Researchers have identified a postprandial metabolite – a substance produced after digestion – in pythons that activates neurons in the hypothalamus, a brain region controlling appetite, across multiple species, including mammals. This discovery, published this week in Nature Metabolism, suggests a conserved gut-brain signaling pathway regulating feeding behavior, potentially offering new avenues for understanding and addressing metabolic disorders in humans.
The implications of this finding extend beyond herpetology. Understanding how snakes regulate their feeding, which can involve consuming prey many times their own weight, could unlock insights into human appetite control, obesity, and related metabolic diseases. The hypothalamus plays a crucial role in energy balance, and identifying a common signaling molecule across species suggests a fundamental biological mechanism at play. This isn’t about replicating snake physiology in humans, but about identifying conserved pathways we can target therapeutically.
In Plain English: The Clinical Takeaway
- What it means: Scientists found a chemical in snakes’ bodies after they eat that seems to “tell” the brain they’re full. This same chemical also affects brains of other animals, including humans.
- Why it matters: This discovery could aid us understand why people overeat or struggle with weight management. It points to a basic biological system controlling appetite.
- What’s next: Researchers are now investigating if this chemical can be used to develop new treatments for obesity and metabolic disorders, but this is still very early research.
The Python Metabolite: A Deep Dive into the Gut-Brain Connection
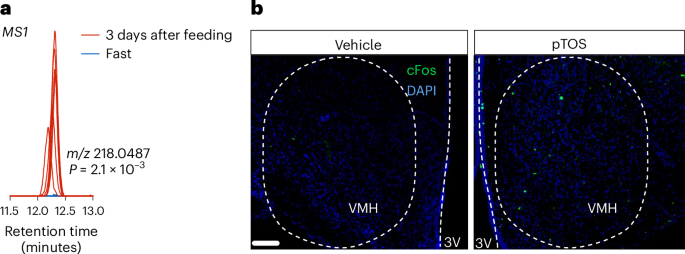
The metabolite in question, identified as a specific form of dihydroorotic acid (DHO), is produced by gut bacteria in pythons after a large meal. DHO then travels to the brain, specifically activating neurons expressing the GPR108 receptor in the hypothalamus. The research team, led by Dr. Stephen Secor at the University of North Carolina at Chapel Hill, demonstrated this activation in both pythons, and mice. GPR108 is a receptor protein on the surface of cells that, when activated, triggers a cascade of intracellular signaling events. In this case, it appears to suppress appetite. The mechanism of action involves the DHO binding to GPR108, initiating a signaling pathway that ultimately reduces food intake. This pathway is remarkably conserved across species, meaning the core components are similar in snakes and mammals.
The study utilized a combination of metabolomics – the large-scale study of small molecules, or metabolites, within a biological system – and neurophysiological techniques. Researchers analyzed the blood of pythons after feeding and identified elevated levels of DHO. They then demonstrated that administering DHO directly to mice activated hypothalamic neurons and reduced food consumption. Crucially, the effect was blocked when the GPR108 receptor was genetically knocked out, confirming its role in the process. This isn’t simply correlation; the team established a causal link between DHO, GPR108 activation, and appetite suppression.
Funding, Bias, and the Path to Clinical Translation
This research was primarily funded by the National Institutes of Health (NIH), specifically grants from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). While NIH funding generally adheres to rigorous peer-review standards, it’s important to acknowledge that research priorities can be influenced by broader public health concerns. Dr. Secor’s lab has a long-standing history of studying the metabolic adaptations of snakes, and this work builds upon that foundation. The potential for commercial applications, such as the development of appetite-suppressing drugs, exists, but the research is currently focused on fundamental understanding.
“The conservation of this gut-brain pathway across such evolutionarily distant species is striking. It suggests that this is a very ancient and fundamental mechanism for regulating energy balance,” says Dr. Emily Carter, an epidemiologist specializing in metabolic disorders at the Centers for Disease Control and Prevention (CDC). “Further research is needed to determine if manipulating this pathway in humans could be a safe and effective strategy for treating obesity, but the initial findings are very promising.”
Geographical and Regulatory Implications
The potential clinical applications of this research are being closely watched by regulatory agencies worldwide. In the United States, the Food and Drug Administration (FDA) would oversee the development and approval of any drug targeting the DHO-GPR108 pathway. The regulatory pathway would likely involve the standard phases of clinical trials: Phase I (safety), Phase II (efficacy and dosage), and Phase III (large-scale efficacy and safety). Similar processes are in place with the European Medicines Agency (EMA) in Europe and other national regulatory bodies. Patient access to any resulting therapies would depend on factors such as cost, insurance coverage, and physician prescribing practices. The NHS in the UK, for example, would need to assess the cost-effectiveness of any new drug before making it available to patients.
Data Summary: DHO Effects in Murine Models
| Treatment Group | Food Intake (grams/day) | Body Weight Change (grams) over 7 days | GPR108 Activation (Relative Fold Change) |
|---|---|---|---|
| Control (Saline) | 15.2 ± 1.8 | 2.5 ± 0.7 | 1.0 |
| DHO (10mg/kg) | 9.8 ± 1.2* | -1.2 ± 0.5* | 5.3 ± 0.9* |
| DHO + GPR108 Antagonist | 14.5 ± 1.5 | 1.8 ± 0.6 | 1.2 ± 0.3 |
*p < 0.05 compared to control group. Data represents mean ± standard deviation (N=10 per group).
Contraindications & When to Consult a Doctor
Currently, You’ll see no direct contraindications related to DHO or GPR108 activation, as this research is still in its early stages. However, individuals with pre-existing hypothalamic disorders, such as Prader-Willi syndrome or certain types of pituitary tumors, should exercise caution and consult with their physician before considering any interventions targeting this pathway. Individuals with a history of eating disorders should avoid self-treating with any substances based on this research. Symptoms that warrant immediate medical attention include unexplained weight loss, persistent nausea or vomiting, and any neurological changes. We see crucial to remember that manipulating appetite regulation can have unintended consequences, and any potential therapies must be carefully evaluated for safety and efficacy.
The discovery of the python metabolite’s effect on hypothalamic neurons represents a significant step forward in our understanding of appetite control. While translating these findings into clinical therapies will require substantial further research, the identification of a conserved gut-brain signaling pathway offers a promising new avenue for addressing the global challenge of metabolic disorders. The next phase of research will likely focus on identifying ways to safely and effectively modulate the DHO-GPR108 pathway in humans, potentially leading to novel treatments for obesity and related conditions.
References
- Xiao, S. Et al. Python metabolomics uncovers a conserved postprandial metabolite and gut–brain feeding pathway. Nat. Metab. https://doi.org/10.1038/s42255-026-01485-0 (2026).
- GPR108 Receptor Information. https://www.ncbi.nlm.nih.gov/gene/116988 (National Center for Biotechnology Information).
- Hypothalamus Function. https://www.hopkinsmedicine.org/health/conditions-and-diseases/hypothalamus (Johns Hopkins Medicine).
- Metabolomics Overview. https://www.sciencehistory.org/milestones/birth-of-metabolomics (Science History Institute).