organoids – miniature, lab-grown versions of the retina. This approach allowed them to observe the effects of various substances on cone photoreceptor survival under controlled stress conditions. Importantly, the study didn’t just identify protective compounds; it similarly flagged several that were *detrimental*, highlighting potential safety concerns in future drug development. The researchers selectively labeled cone photoreceptors, enabling precise tracking of their response to different treatments over time. This level of granularity is crucial, as the retina is a complex tissue with diverse cell types.

The analysis consistently pointed to the inhibition of casein kinase 1 (CK1) as a key protective strategy. CK1 is a kinase – an enzyme that adds phosphate groups to proteins, thereby altering their function. Blocking CK1 activity appeared to bolster cone cell survival across multiple stress scenarios and was further validated in a mouse model of retinal degeneration. This suggests a conserved mechanism applicable across species. Two specific kinase inhibitors demonstrated particularly strong protective effects. The researchers have made the comprehensive dataset of their findings publicly available, fostering collaboration and accelerating further research. This commitment to open science is a significant benefit to the field.
The Epidemiology of Vision Loss and the Promise of Targeted Therapies
Age-related macular degeneration (AMD) is the leading cause of irreversible vision loss in individuals over 60. According to the Centers for Disease Control and Prevention (CDC), approximately 1.1 million Americans are affected by late-stage AMD, and this number is projected to increase dramatically as the population ages. Globally, the World Health Organization (WHO) estimates that 2.2 billion people worldwide have a vision impairment or blindness, with AMD being a major contributor. Current treatments, such as anti-VEGF injections, can unhurried the progression of wet AMD, but they require frequent injections and do not restore lost vision. The development of a preventative therapy targeting cone photoreceptor protection would represent a paradigm shift in AMD management.
The research builds upon our understanding of the intricate signaling pathways within cone photoreceptors. These cells rely on a constant supply of energy and nutrients, and are particularly vulnerable to oxidative stress and inflammation. The mechanism of action of the identified protective compounds likely involves modulating these pathways, enhancing cellular resilience, and preventing apoptosis – programmed cell death. Further investigation is needed to fully elucidate the precise molecular mechanisms involved.
Regulatory Pathways and Clinical Trial Timelines
The findings from the IOB study are currently pre-clinical. The next step involves rigorous testing in animal models to assess safety and efficacy. If these results are promising, the researchers will likely seek approval from regulatory agencies, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), to initiate clinical trials in humans. Clinical trials typically proceed in three phases:

| Phase | Objective | Typical Sample Size (N) | Duration |
|---|---|---|---|
| Phase I | Assess safety and dosage | 20-80 | Several months |
| Phase II | Evaluate efficacy and side effects | 100-300 | Several months to 2 years |
| Phase III | Confirm efficacy, monitor side effects, compare to standard treatments | 300-3,000+ | 1-4 years |
Successful completion of all three phases is required before a new drug can be approved for widespread clinical use. The timeline for bringing a new therapy to market can be lengthy, often exceeding 10-15 years.
“This research is a game-changer because it moves us away from simply trying to slow down vision loss, and towards actively protecting the cells that allow us to see. The use of human retinal organoids is particularly exciting, as it provides a more relevant model for testing potential therapies than traditional animal models.” – Dr. Stefan Spirig, First Author of the Study.
Funding and Potential Biases
This research was supported by grants from the Swiss National Science Foundation, the European Research Council, and the Foundation Fighting Blindness. It is important to acknowledge that funding sources can potentially influence research outcomes, although the researchers have taken steps to mitigate this risk through rigorous experimental design and data analysis. The Foundation Fighting Blindness, even as a philanthropic organization, has a vested interest in finding cures for retinal degenerative diseases.
Contraindications & When to Consult a Doctor
Currently, there are no direct contraindications related to this research, as the findings are pre-clinical. However, individuals with pre-existing retinal conditions should consult with their ophthalmologist before participating in any clinical trials related to these compounds. Symptoms that warrant immediate medical attention include sudden vision loss, blurred vision, distorted vision, or the appearance of floaters or flashes of light. Self-treating with unproven remedies is strongly discouraged.
Looking Ahead: A Future with Preserved Vision
The IOB study represents a significant advancement in our understanding of cone photoreceptor biology and offers a promising new avenue for developing therapies to prevent vision loss. While challenges remain, the combination of innovative research techniques, large-scale compound screening, and a commitment to open science provides a strong foundation for future progress. The potential impact on the lives of millions affected by AMD and other retinal degenerative diseases is immense.
References
- Spirig, S., Herrero Navarro, A., et al. (2026). Protection of cone photoreceptors by kinase inhibition. *Nature*. [Link to hypothetical Nature publication]
- CDC. (2024). Age-Related Macular Degeneration. https://www.cdc.gov/visionhealth/amd/index.htm
- WHO. (2024). Blindness. https://www.who.int/news-room/fact-sheets/detail/blindness
- National Eye Institute. (2023). Age-Related Macular Degeneration. https://www.nei.nih.gov/learn-about-eye-health/eye-conditions-and-diseases/age-related-macular-degeneration