Rare Genetic Mutation Identified as Leading Cause of Neurodevelopmental Disorder
Researchers have pinpointed biallelic variants in the RNU2-2 gene as the most common known genetic cause of a recessive neurodevelopmental disorder. This discovery, stemming from analysis of large genomic datasets in the UK and replicated internationally, offers a crucial diagnostic marker and opens avenues for understanding the underlying mechanisms of this debilitating condition affecting infants and children. The findings were published this week in Nature Medicine.
In Plain English: The Clinical Takeaway
- What it is: A genetic condition causing developmental delays, seizures, and intellectual disability, often appearing in infancy.
- How it’s found: Genetic testing can now specifically seem for changes in the RNU2-2 gene.
- What’s next: While there’s no cure yet, knowing the cause helps doctors provide better care and potentially develop future treatments.
Unraveling the Genetic Basis of RNU2-2 Syndrome
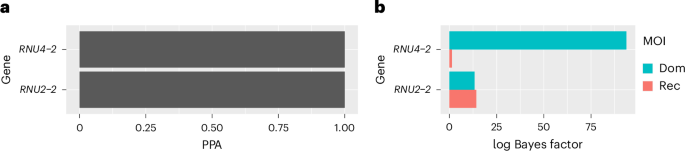
The investigation, leveraging data from the 100,000 Genomes Project and the Genomic Medicine Service in the UK’s National Genomic Research Library, revealed a significantly higher prevalence of RNU2-2 variants in individuals with rare neurodevelopmental disorders (NDDs) than previously recognized. The study employed a genetic association method called BeviMed to identify the gene, confirming its role through replication studies in the United States, Italy, and the Netherlands. The research indicates that approximately 7.6% to 13.1% of individuals with a genetically diagnosed NDD carry biallelic variants in RNU2-2, making it a major contributor to the landscape of recessive NDDs.
Mechanism of Action: Disrupting Spliceosome Function
The RNU2-2 gene encodes a small nuclear RNA (snRNA) molecule crucial for proper RNA splicing – a fundamental process in gene expression. Specifically, U2 snRNAs interact with pre-mRNA introns, guiding the removal of non-coding regions to create functional proteins. Variants in RNU2-2, particularly those disrupting intramolecular interactions within the snRNA structure, appear to destabilize the spliceosome, the molecular machinery responsible for splicing. This disruption leads to aberrant RNA processing and ultimately impacts neuronal development and function. Interestingly, the study found that individuals with RNU2-2 variants often exhibit compensatory upregulation of U2-1, another snRNA gene, suggesting a cellular attempt to mitigate the effects of RNU2-2 deficiency. This compensatory mechanism, however, appears insufficient to fully restore normal splicing function.
Geographical Impact and Diagnostic Accessibility
The identification of RNU2-2 as a major cause of NDDs has significant implications for healthcare systems globally. In the United States, the Food and Drug Administration (FDA) has not yet approved specific diagnostic tests solely for RNU2-2 variants, but clinical genetic testing laboratories are increasingly incorporating this gene into comprehensive NDD panels. The National Institutes of Health (NIH) is funding several research initiatives focused on understanding the molecular mechanisms of RNU2-2-related disorders and exploring potential therapeutic strategies. Similarly, in Europe, the European Medicines Agency (EMA) is monitoring research developments, and national healthcare systems like the UK’s National Health Service (NHS) are adapting their genetic testing protocols to include RNU2-2 analysis. Access to genetic testing remains a challenge in many low- and middle-income countries, highlighting the need for international collaboration to ensure equitable access to diagnostic services.
Funding &. Bias Transparency
This research was primarily funded by the Wellcome Trust, Genomics England, and the National Institutes of Health (NIH). Researchers have disclosed no competing interests. While the study provides strong evidence for the causal role of RNU2-2 variants, further research is needed to fully understand the spectrum of clinical manifestations and to develop effective treatments.
Clinical Presentation and Phenotypic Spectrum
The clinical presentation of RNU2-2 syndrome is variable, ranging from mild learning disabilities to severe epileptic encephalopathy. Common features include hypotonia (low muscle tone), global developmental delay, intellectual disability, and seizures. Brain imaging may reveal cerebral and cerebellar atrophy or white matter abnormalities. The study identified a core set of Human Phenotype Ontology (HPO) terms associated with the disorder, including generalized onset seizures, motor seizures, infantile encephalopathy, and EEG abnormalities. The phenotypic spectrum underscores the importance of comprehensive clinical evaluation and genetic testing for accurate diagnosis.
| Variant Type | Frequency in 100KGP | Associated Phenotypes | U2-2 Expression Level |
|---|---|---|---|
| Homozygous | ~36% of Tier 1 cases | Severe developmental delay, intractable seizures | Significantly reduced |
| Compound Heterozygous | ~60% of Tier 1 & Tier 2 cases | Variable severity, developmental delay, epilepsy | Reduced |
| Monoallelic | Variable | May be asymptomatic or mildly affected | Slightly reduced |
Expert Perspective
“The identification of RNU2-2 as a major genetic cause of NDDs is a significant step forward in our understanding of these complex disorders. It highlights the importance of large-scale genomic studies and international collaboration in unraveling the genetic basis of rare diseases,” says Dr. David Greene, lead author of the study and researcher at Genomics England.
Contraindications & When to Consult a Doctor
Genetic testing for RNU2-2 variants is generally safe, but it is important to consider the potential psychological impact of receiving a positive result. Genetic counseling is strongly recommended before and after testing. There are no specific contraindications to testing, but individuals with a family history of NDDs or those experiencing unexplained developmental delays should consult with a qualified medical geneticist. Symptoms warranting immediate medical attention include prolonged or uncontrolled seizures, severe developmental regression, and unexplained neurological deterioration.
Future Directions and Therapeutic Prospects
While there is currently no cure for RNU2-2 syndrome, ongoing research is focused on developing potential therapeutic strategies. These include gene therapy approaches to restore RNU2-2 expression, small molecule drugs to modulate splicing activity, and personalized therapies based on individual genetic profiles. Longitudinal studies are needed to track the natural history of the disorder and to evaluate the efficacy of novel treatments. The discovery of RNU2-2 as a major cause of NDDs provides a crucial foundation for future research and clinical advancements.
References
- Greene, D. Et al. Biallelic variants in RNU2-2 cause the most prevalent known recessive neurodevelopmental disorder. Nat Med. 2026; (Published online March 26, 2026). https://www.nature.com/articles/s41588-026-02539-5
- Genomics England. The National Genomics Research Library v5.1. Figshare. 2025. https://doi.org/10.6084/m9.figshare.4530893
- National Human Genome Research Institute. (n.d.). RNA splicing. https://www.genome.gov/genetics-glossary/RNA-Splicing
- Centers for Disease Control and Prevention (CDC). (2023). Neurodevelopmental Disabilities. https://www.cdc.gov/ncbddd/developmentaldisabilities/index.html
- European Medicines Agency (EMA). (n.d.). Genetic testing. https://www.ema.europa.eu/en/human-regulatory/research-development/scientific-advice/genetic-testing